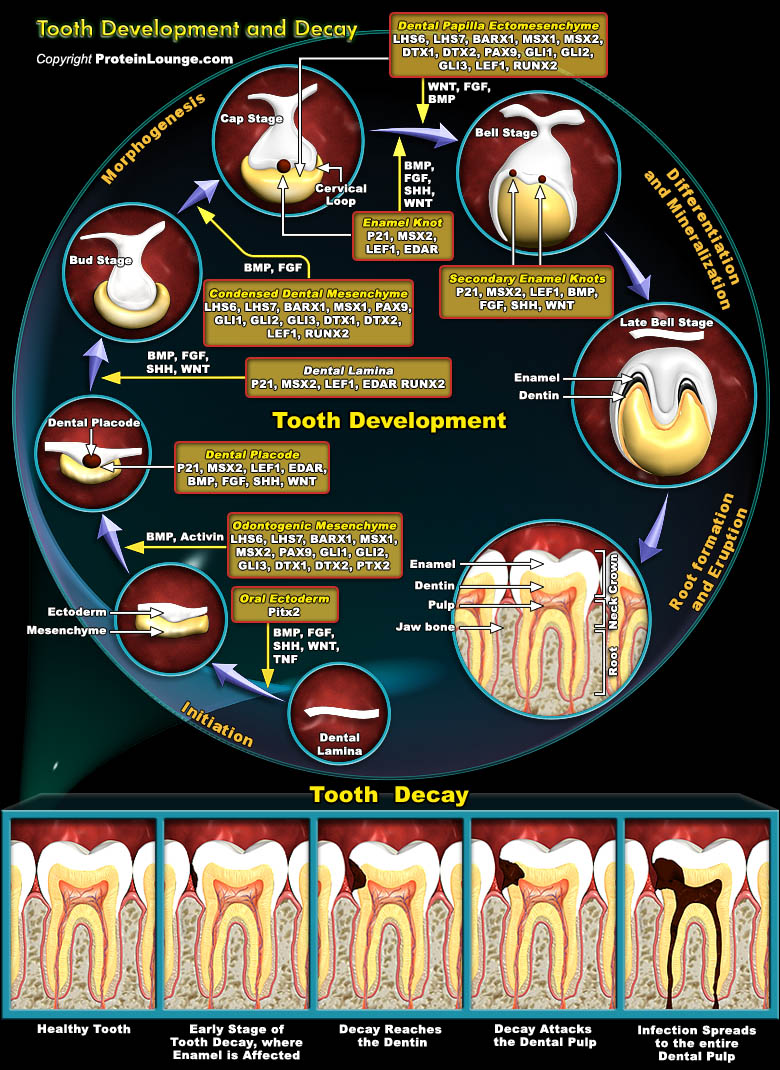
Teeth develop as ectodermal appendages in vertebrate embryos, and their early development resembles morphologically as well as molecularly other organs such as Hairs and Glands. Interactions between the Ectoderm and underlying Mesenchyme constitute a central mechanism regulating the morphogenesis of all these organs. Central features of Tooth morphogenesis are the formation of the Epithelial Placode, the budding of the Epithelium, the condensation of Mesenchyme around the bud, and the folding and growth of the Epithelium generating the shape of the Tooth Crown. The mineralized structures characteristic for Teeth, which is, Dentin and Enamel, are formed by specialized cells, the Odontoblasts and Ameloblasts differentiating from the Mesenchyme and Epithelium,[..]
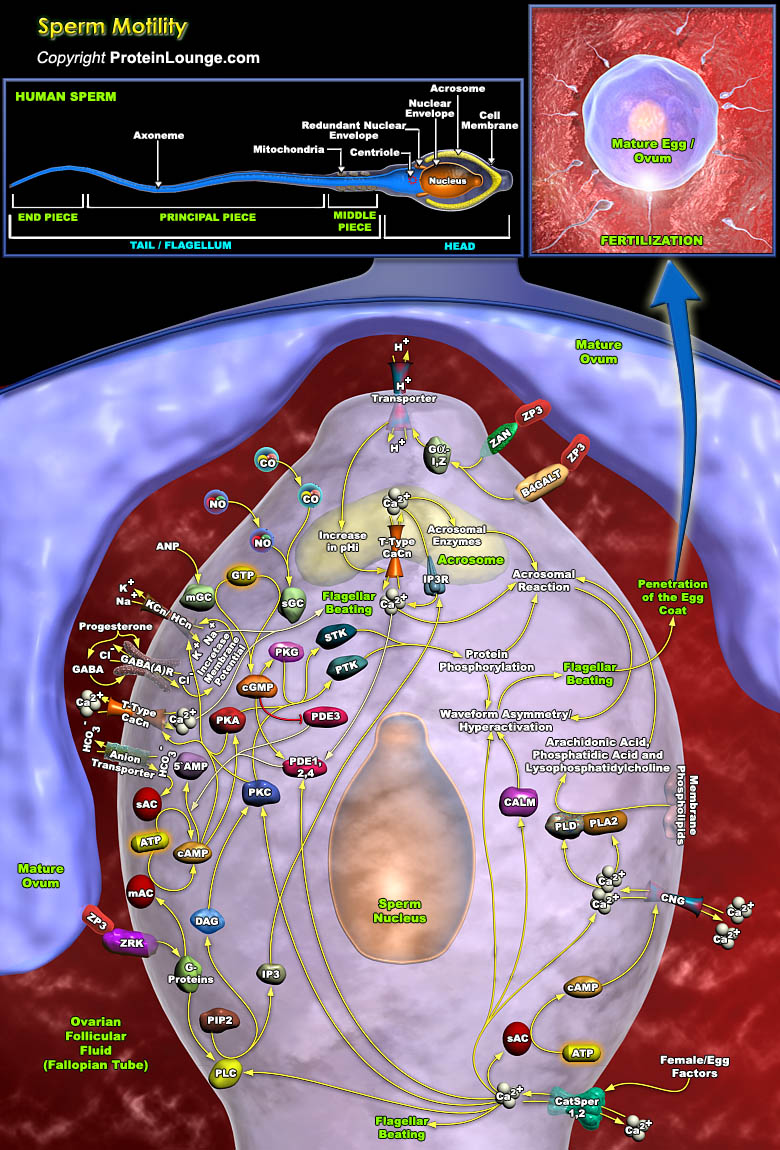
In humans, roughly 300 million spermatozoa on average are ejaculated in the female reproductive tract, but only about one of every million actually enters the Fallopian tube. Upon entry, these spermatozoa apparently bind strongly to the oviductal epithelium in the isthmus, forming a sperm storage site. Human spermatozoon formed in the testes via spermatogenesis is morphologically complete but functionally immature and incapable of fertilizing an egg. Spermatozoa to become fertile must undergo maturation in the epididymis followed by capacitation, and acrosome reaction in the female reproductive system. For reaching the oocyte at the fertilization site, the limited number of capacitated spermatozoa in the isthmus has to swim a long way full of obstacles. This[..]
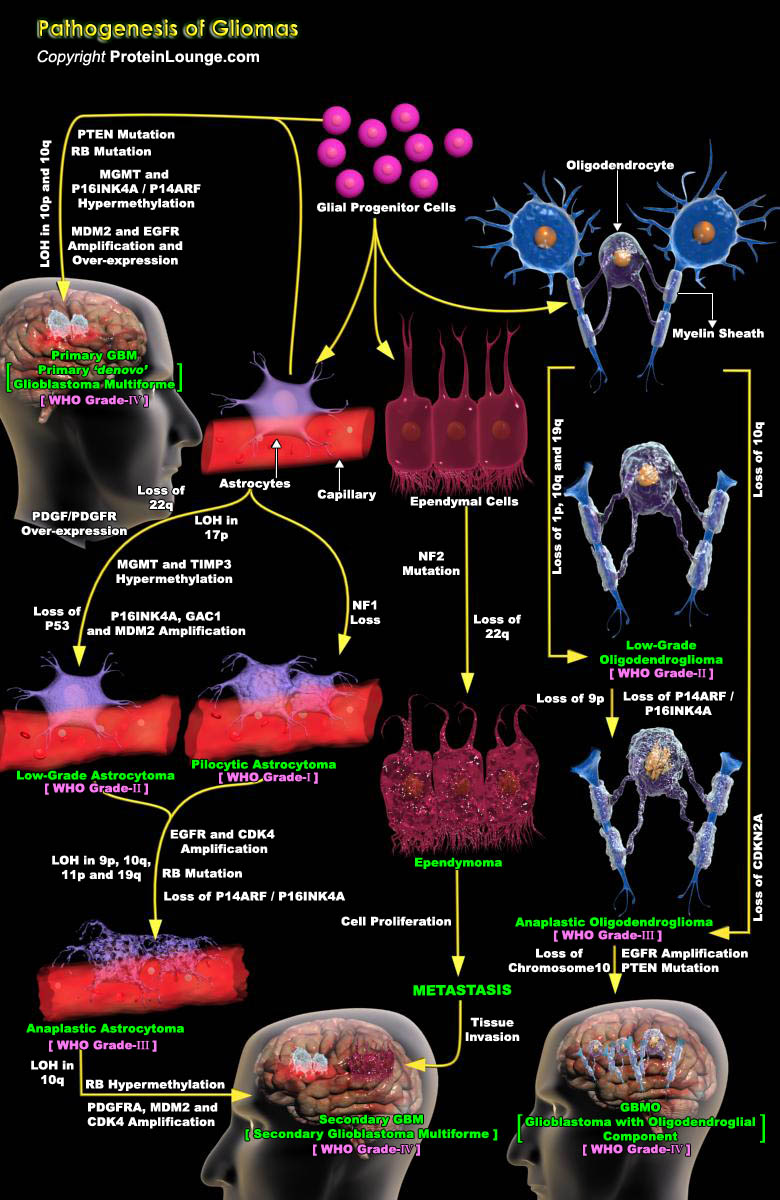
Tumors of the Central Nervous System (CNS) are devastating as they are difficult to treat and may cause grave disability or death. CNS Gliomas pose particularly difficult problems because of their tendency toward malignancy, rate of tumor spread, and the lack of effective therapy. Gliomas are the most common intracranial malignant tumors in humans (Ref.1). In vertebrates, the embryonic neural tube (neuroectoderm) gives rise to the main cell types of the CNS, including neurons, astrocytes, oligodendrocytes and ependymal cells. The neural crest derives from the dorsal lip of the neural tube, and its cells migrate extensively during embryonic development, giving rise to various tissues, including the Peripheral Nervous System (PNS). In both the CNS and PNS, the appearance[..]
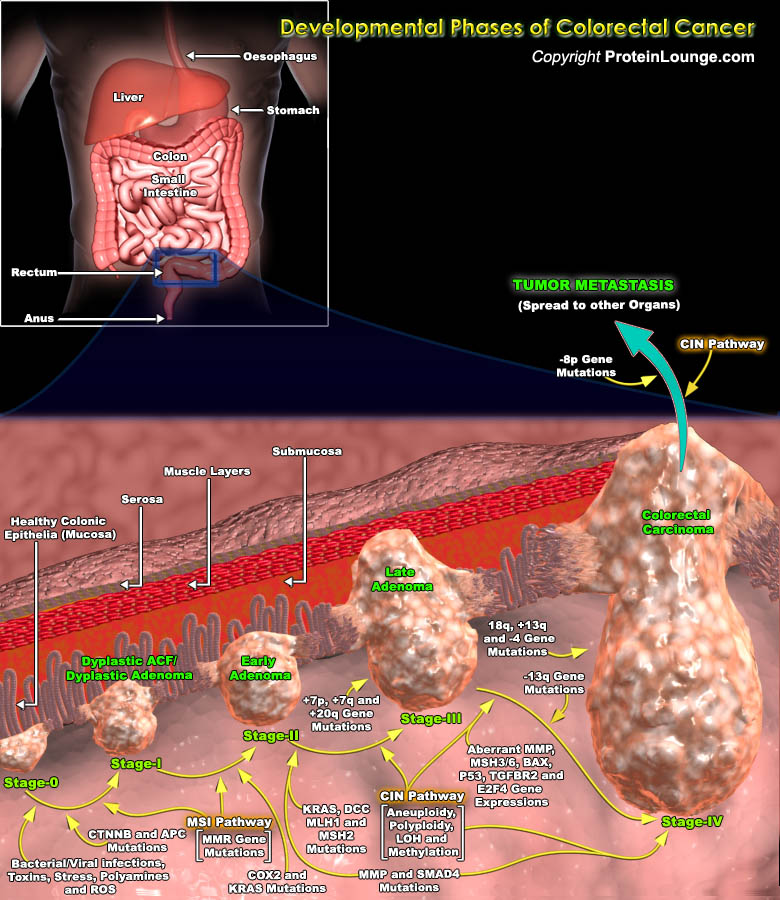
Colorectal tumors arise as a result of the mutational activation of oncogenes coupled with the mutational inactivation of tumor suppressor genes without a major role for gene amplification or rearrangement. These tumors affect the colon and rectum, and most colorectal cancers arise from adenomatous polyps. The development of colorectal neoplasms is characterized by an ordered series of events that are referred to as the “Dysplasia-Carcinoma Sequence” or “Adenoma-Carcinoma Sequence”. Mutations in at least four to five genes are required for the formation of malignant tumors and fewer changes only suffice for benign tumorigenesis. Although genetic alterations occur according to a preferred sequence, the total accumulation of changes, rather than[..]
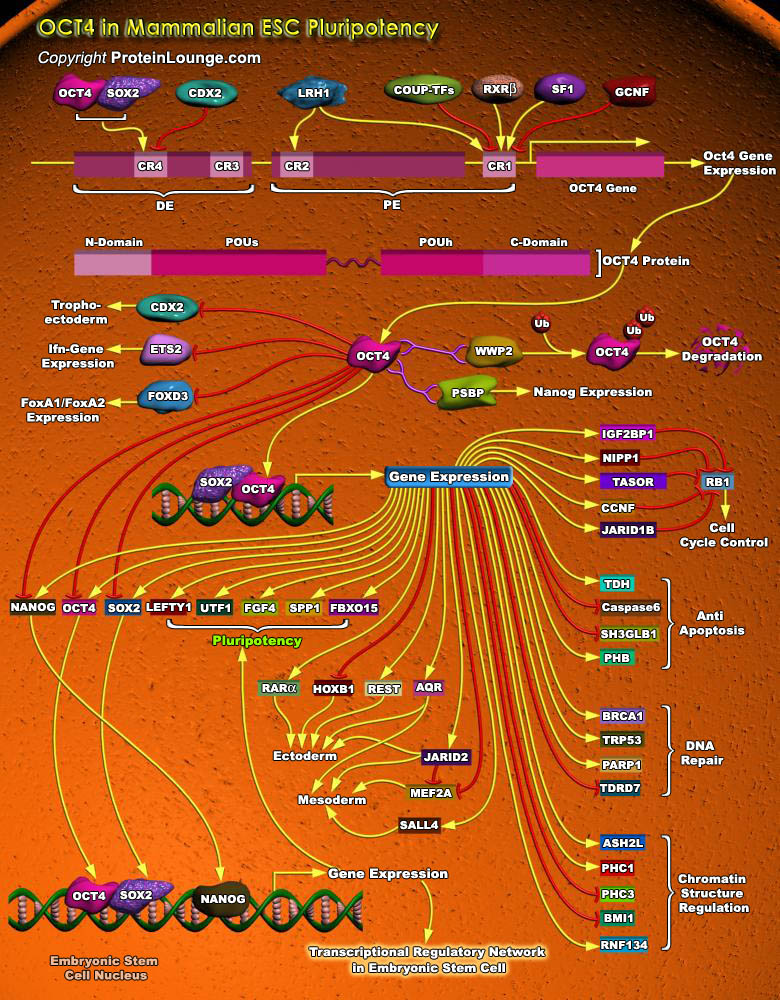
Stem cells are undifferentiated cells capable of producing virtually all cell types in our body. They are characterized by the ability to Self-renew and maintain Pluripotency. For proper developmental outcome, ESCs (Embryonic Stem Cells) must tightly regulate their differentiation status. Hundreds of genes have been identified, including several transcription factors, which have expression patterns tightly correlated with ESC differentiation. Very low number of transcription factors is responsible for regulating the development of an entire organism. These transcription factors form multiprotein complexes on DNA, thereby orchestrating the correct temporal–spatial expression of developmental genes. The process leads to the establishment of functional[..]
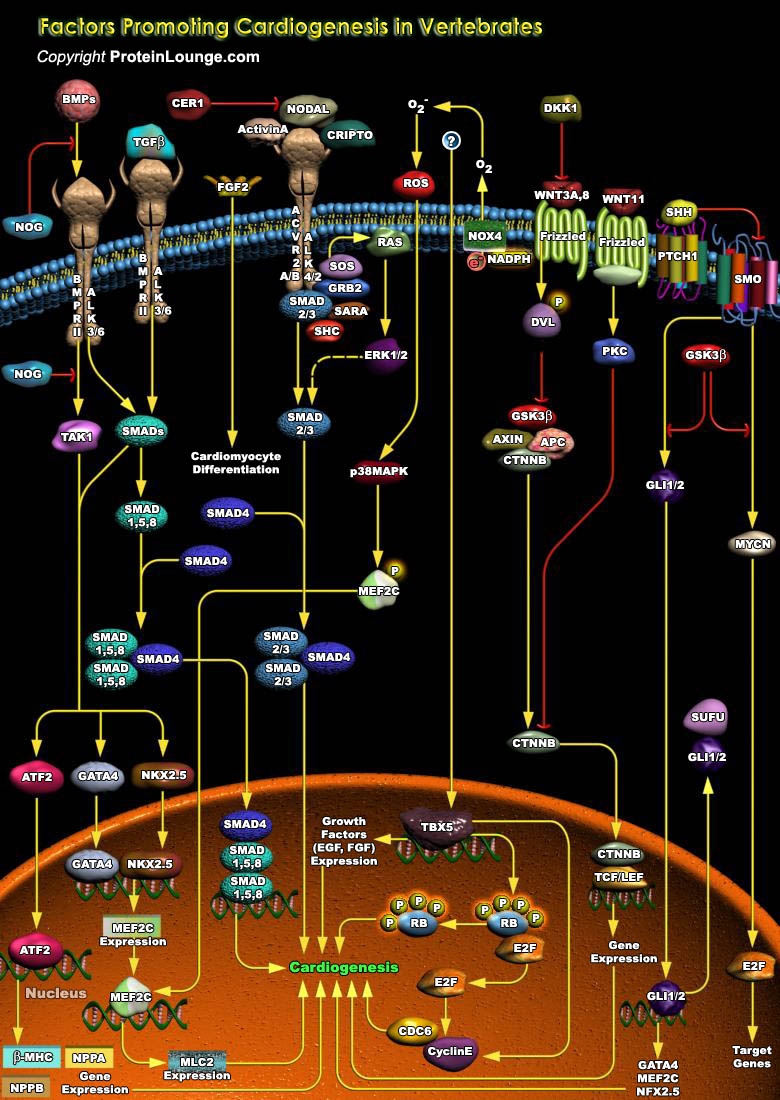
Cardiogenesis is the formation of new heart tissue from embryonic, postnatal, or adult multipotent cardiovascular progenitor cells. Cardiogenesis in vertebrate is a complex process, where various genetic and epigenetic factors play crucial role in driving the interaction between different structures and diverse cell types. Cardiomyocytes are the main cell type found in the heart that are responsible for the contraction of the chambers and efficient blood flow throughout the body (Ref.1 and 2). Multiple transcription factors and extracellular growth factors specify the Cardiac lineage in mesodermal progenitor cells. The earliest expressed transcription factors that initiate Cardiac fate are the homeobox transcription factor NKX2.5 (NK2 Transcription Factor Related[..]
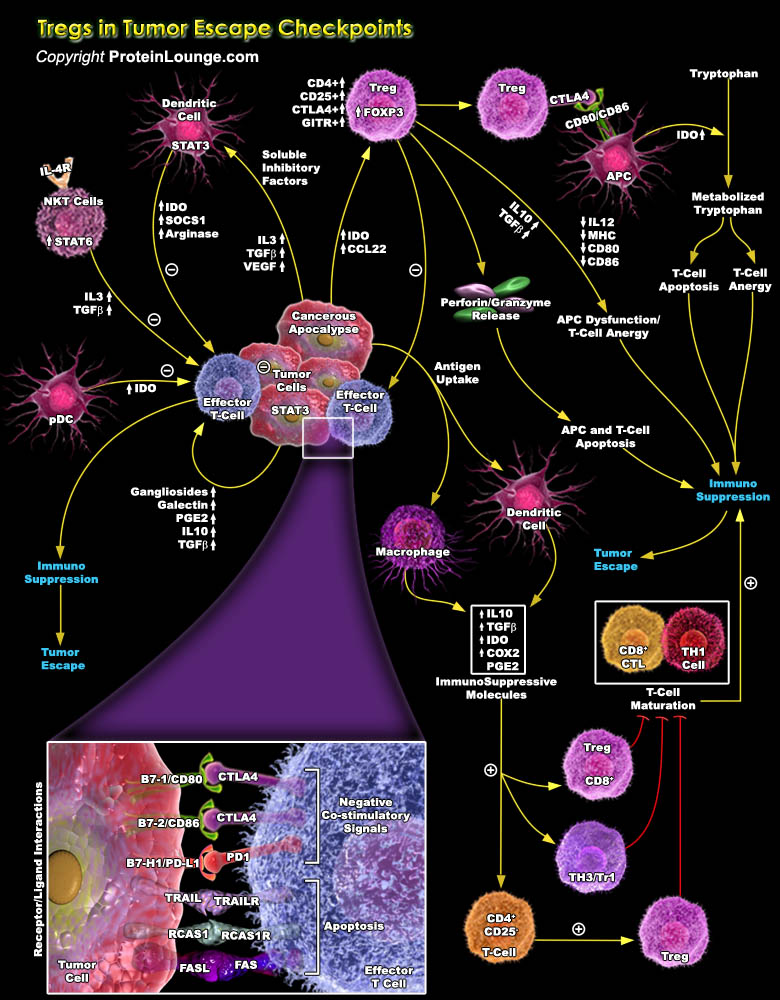
Specific T cell populations have suppressive/regulatory cells known as Regulatory T-cell or Tregs (Previously known as suppressor T-cell). Among them CD4+ regulatory T cells (Tregs) basically has two different subsets Tr1 and Th3 cells which are differentiated by their distinct suppressive mechanisms. The thymus-derived Tregs or natural Tregs (nTregs) express CD4 and high CD25 with FOXP3 (Forkhead Box-P3) to be a key regulatory gene. Apart from these, other markers of Treg include CTLA4 (Cytotoxic T-Lymphocyte-Associated protein-4), GITR (Glucocorticoid-Induced TNFR-Related protein) and LAG-3 (Lymphocyte Activation Gene-3). Treg cells are implicated in the development of autoimmunity, allergy and rejection of organ transplants, as well as the suppression of immune[..]
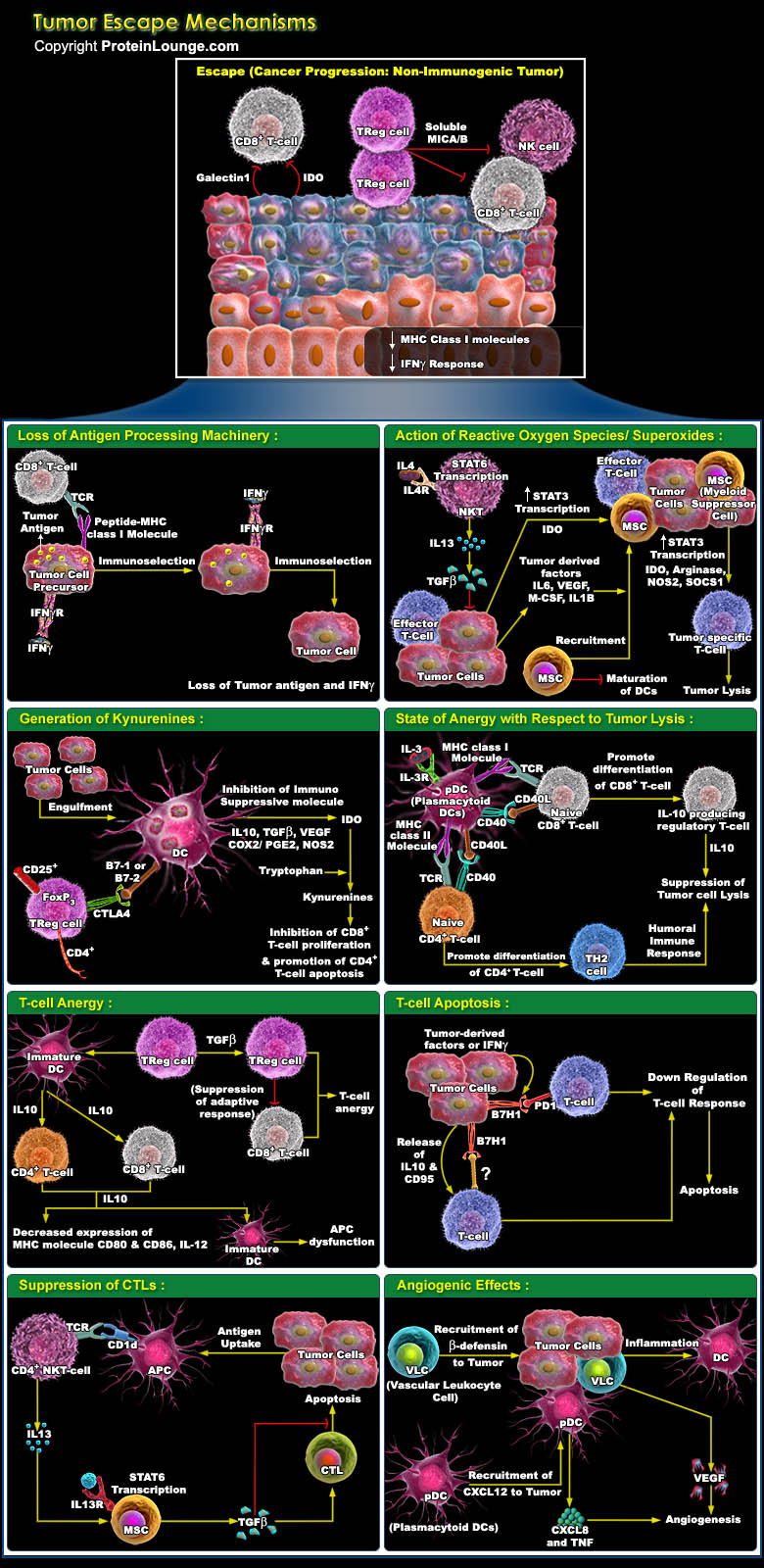
Tumour cells characteristically provides their own growth signals, and ignore growth-inhibitory signals, avoid cell death, replicate without limits, sustain angiogenesis, and invade tissues through basement membranes and capillary walls. Whereas cancer immunosurveillance predicts that the immune system can recognize precursors of cancer and, in most cases, destroy these precursors before they become clinically apparent. But sometimes tumour cells use to escape these innate and adaptive immune responses by immunoselection (that is, selection of non-immunogenic tumour-cell variants, a process that is also known as immunoediting) or by immunosubversion (that is, active suppression of the immune response) and heads towards a difficult situation which is refer as cancer.[..]
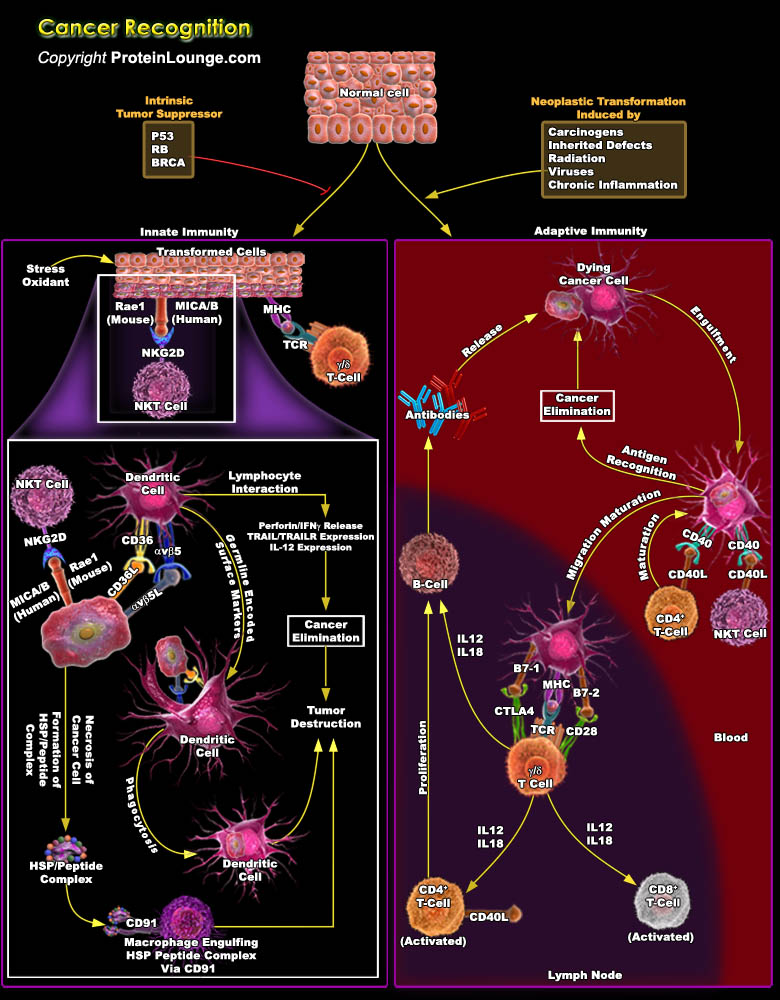
Cancer immunotherapy attempts to exploit the exquisite power and specificity of the immune system for the treatment of malignancy. Although cancer cells are less immunogenic than pathogens, the immune system is clearly capable of recognizing and eliminating tumour cells. However, tumors frequently interfere with the development and function of immune responses. Thus, the challenge for immunotherapy is to use advances in cellular and molecular immunology to develop strategies that effectively and safely augment antitumour responses. By contrast, the immune system has evolved strategies, largely in response to infections, to efficiently search for and specifically destroy diseased targets. Advances in cellular and molecular immunology have provides enormous insights in[..]
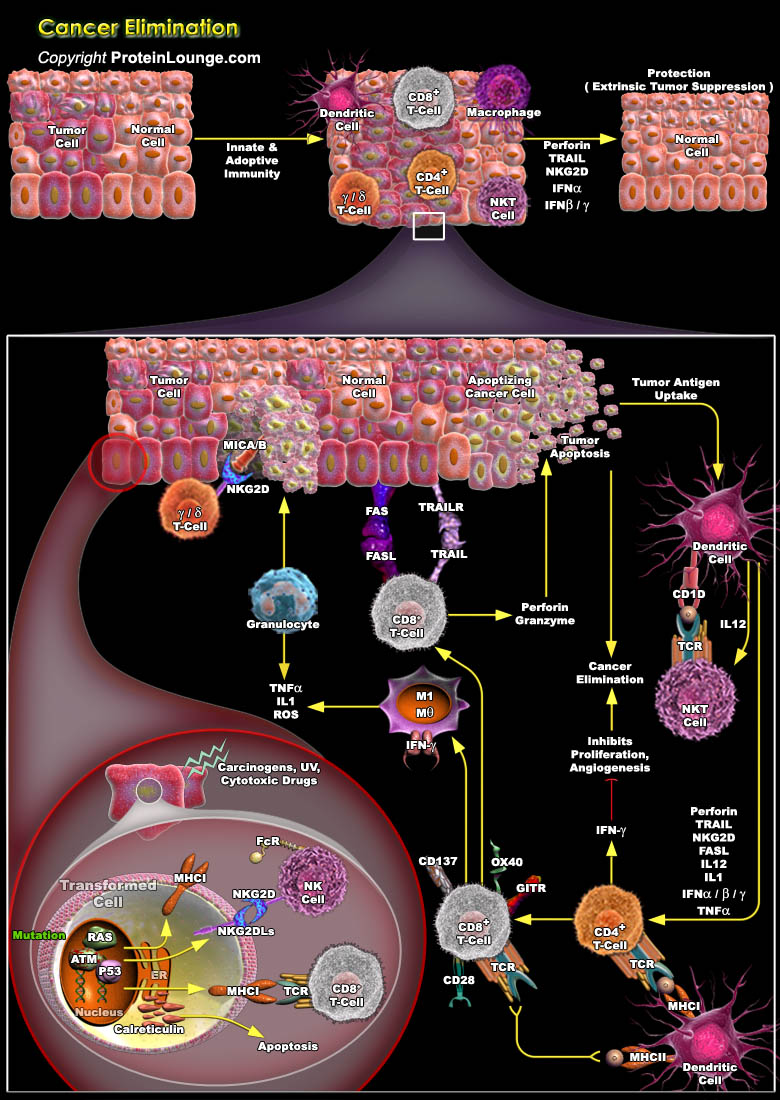
The immune system has three primary roles in the prevention of tumors. First, the immune system can protect the host from virus-induced tumors by eliminating or suppressing viral infections. Second, the timely elimination of pathogens and prompt resolution of inflammation can prevent the establishment of an inflammatory environment conducive to tumorigenesis. Third, the immune system can specifically identify and eliminate tumor cells on the basis of their expression of tumor-specific antigens or molecules induced by cellular stress. The evolution of the cancer immunoediting concept from the older and perhaps more controversial ‘cancer immunosurveillance’ hypothesis has helped interpret the predictive and prognostic significance of immune infiltrates into[..]
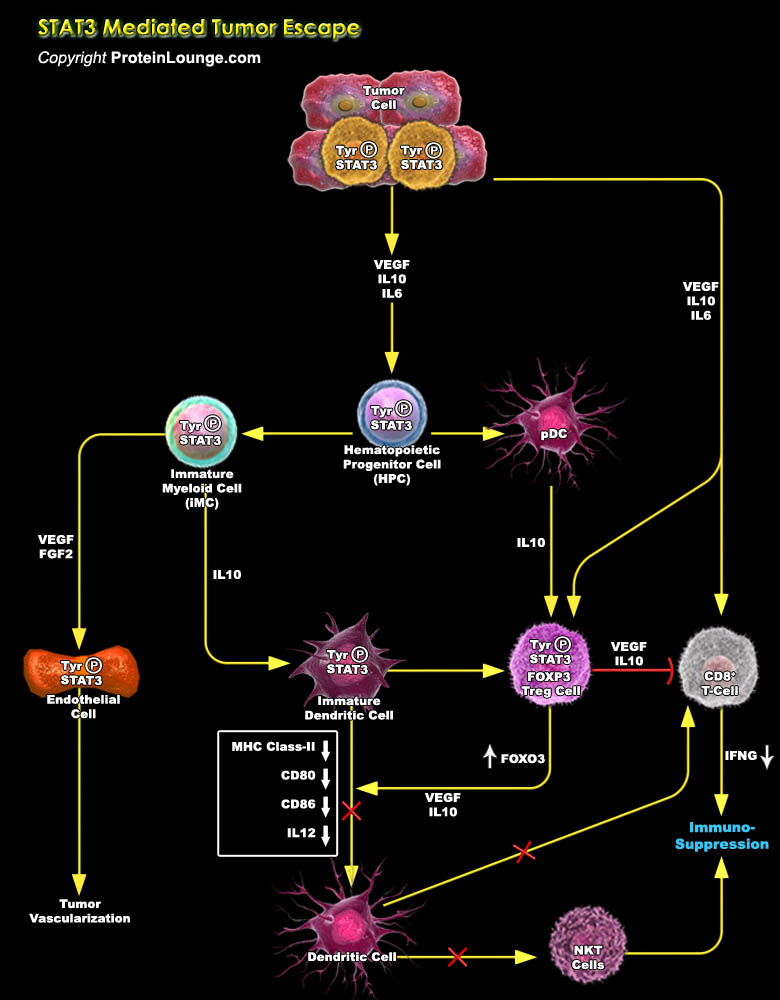
Immune cells in the tumour microenvironment not only fail to mount an effective anti-tumour immune response, but also interact intimately with the transformed cells to promote oncogenesis actively. STAT (Signal Transducer and Activator of Transcription) proteins act as a mediator of cytokine receptor signaling. This protein plays a role in transmitting the signals of growth factor receptors and is both cytoplasmic signaling molecules and nuclear transcription factors that activate diverse genes. In normal cells, STAT activation is transient and tightly regulated. While STAT3 (Signal Transducer and Activator of Transcription 3) acts as a point of convergence for numerous oncogenic signalling pathways, and constitutively remain active both in tumour[..]
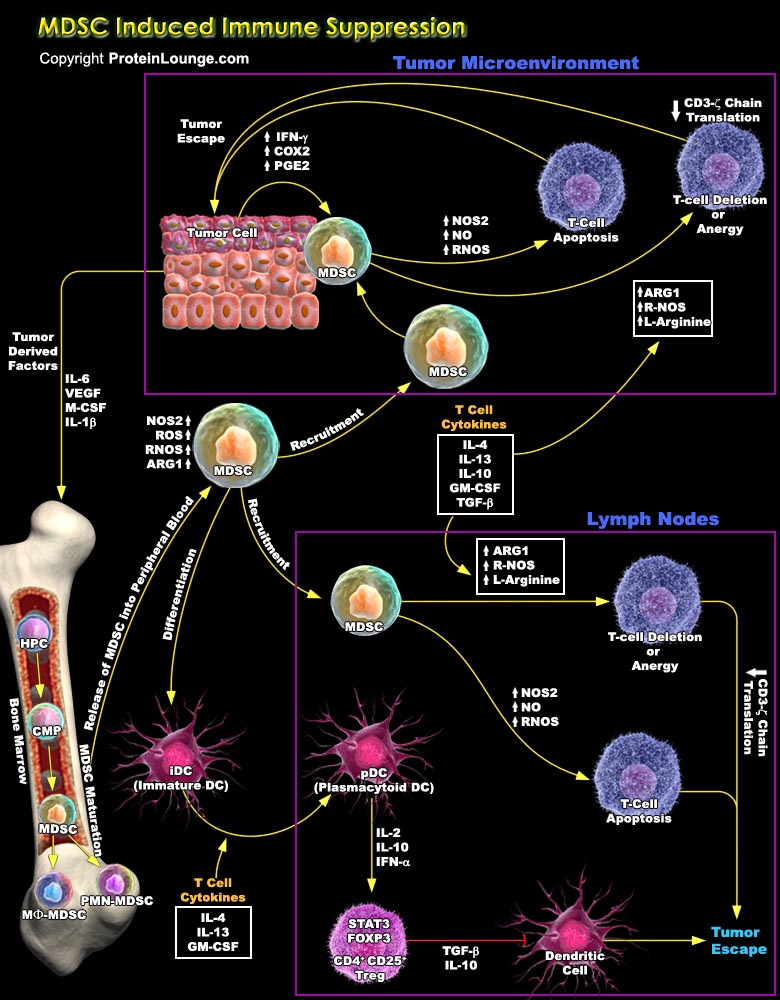
MDSCs (Myeloid-Derived Suppressor Cells) are recently been recognized as critical mediators of tumor progression in numerous solid tumours through their inhibition of tumor-specific immune responses. These cells are increased in numerous pathologic conditions, including infections, inflammatory diseases, graft-versus-host disease, traumatic stress, and neoplastic diseases. MDSCs inhibit not only activation of T cells by anti-CD3 and super antigen, but also antigen-specific CD4+ and CD8+ T-cell responses. The mechanisms of MDSC immunosuppression are diverse, it includes up-regulation of ROS (Reactive Oxygen Species), NO (Nitric Oxide), and L-Arginine metabolism. It also facilitates tumour-induced immune suppression and tumour progression by inducing the[..]

