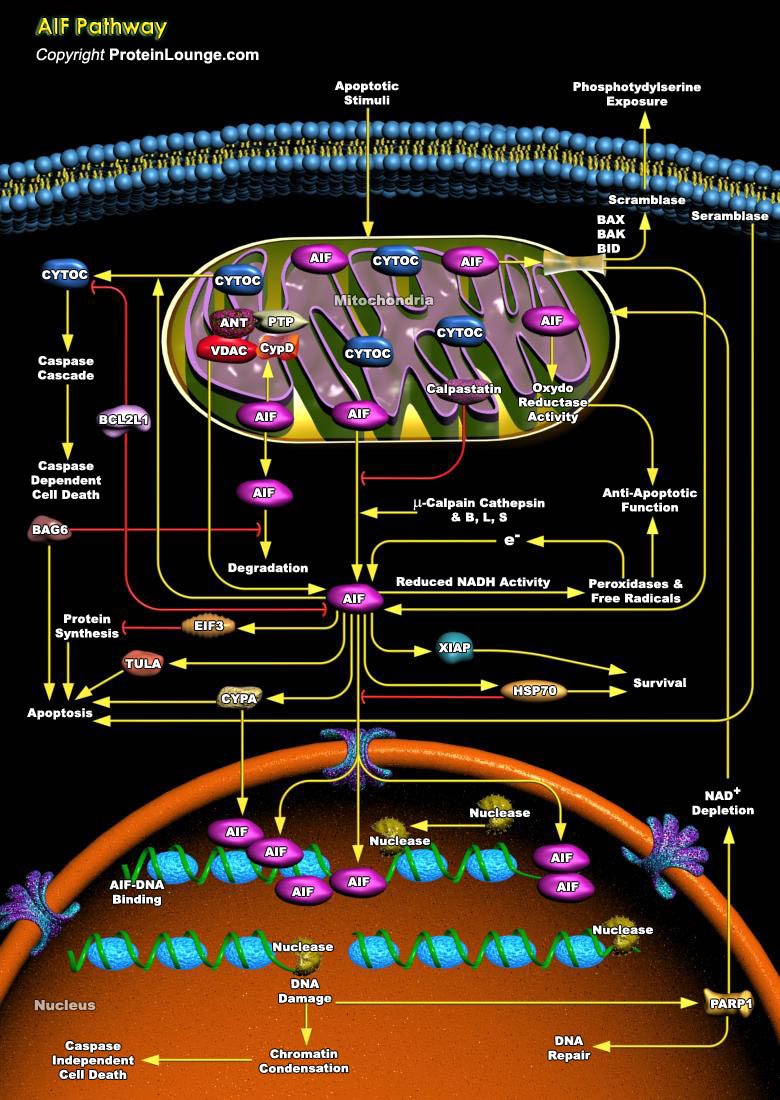
AIF (apoptosis-inducing factor), is a phylogenetically old flavoprotein which is confined to the mitochondrial intermembrane space in healthy cells but upon lethal signaling, it translocates, via the cytosol, to the nucleus where it binds to DNA and provokes Caspase-independent chromatin condensation [Ref.1]. Proteolysis of the membrane tether in mature AIF can be mediated by local or cytoplasmic proteases entering the intermembrane space upon permeabilization of the outer membrane. Mitochondrial membrane permeabilization leads to the release of other IMS (Intermembrane Space) proteins, such as CYCS (Cytochrome-C), SMAC (Second Mitochondria-Derived Activator of Caspase12)/DIABLO and ENDOG that take part in the apoptotic process. BCL2L1 (Bcl2 Related Protein Long[..]
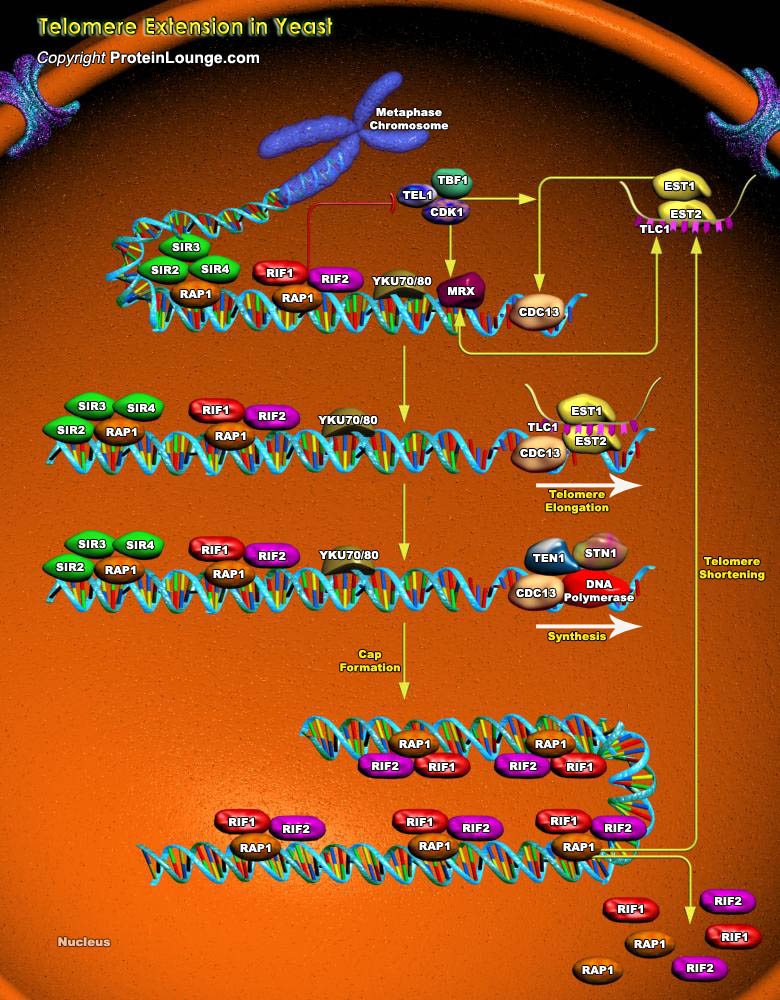
Telomeres are specialized nucleoprotein structures found at the ends of linear eukaryotic chromosomes. Telomeres confer stability to chromosomes by preventing nucleolytic degradation and recombination. They also function in chromosomal localization, nuclear architecture, and repression of nearby genes. The telomeric DNA of most organisms consists of simple tandem repeats that are rich in dG and dT residues on the 3' end-containing strand. This strand is synthesized by a ribonucleoprotein complex called telomerase, an enzyme that is critical to the maintenance of telomere length and function (Ref.1).The telomeres of the budding yeast Saccharomyces cerevisiae contain ~300 base pairs of double-stranded (TG1-3/C1-3A) sequences. Telomerase recruitment is mediated by a[..]
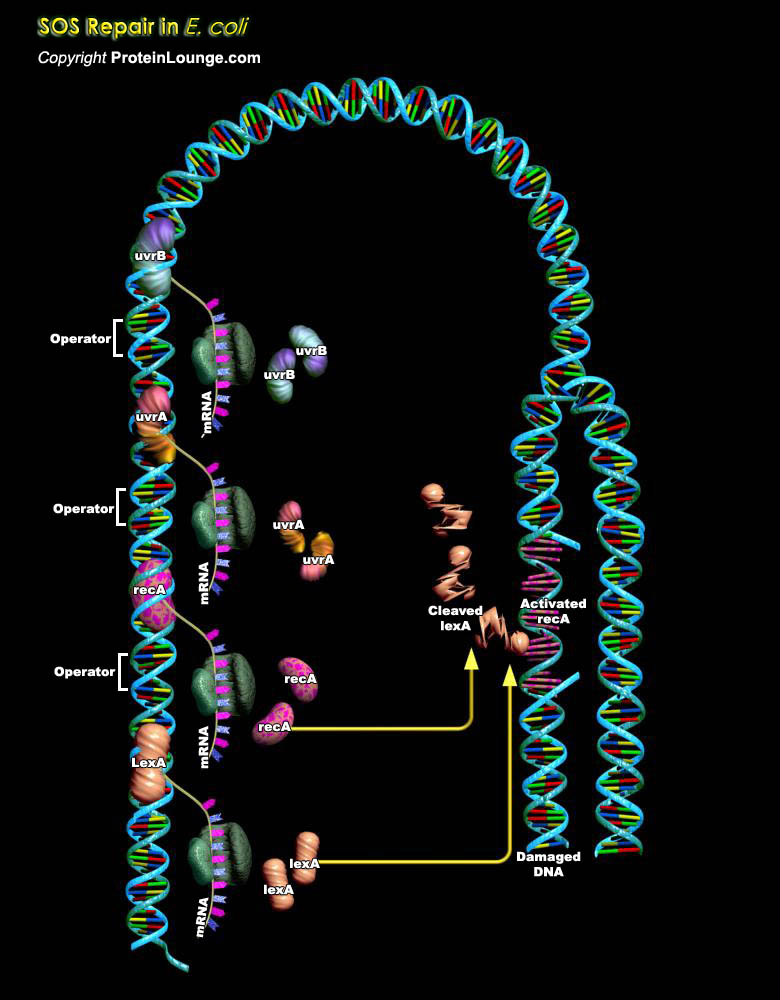
All organisms are exposed to a multitude of DNA damaging agents ranging from UV (Ultraviolet) light to fungal metabolites, like Aflatoxin B1. Furthermore, DNA damaging agents, such as ROS (Reactive Oxygen Species), can be produced by cells themselves as metabolic byproducts and intermediates. Together, these agents pose a constant threat to an organism's genome. As a result, organisms have evolved a number of vitally important mechanisms to repair DNA damage in a high fidelity manner. They have also evolved systems (cell cycle checkpoints) that delay the resumption of the cell cycle after DNA damage to allow more time for these accurate processes to occur. If a cell cannot repair DNA damage accurately, a mutagenic event may occur (Ref.1). Most bacteria,[..]
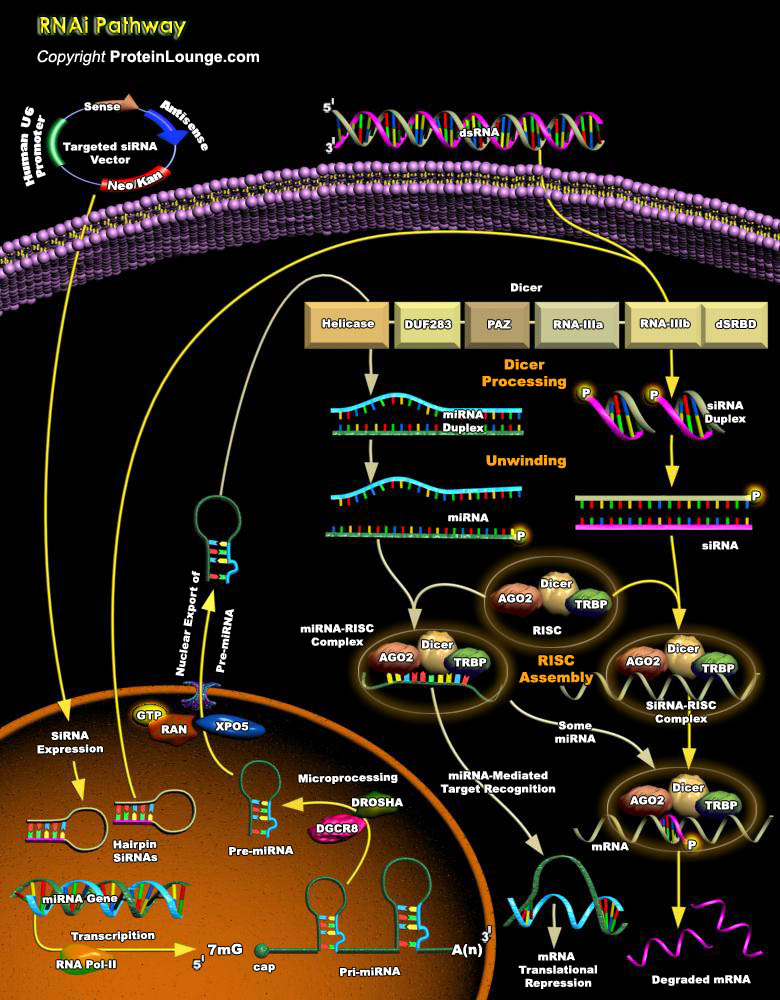
RNAi (RNA interference) is broadly defined as a gene silencing pathway that is triggered by dsRNA (double-stranded RNA). The dsRNA trigger can be supplied exogenously, as an experimental tool, or can derive from the genome. RNAi involves a number of steps from biogenesis of the trigger RNA, processing of dsRNA to small RNAs and formation of an effector complex containing the small RNA which goes on to guide mRNA cleavage, translational repression and/or chromatin modifications in a sequence-specific manner. Two types of small RNA molecules – miRNA (microRNA) and siRNA (small interfering RNA) – are central to RNA interference (Ref. 1, 2 and 9). siRNAs and miRNAs are 20–25-nt RNAs involved in silencing homologous genes or their transcripts. miRNAs[..]
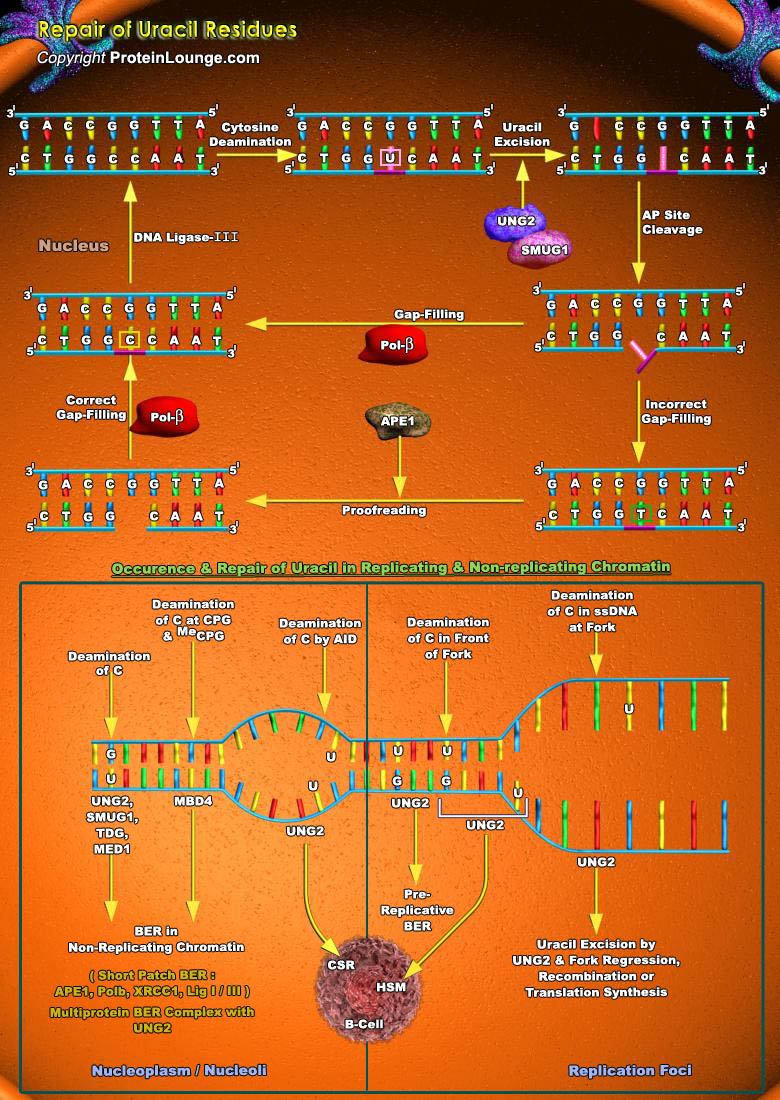
Hydrolytic deamination of cytosine to uracil is a major source of genome instability in human cells. This mutagenic process is mostly enhanced in single stranded DNA . Single stranded DNA can be found in transcribed genes, near forks, in mitochondrial DNA as well as in breathing DNA. It is believed to be much higher in replicating cells as compared to non-replicating cells. The cytosine deamination to uracil needs to be repaired soon to restore genomic stability. Otherwise uracil residues may give rise to C→T transitions, which are the most common spontaneous mutations occurring in living organisms. Uracil in DNA arises by spontaneous deamination of cytosine to generate pro-mutagenic U:G mispairs. Uracil in DNA is also produced by the incorporation of dUMP[..]
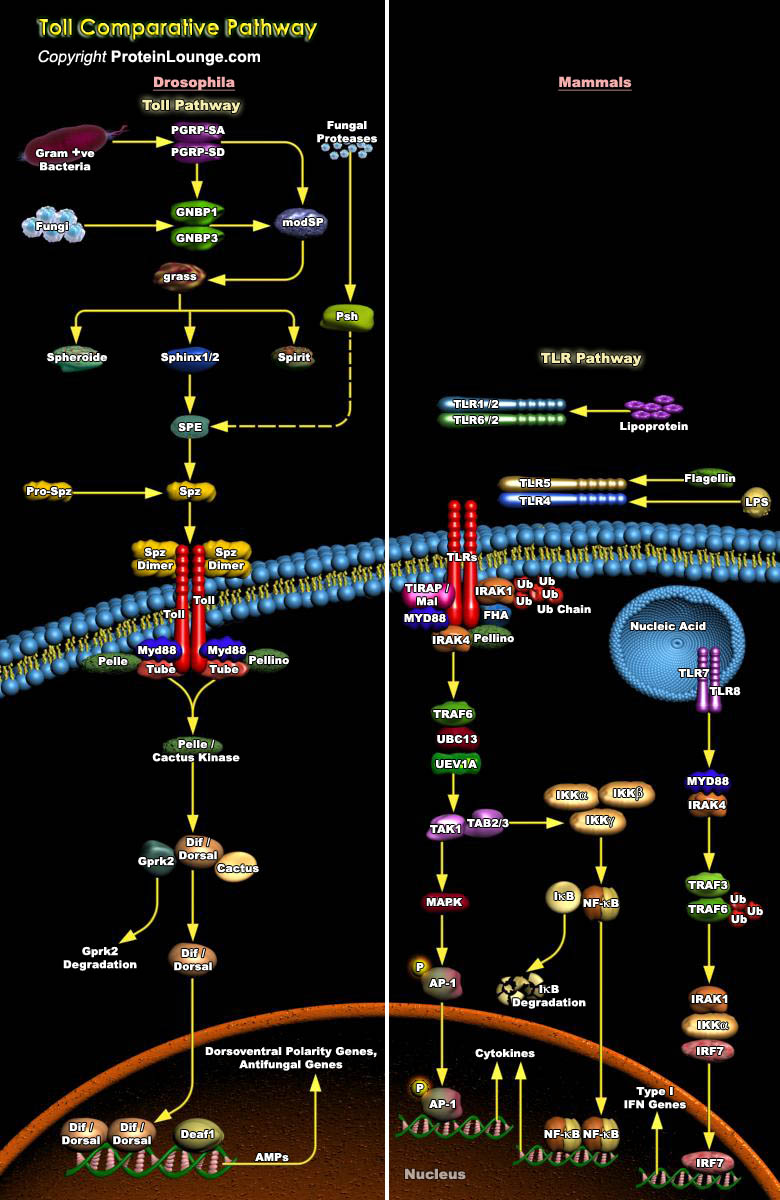
Innate immune system plays an important role in immune responses against pathogen invasion in vertebrates and invertebrates. In mammalian systems, it provides the first line of defense against multiple pathogens, whereas in insects, the entire immune system is innate. The Toll pathway plays important functions in innate immunity against infectious pathogens in vertebrates and invertebrates. Activation of the Toll pathway is different between vertebrates and invertebrates. In vertebrates like Homo sapiens, Toll pathway is directly activated by Toll-like receptors (TLRs) binding to various pathogen-associated molecular patterns (PAMPs) from different pathogens, whereas in invertebrates like Drosophila, Toll receptors bind to the cytokine-like molecule Spätzle but not to[..]
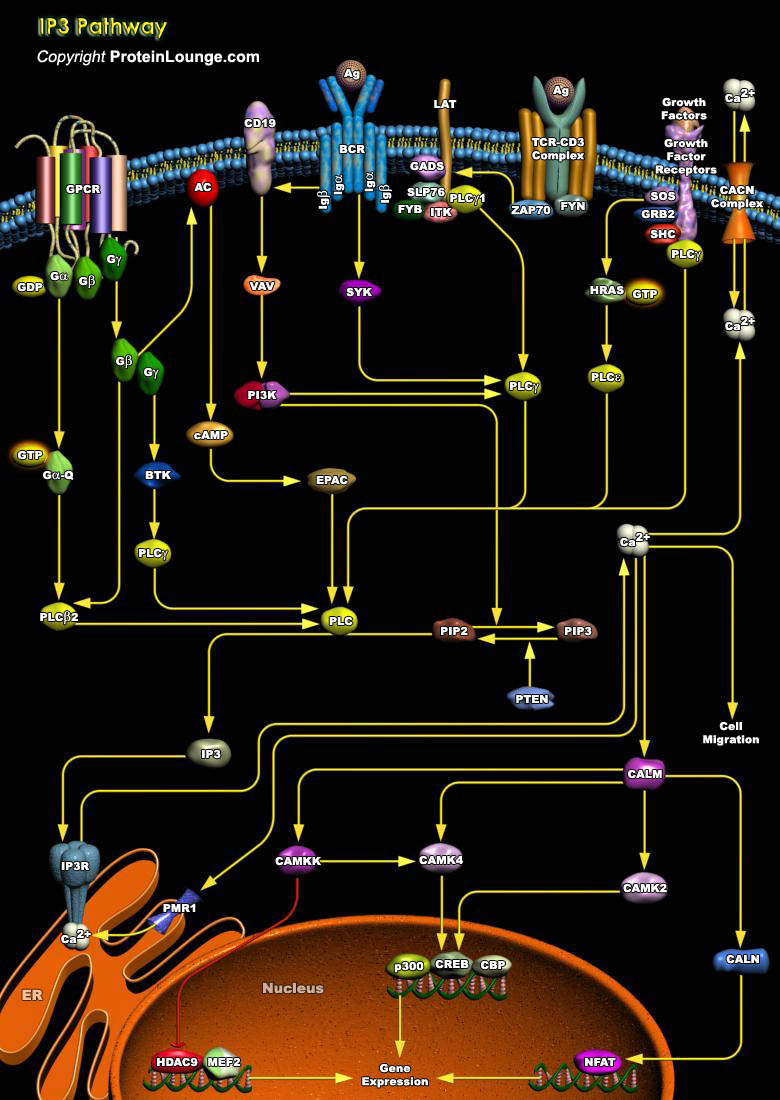
IP3 (Inositol 1,4,5-triphosphate), also known as a second messenger, is a molecule that functions to transfer a chemical signal received by the cell, such as from a hormone, neurotransmitters, growth factors and hypertrophic stimuli such as AngII (Angiotensin-II), Beta-adrenergic receptor agonists, and ET1 (Endothelin-1) to various signaling networks within the cell. IP3 is known to play a crucial role in initiating and propagating these messages; however, the precise mechanism of how IP3 relates to the next element in its signaling pathway, the calcium wave, remains highly controversial. The receptors for IP3, IP3R (IP3 Receptor) constitute a family of Ca2+ channels responsible for the mobilization of intracellular Ca2+ stores. Three different receptor types have been[..]
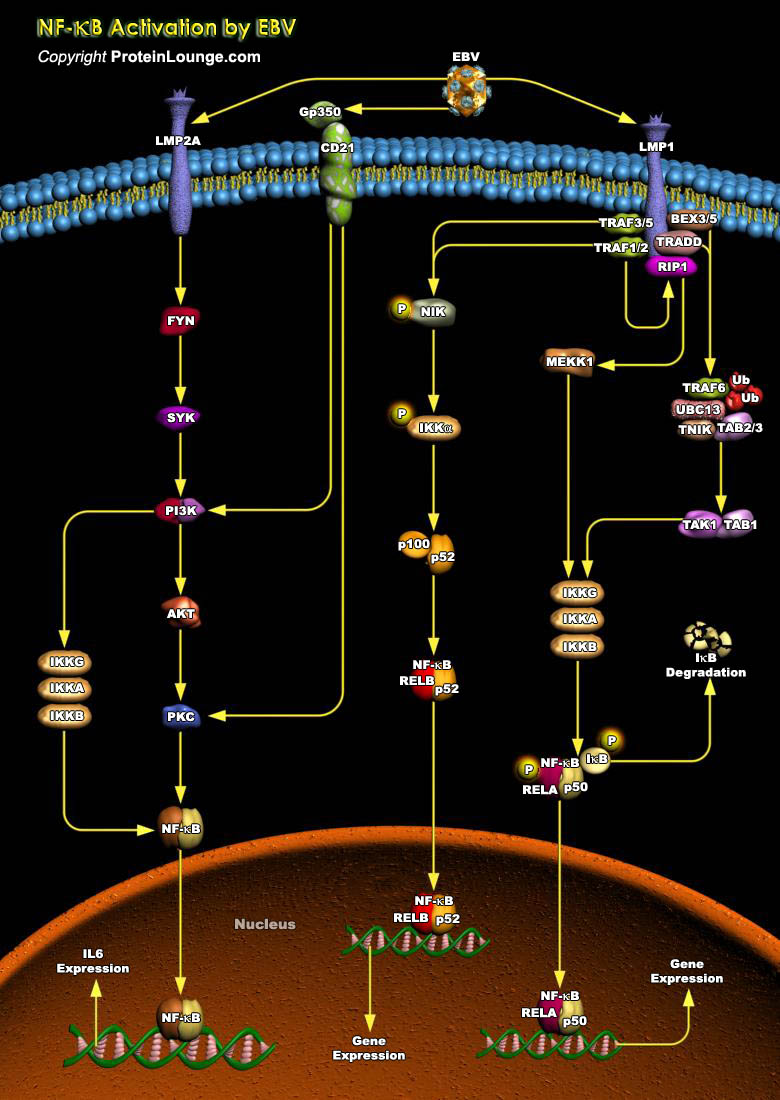
Epstein-Barr virus (EBV) is a gamma-herpesvirus that infects more than 90% of people worldwide, is the etiologic agent of infectious mononucleosis, and is associated with multiple human malignancies, primarily of lymphoid and epithelial cell origin, including Burkitt’s lymphoma, post-transplant lymphoma, AIDS-associated lymphomas, Hodgkin’s disease, T-Cell lymphoma, NPC (Nasopharyngeal Carcinoma), parotid gland carcinoma, and gastric carcinoma. EBV-encoded RNAs (EBERs) are the most abundant viral transcripts in latently EBV-infected cells (Ref.1). The canonical NF-kappaB pathway plays a pivotal role in regulating a variety of essential processes, such as immunity, cell survival, and proliferation. A large number of both positive and negative regulators of NF-kappaB[..]
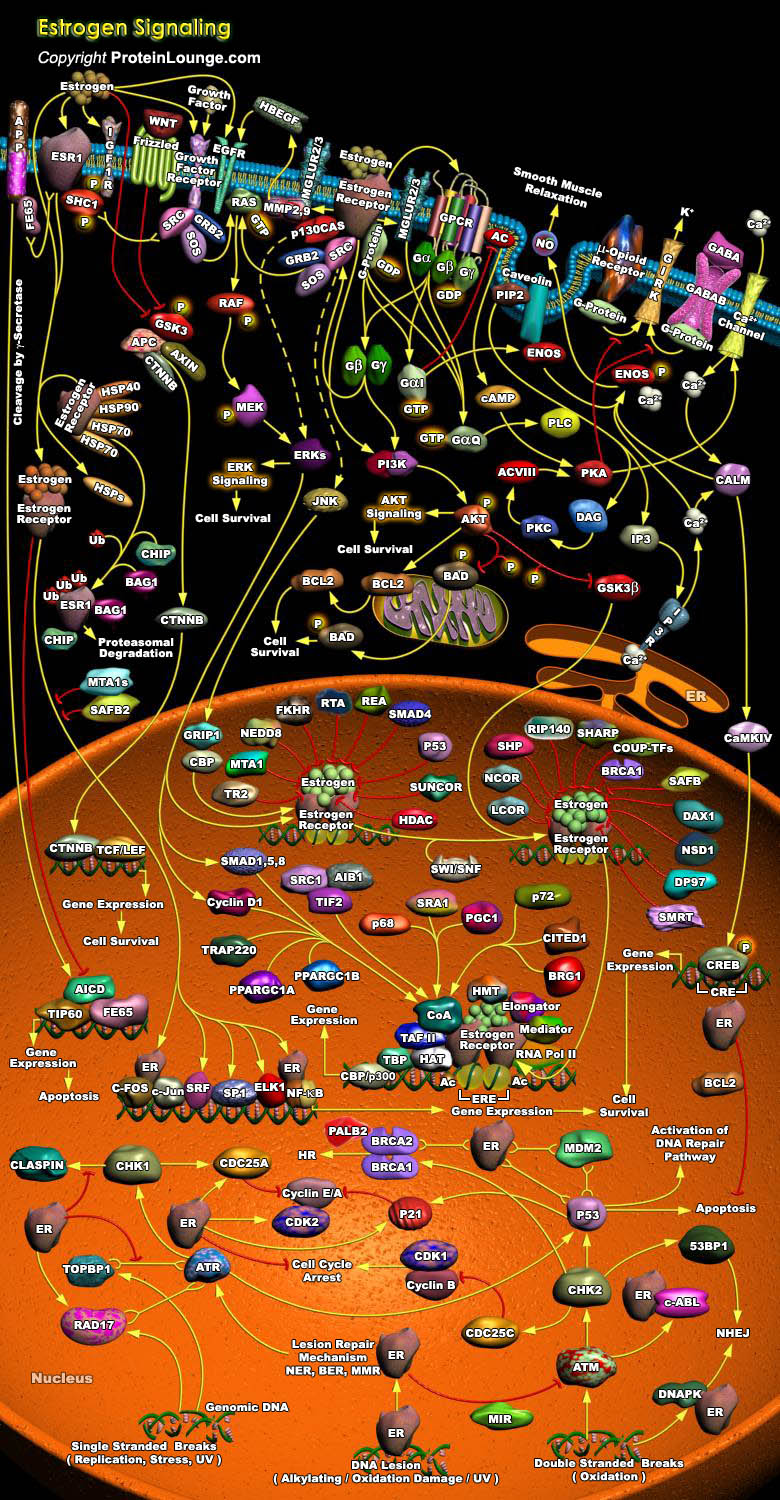
Estrogens play important roles in growth, development, reproduction, and maintenance of a diverse range of mammalian tissues. The physiological effects of estrogens are mediated by the intracellular ERs (Estrogen Receptors), which regulate transcription of target genes through binding to specific DNA target sequences. The ERs orchestrate both transcriptional and non-genomic functions in response to estrogens, xenoestrogens and signals emanating from growth factor signalling pathways. The pleiotropic and tissue-specific effects of estrogens are mediated by the differential expression of two distinct ER subtypes: ER-Alpha and ER-Beta, and their coregulators (Ref.1). The activities of a plethora of ER-interacting proteins converge to confer distinct functionalities on[..]
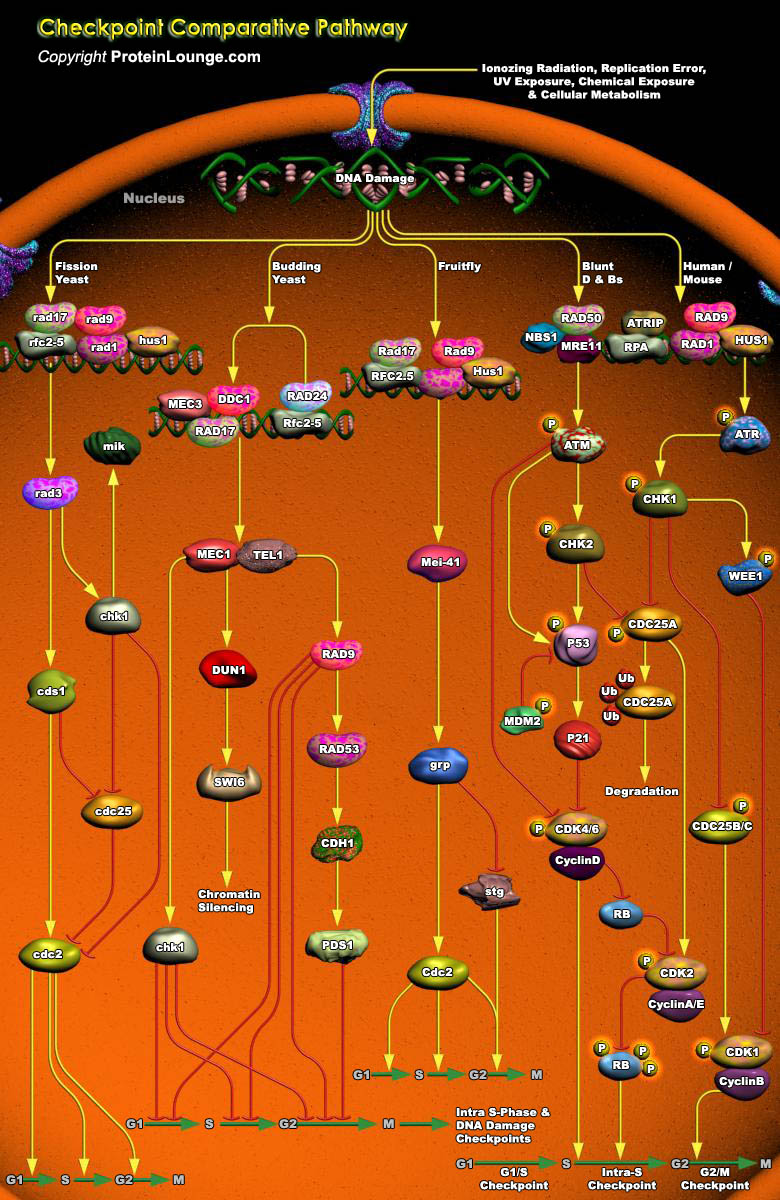
Cell cycle checkpoints are surveillance mechanisms that monitor the order, integrity, and fidelity of the major events of the cell cycle. These include growth to the appropriate cell size, the replication and integrity of the chromosomes, and their accurate segregation at mitosis. Many of these mechanisms are ancient in origin and highly conserved, and hence have been heavily informed by studies in simple organisms such as the yeasts. Others have evolved in higher organisms, and control alternative cell fates with significant impact on tumor suppression.Recent work on the mechanisms of DNA damage and replication cell cycle checkpoints has revealed great similarity between the checkpoint pathways of organisms as diverse as yeasts, flies, humans and mouse. However, there[..]
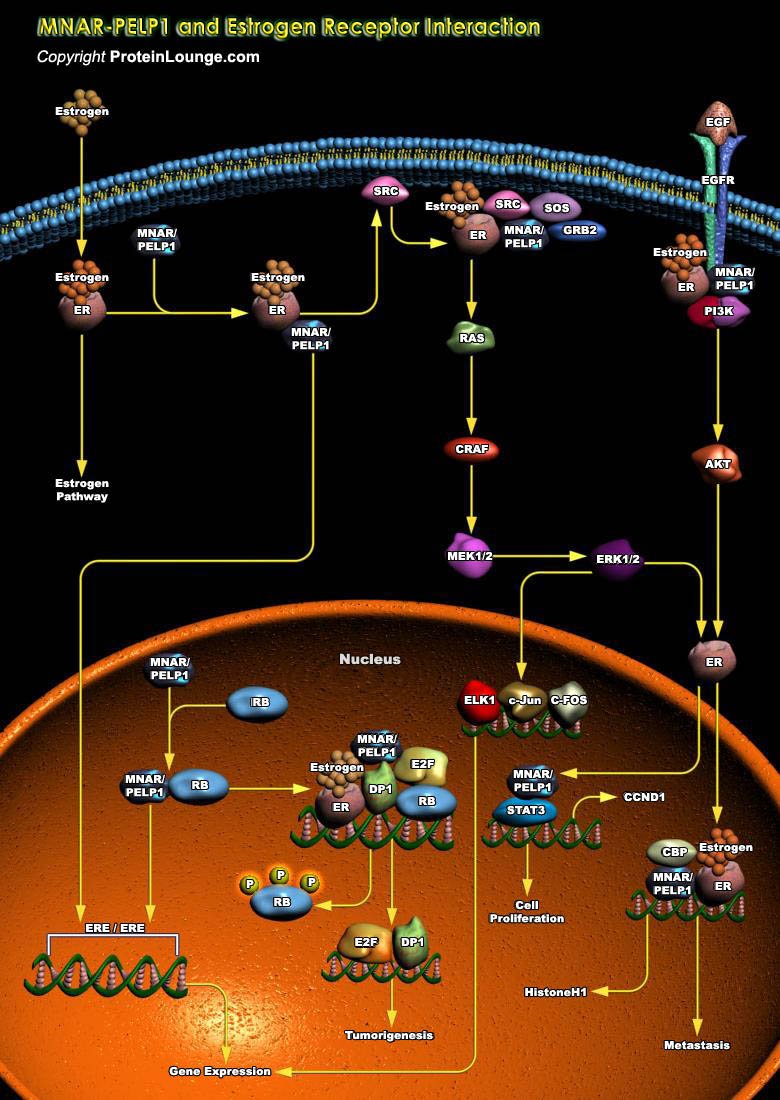
Estrogen contributes to the development of three of the top five cancers of women--those of the breast, uterus, and ovaries--which together account for an estimated 240,000 new cancer cases a year in the United States alone (Ref.1). The ER (Estrogen Receptor) signaling pathway is implicated in the progression of cancer tumorigenesis and the activity of the hormone receptor is regulated by ligands (Estrogens), and coactivators or corepressors (Ref.2). MNAR (Modulator of Nongenomic Activity Of Estrogen Receptor)/PELP1 (Proline Glutamic Acid-Rich Nuclear Protein)/p160, a recently identified novel coactivator of ERs, has been shown to be amplified and overexpressed in breast cancer cell lines, whose expression in some cases, may be as high as, 3-5 times higher in breast[..]
.jpg)
NF-KappaB (Nuclear factor-KappaB)/Rel proteins are dimeric, sequence-specific transcription factors involved in the activation of an exceptionally large number of genes in response to inflammation, viral and bacterial infections, and other stressful situations requiring rapid reprogramming of gene expression. NF-kappB pathway is crucial for the regulation of immune responses, inflammation, and stress responses. Rel/NF-KappaB transcription factors include a collection of proteins, conserved from the fruit fly Drosophila melanogaster to humans. The fruit fly Drosophila melanogaster has proved to be a valuable model system to study these evolutionarily conserved NF-KappaB mediated immune responses (Ref.1 and 2).NF-KappB pathways play a major role in Drosophila host[..]

