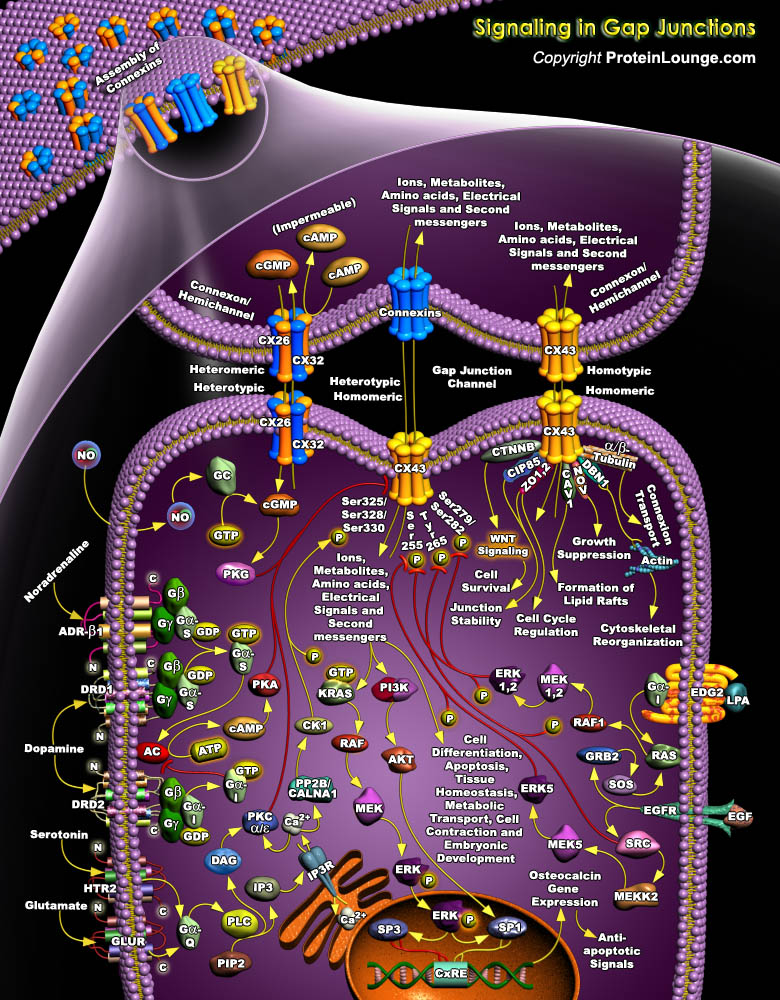
Gap Junction (GJ) channels span two plasma membranes and are formed by the alignment of two hemichannels, each consisting of an oligomer of structural subunit proteins, called Cxs (Connexins). These junctional proteins constitute a multigene family whose members are distinguished according to their predicted molecular weight in kilodaltons. A Connexin structure consists of two extracellular loops (EL), four membrane-spanning domains (TM), one cytoplasmic loop (CL), one N-terminal tail (NT), and one C-terminal tail (CT) (Ref.1 & 2). During intercellular channel formation, six Connexins oligomerize into a Connexon or hemichannel that docks in homotypic, heterotypic and combined heterotypic/heteromeric arrangements. In total, as many as 14 different Connexon[..]
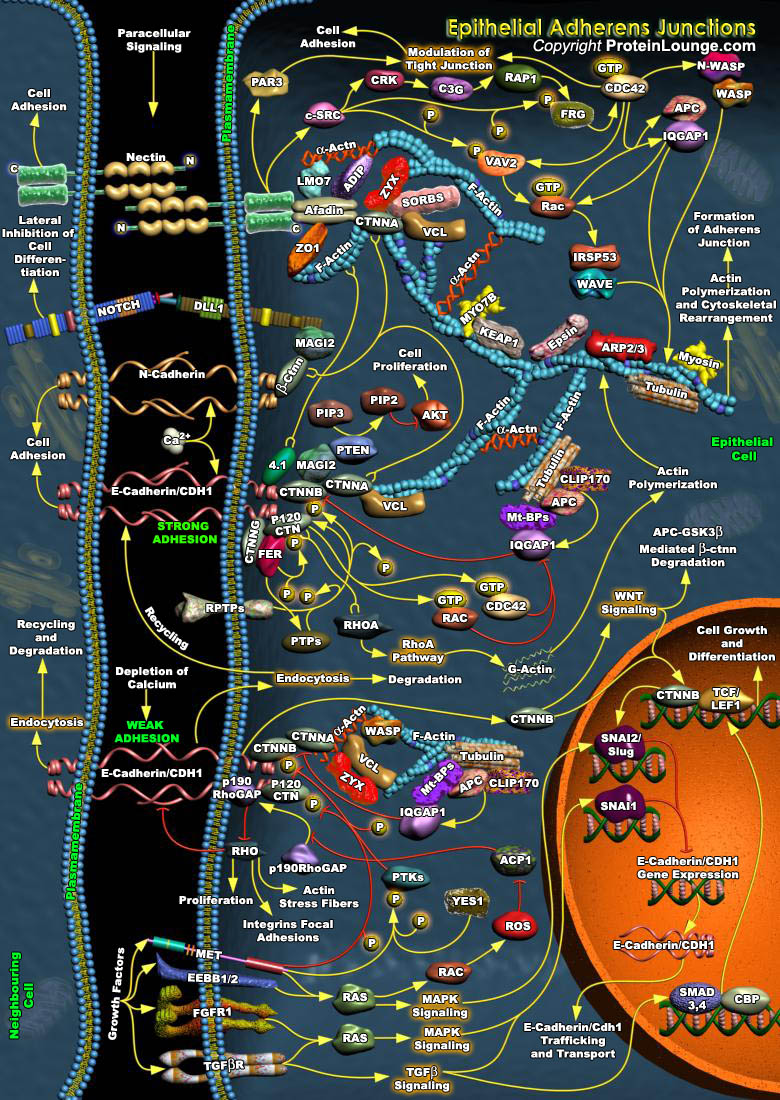
Adhesion between neighboring epithelial cells is a crucial and tightly controlled process. The integrity of cell-cell contacts is essential for the regulation of electrolyte absorption and for the prevention of tumor metastasis. In polarized epithelia, specialized structures such as Adherens Junctions (AJs) and Tight Junctions (TJs) are responsible for the establishment of contacts between neighboring cells. The establishment and stability of Adherens Junction is tightly regulated-in particular, by Growth Factors, cytokines and hormones. Such regulation, although poorly understood, is quite essential for the modulation of paracellular permeability in various epithelia, for the epithelium mesenchyme transition, and for development, morphogenesis and wound healing[..]
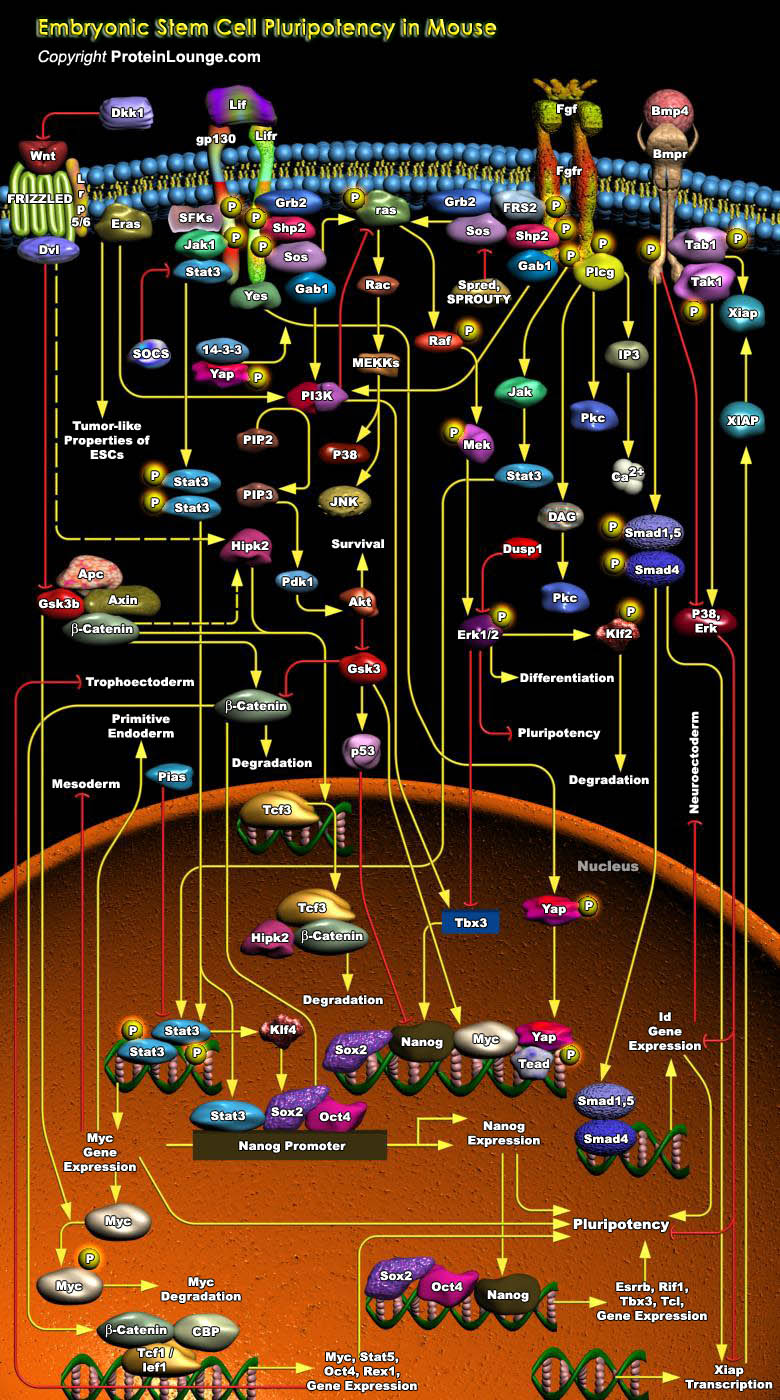
ESCs (Embryonic Stem Cells) are a population of Pluripotent, Self-renewing cells which can proliferate indefinitely and contribute to the formation of basically all cell types in vitro and in vivo. The study of mammalian ESCs, especially Mouse ESCs, has provided valuable insights into early embryogenesis in mammals. Mouse ES cells are derived mainly from the ICM (Inner Cell Mass) of the Mouse Blastocyst embryos and retain this developmental identity even after prolonged culture in vitro. Both cell Extrinsic and Intrinsic factors regulate Mouse ESC Self-renewal and Pluripotency. Cell extrinsic factors include LIF (Leukaemia Inhibitory Factor) and BMP (Bone Morphogenic Protein), which signal through STAT (Signal Transducer and Activator of Transcription) and SMAD (Sma[..]
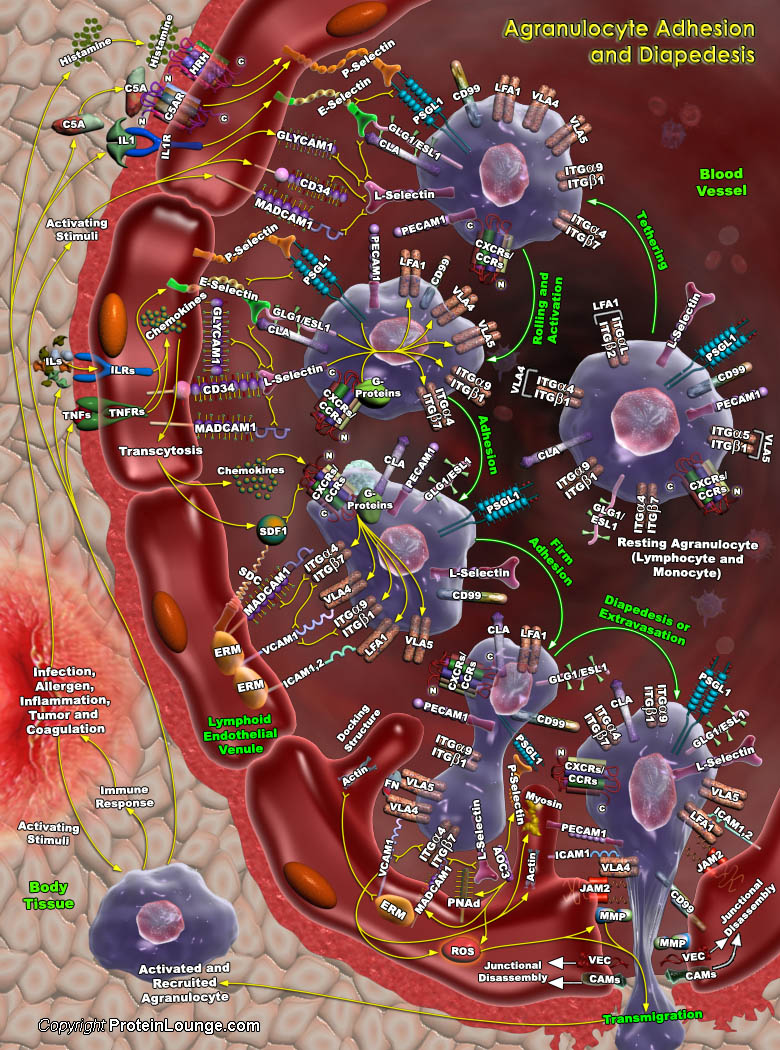
Cell adhesion and migration is the fundamental feature of multicellular organisms during defense mechanisms, where leukocytes play the central role. They bind bacteria, parasites, viruses, tumor cells etc. Furthermore, their interactions with the endothelium are of special importance. The migration of leukocytes or WBCs (White Blood Cells) from the vascular system to sites of pathogenic exposure is a key event in the process of inflammation. Generally, agranulocyte (which includes Lymphocytes and Monocytes) adhesion and passage from the bloodstream to the lymphatic system occurs in the high endothelial venules of the lymphnodes, also known as the lymphoid endothelial venule. This way the agranulocytes communicate with each other in the lymphatic system and search for[..]
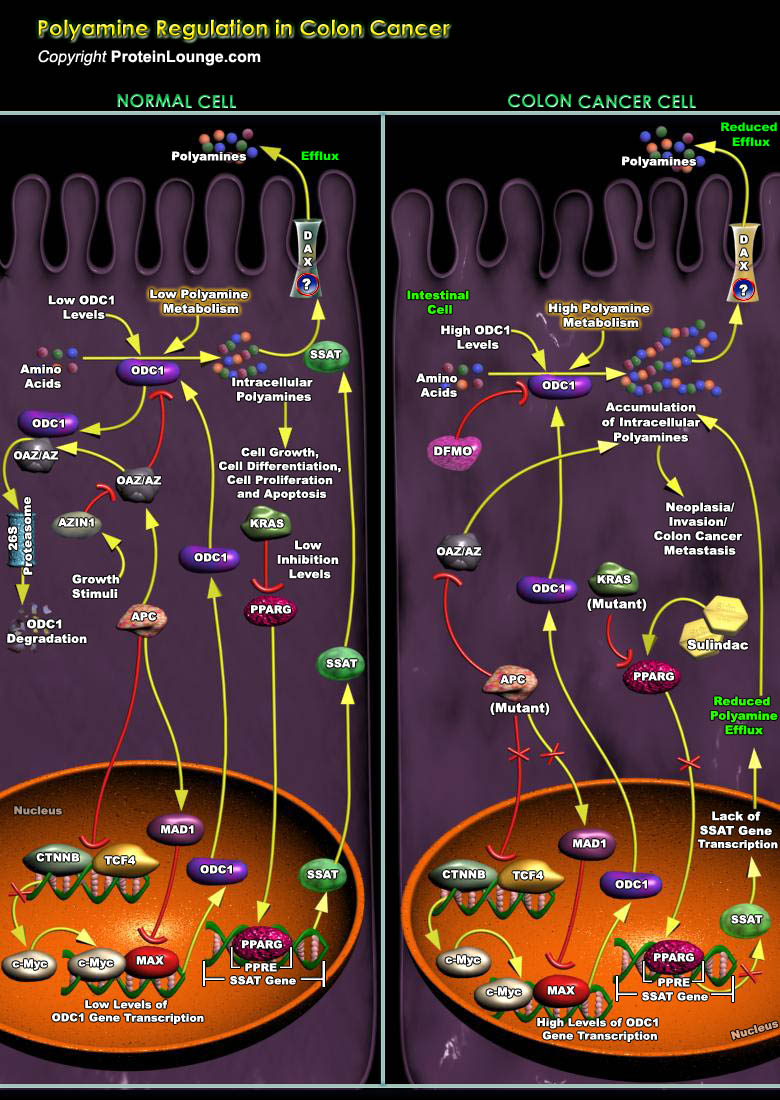
Polyamines are vital for the growth and function of normal cells. The complexity of polyamine metabolism and the multitude of compensatory mechanisms that are invoked to maintain polyamine homoeostasis argue that these amines are critical to cell survival. The regulation of polyamine content within cells occurs at several levels, including transcription and translation (Ref.1). The amino-acid derived polyamines like Putrescine, Spermidine and Spermine that are the main polyamines found in prokaryotes and eukaryotes, have long been associated with cell growth and cancer, and specific oncogenes and tumor suppressor genes regulate polyamine metabolism. Regulation of these polyamines (otherwise known as “organic cations”) is chiefly associated with cancers[..]
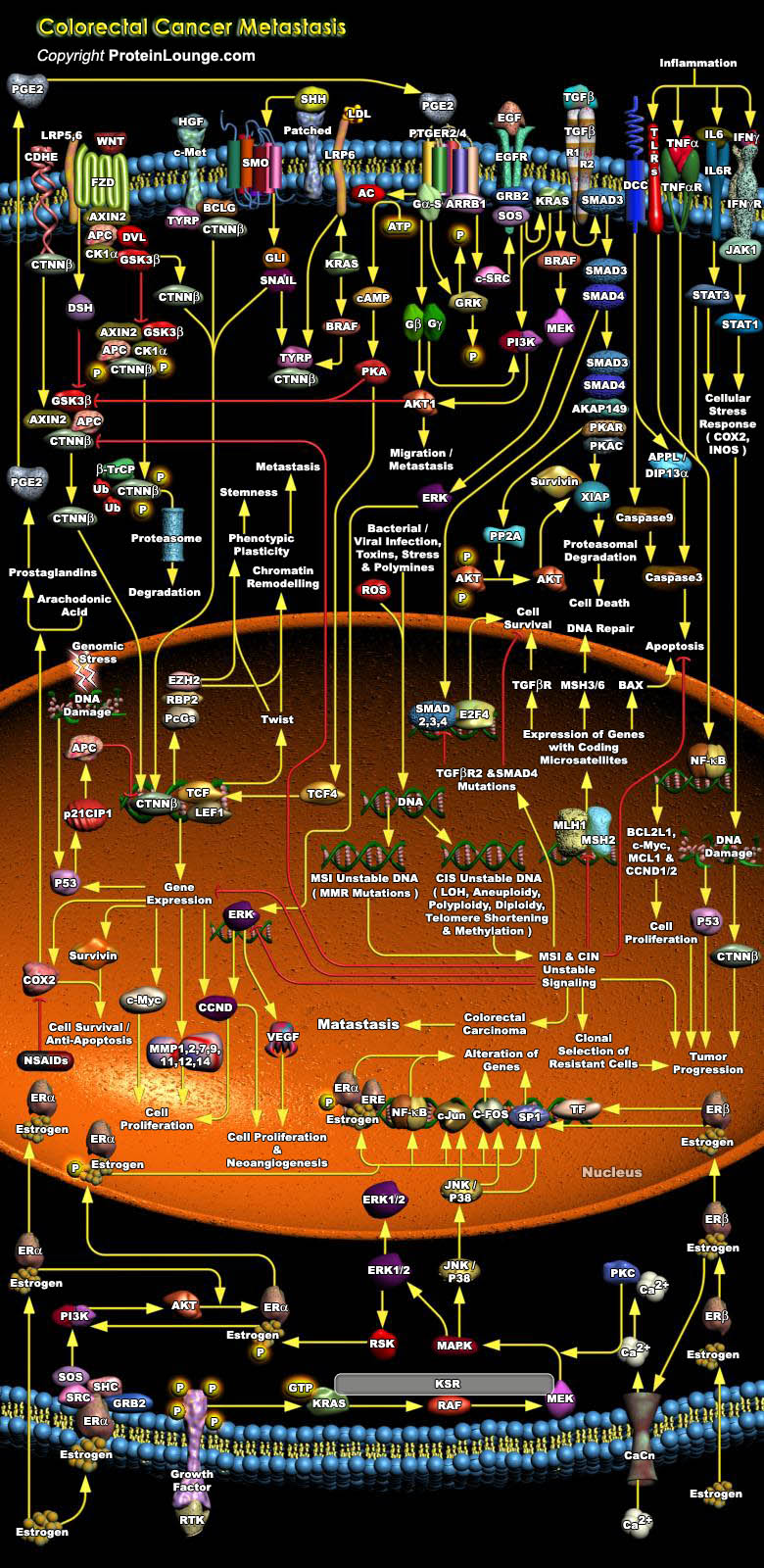
Colorectal cancer represents a relatively well-characterized tumorigenesis paradigm and colorectal carcinoma is one of the leading causes of cancer-related death. Colorectal cancer results from the accumulation of genetic alterations. Genomic instability creates a permissive state in which a potential cancer cell is allowed to acquire enough mutations to become a cancer cell. Several forms of genomic instability are common in colon cancer: MSI (Microsatellite Instability), CIN (Chromosome Instability), and chromosome translocations. MSI occurs in approximately 15% of colon cancers and results from inactivation of the MMR (Mismatch Repair) system secondary to either MMR gene mutations or hypermethylation of MMR gene promoters. It promotes tumorigenesis by generating[..]
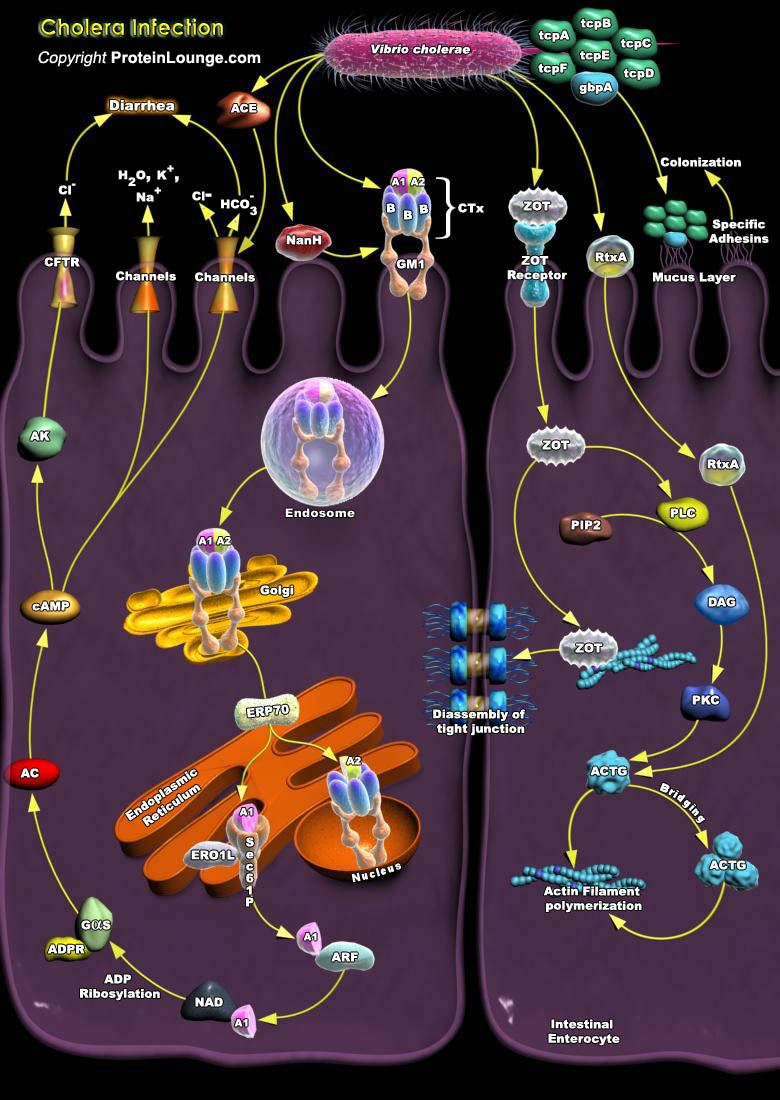
Cholera is one of the most severe diarrheal diseases that affect humans and is responsible for significant morbidity and mortality especially among children in developing countries. It is characterized by numerous, voluminous watery stools, often accompanied by vomiting, and resulting in hypovolemic shock and acidosis. It is caused by certain members of the species Vibrio cholerae which can also cause mild or inapparent infections. Other members of the species may occasionally cause isolated outbreaks of milder Diarrhea whereas others the vast majorities are free-living and not associated with disease. The causative agent of Cholera, Vibrio cholerae, is a Gram-negative highly motile bacterium with a single polar flagellum that inhabits rivers, estuaries or other[..]
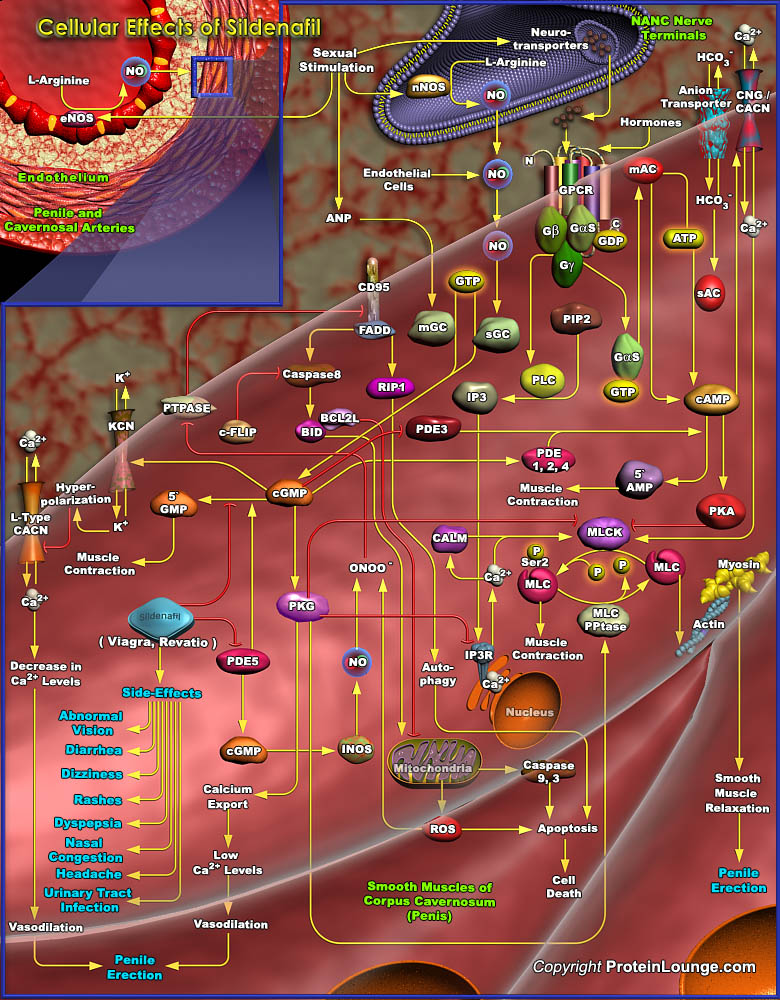
Erectile Dysfunction commonly known as ED or Impotence, affects a large segment of the male population that results in impaired relaxation of the smooth muscle cells in the corpus cavernosum and in the penile arteries. It is an age-related problem that is increasingly common in men aged over 40 years. In the past, ED was often assumed to be either a psychological problem or a normal part of the ageing process, to be tolerated with other signs of aging. However, ED is now known to be primarily organic resulting from vascular, hormonal or neurological complications. The corpus cavernosum (cavernous space) contains sinusoids that are surrounded by trabecular smooth muscle (Ref.1). Upon sexual stimulation, the increase in parasympathetic activity results in dilatation of[..]
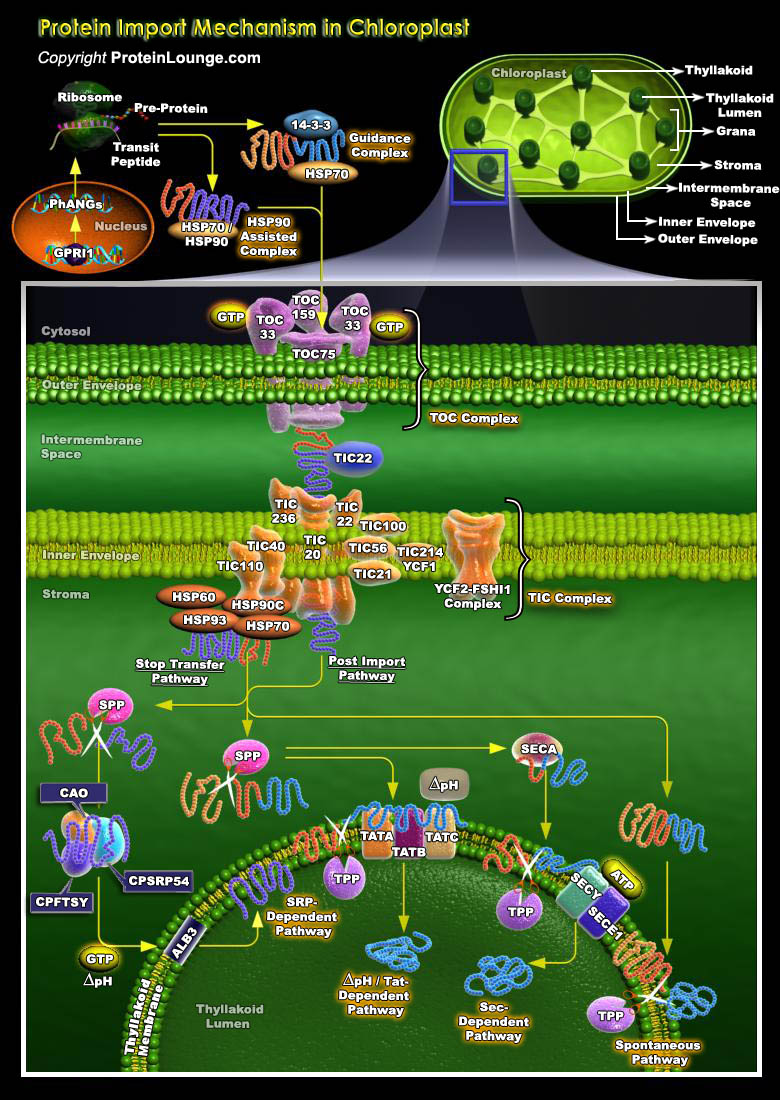
Chloroplasts represent a diverse group of essential organelles collectively as the plastids. Plastids are a heterogeneous family of organelles found ubiquitously in plant and algal cells. Chloroplasts perform a variety of biochemical functions within plant cells. They contain the green pigment chlorophyll and are responsible for the light-harvesting and carbon-fixation reactions of photosynthesis, as well as for the synthesis of many essential metabolites, such as fatty acids and amino acids. Other members of the plastid family include the Amyloplasts( contain large quantities of starch and play important roles in energy storage and gravitropism), the Chromoplast ( accumulate the red-orange-yellow), Carotenoid (pigments that act as attractants in flowers and[..]
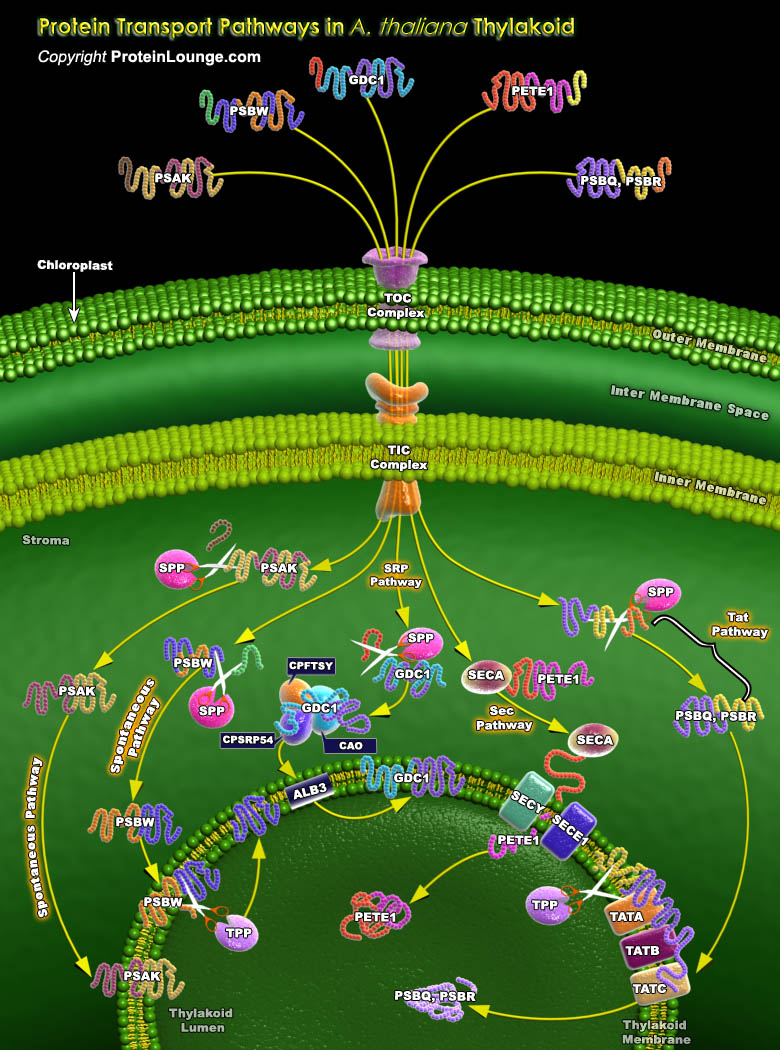
Chloroplasts are exceptionally complex organelles found ubiquitously in Plant and Algal cells. Chloroplasts contain at least six suborganellar compartments: Outer and Inner membranes, Intermembrane Space, Stroma, and Thylakoid membrane and Lumen, all of which require specific trafficking systems. The Thylakoid membrane of the Chloroplast accounts for the bulk of the Chloroplast lipid content and contains the abundant proteins associated with light capture and photosynthetic electron transport.The targeting systems of the Thylakoid appear to be conserved from the Protein export systems that exist in Gram-negative bacteria. The Thylakoid membrane contains four especially prominent complexes (Photosystems I and II, Cytochrome b/f complex and the ATP Synthase), each of[..]
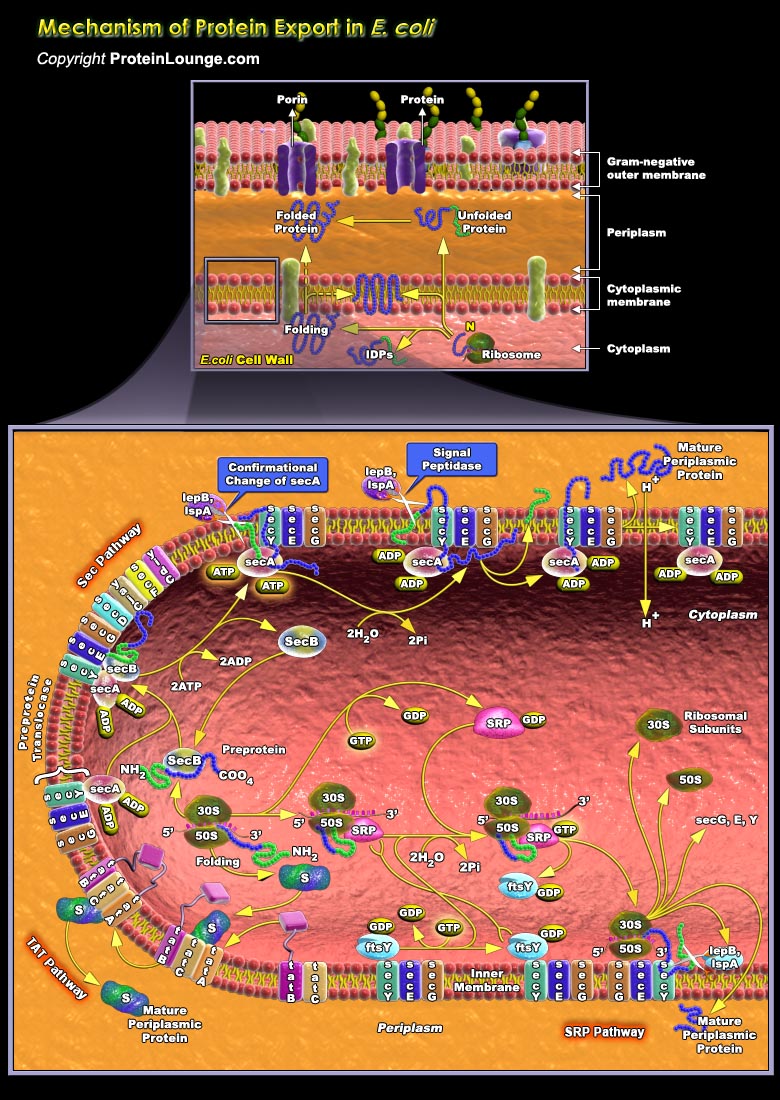
The translocation of proteins from one compartment to another is an essential feature of cellular life. The proper functioning of extracytoplasmic proteins requires their export to, and productive folding in, the correct cellular compartment. Gram-negative bacteria secrete a wide range of proteins whose function includes biogenesis of organelles, such as Pili and flagella; nutrient acquisition; virulence; and efflux of drugs and other toxins. Export of these proteins to the bacterial surface involves transport across the IM (Inner Membrane), Periplasm, and OM (Outer Membrane) of the cell envelope. All proteins in Escherichia coli are initially synthesized in the cytoplasm then follow a pathway that depends upon their ultimate cellular destination. Many proteins[..]
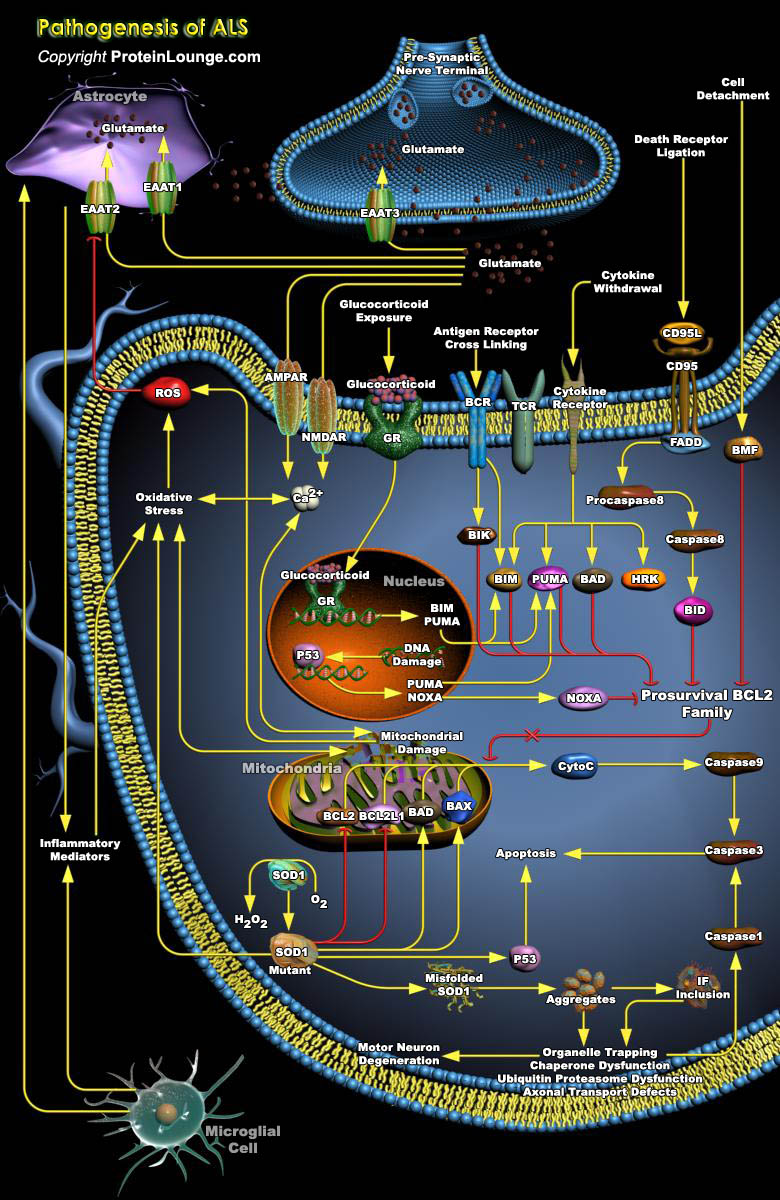
ALS (Amyotrophic Lateral Sclerosis), also known as Lou Gehrig’s disease, is a devastating neurodegenerative disorder, which is characterized by the selective degeneration of upper and lower motor neurons, the large nerve cells connecting the brain to the spinal cord and from the spinal cord to muscles, which control muscle movement. The loss of motor neurons leads to progressive atrophy of skeletal muscles. ALS is a relentless disease that manifests as progressive decline in muscular function resulting in eventual paralysis, speech deficits and, ultimately, death due to respiratory failure in the majority of ALS patients within 2 to 5 years of clinical onset. In spite of its notoriety, the mechanisms underlying ALS remain obscure and therapies with long-term[..]

