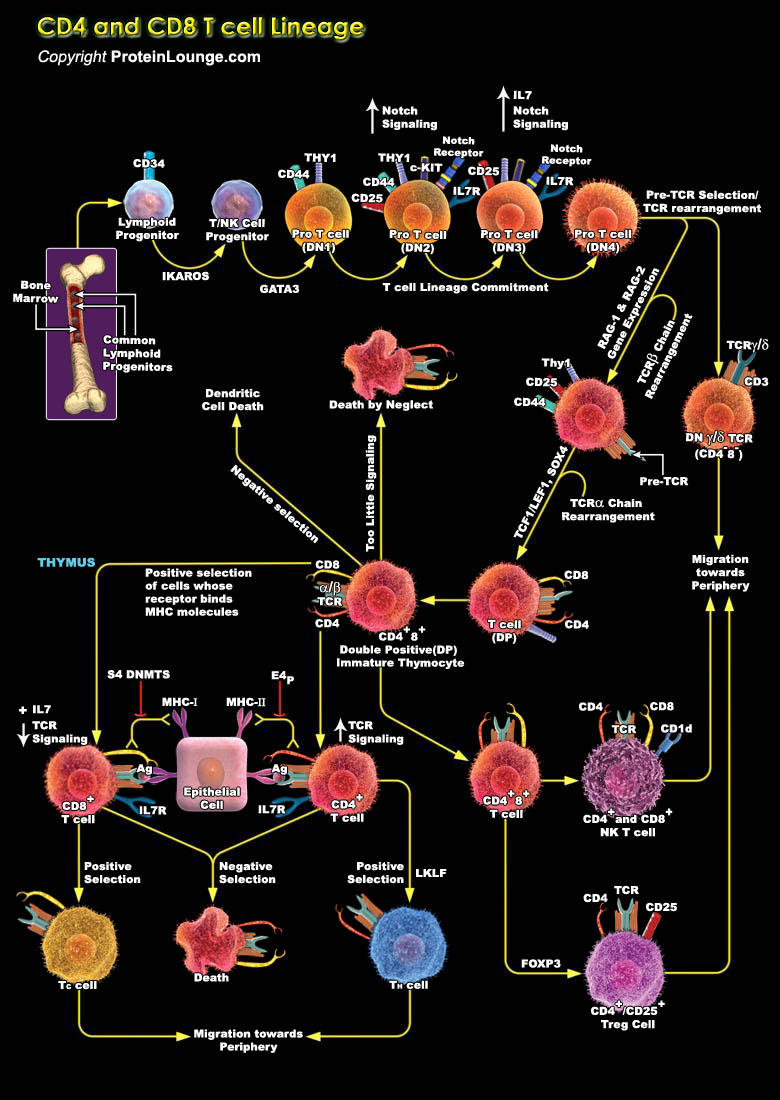
Cell-fate decisions are controlled typically by conserved receptors that interact with co-evolved ligands. Therefore, the lineage-specific differentiation of immature CD4+CD8+ T cells into CD4+ or CD8+ mature T cells is unusual in that it is regulated by clonally expressed, somatically generated T-cell receptors (TCRs) of unpredictable fine specificity. Each mature T cell generally retains expression of the co-receptor molecule (CD4 or CD8) that has an MHC-binding property that matches that of its TCR. Two models were proposed initially to explain this remarkable outcome--'instruction' of lineage choice by initial signalling events or 'selection' after a stochastic fate decision that limits further development to cells with coordinated TCR and co-receptor[..]
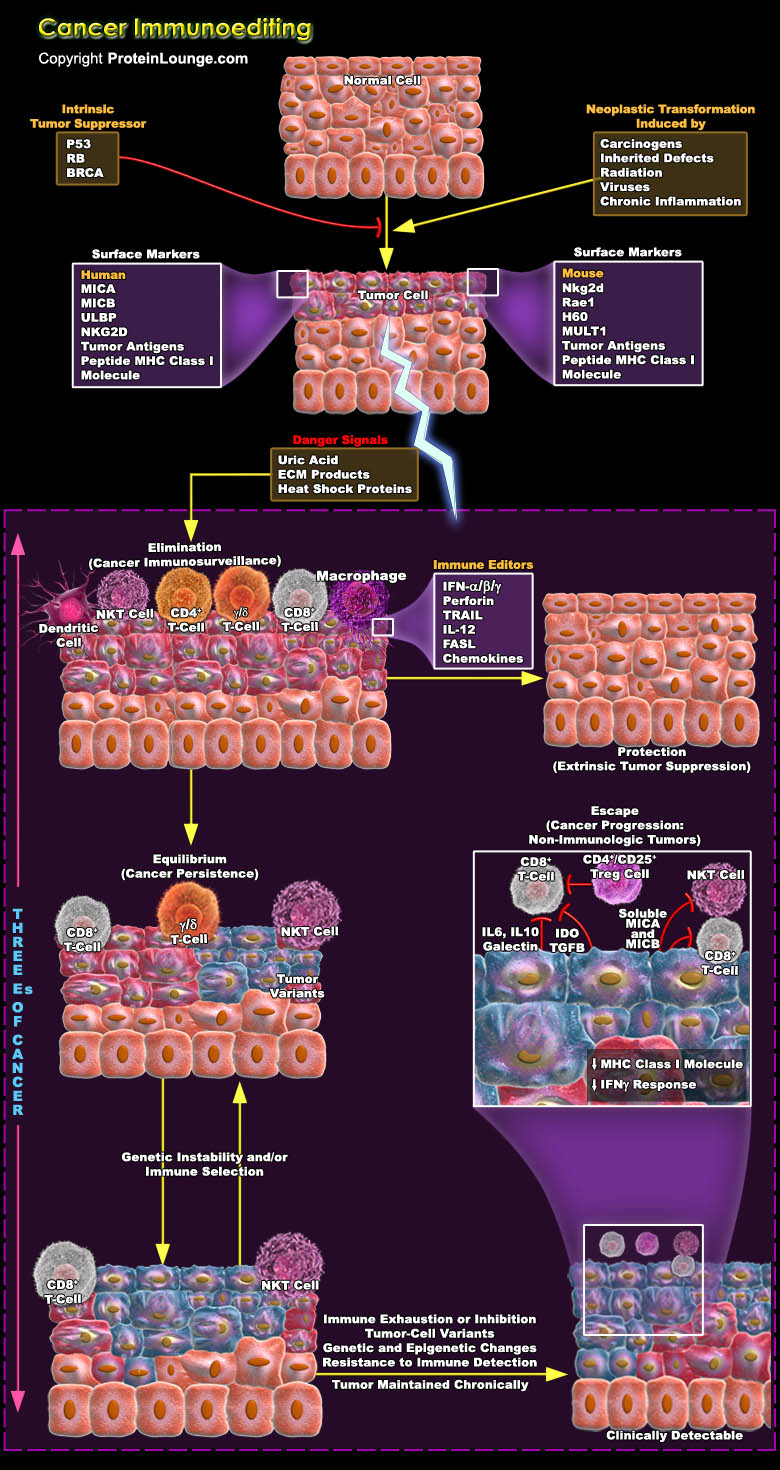
As research into tumour immunology continues at an incredible pace, a considerable amount of work is aimed at exploring the mechanisms that underlie the immunological recognition and elimination of cancer and the downstream consequences of these processes. The capacity of the immune system for recognition is not limited solely to the classic models of self versus pathogen or self versus non-self but encompasses the more-subtle differences that exist between self and transformed self. This conclusion provides the argument for reconsidering the largely discarded hypothesis of cancer immunosurveillance. Immune system attempts to constrain tumour growth, but sometimes tumour cells might escape or attenuate this immune pressure, similar to the way in which these cells[..]
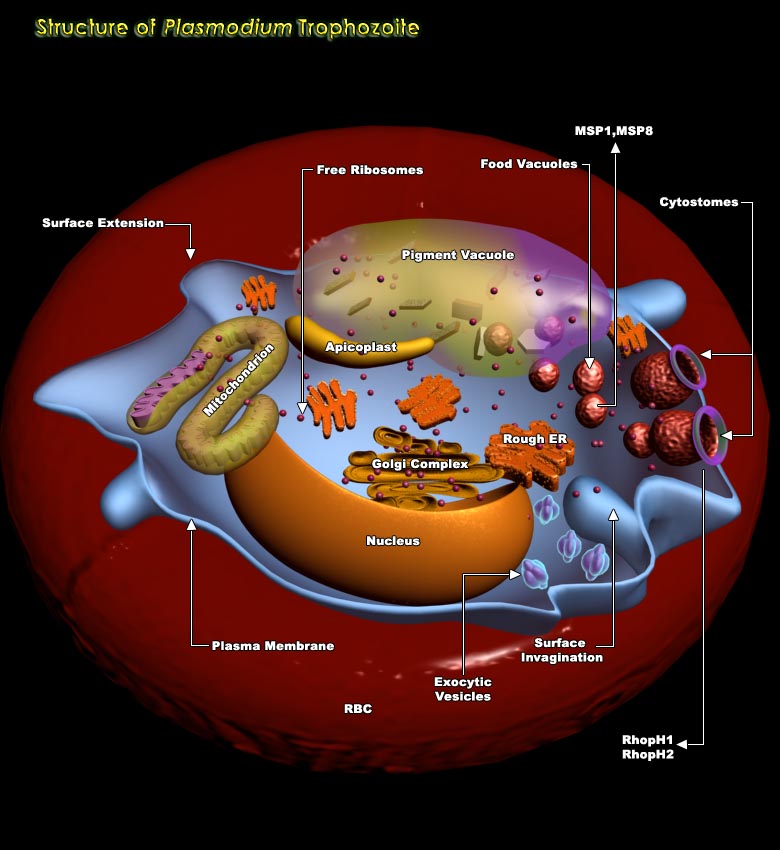
In the life cycle of Plasmodium spp, Erythrocytic stage is very important involving four stages viz. Merozoite stage, ring stage, Trophozoite stage and Schizont stage. On being released from the hepatocytes, the Merozoites enter the bloodstream prior to infecting red blood cells. They use the Apicomplexan invasion organelles to recognize and enter the host erythrocyte. The parasite first binds to the erythrocyte in a random orientation. It then reorients such that the apical complex is in proximity to the erythrocyte membrane. A tight junction is formed between the parasite and erythrocyte. As it enters the red blood cell, the parasite forms a parasitophorous vesicle, to allow for its development inside the erythrocyte. After invading the erythrocyte, the parasite[..]
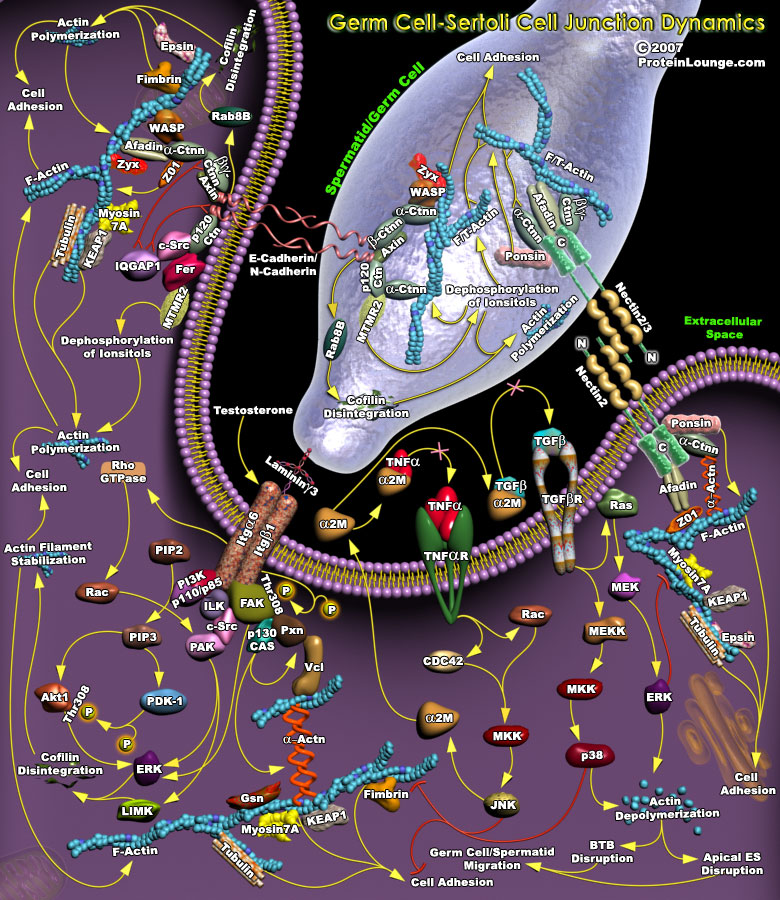
Sertoli-Germ (Spermatid)-cell interactions affect spermatogenesis at the molecular, cellular and biochemical levels. Germ cell movement within the epithelium is vital because germ cells, if induced to release into the tubule lumen prematurely, will be unable to fertilize the ovum. On the other hand, if germ cells are forced to remain attached to the seminiferous epithelium for a period of time longer than necessary to complete their development, they will degenerate and eventually be phagocytosed by Sertoli cells and for this, proper regulation of germ cell migration in the seminiferous epithelium and at blood-testes barrier (abbreviated as BTB) is essential (Ref.1). Spermatid/germ cell and sertoli cell interactions in the testis occur through specialized junctions[..]
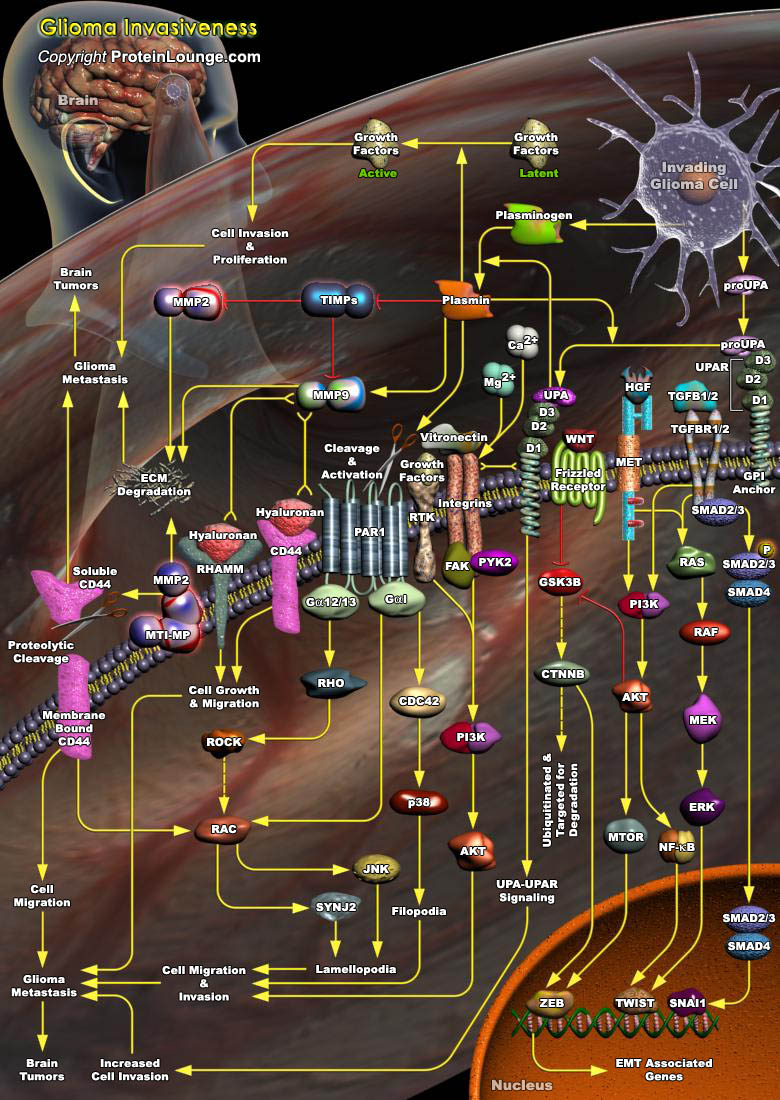
Gliomas are the most common intracranial malignant tumors in humans, and high-grade Gliomas in particular pose a unique challenge due to their propensity for proliferation and tissue invasion. The invasion of neoplastic cells into healthy brain tissue is a pathologic hallmark of Gliomas and contributes to the failure of current therapeutic modalities (surgery, radiation and chemotherapy). Transformed glial cells share the common attributes of the invasion process, including cell adhesion to ECM (Extracellular Matrix) components, cell locomotion, and the ability to remodel extracellular space. However, Glioma cells have the ability to invade as single cells through the unique environment of the normal CNS (Central Nervous System). The brain parenchyma has a unique[..]
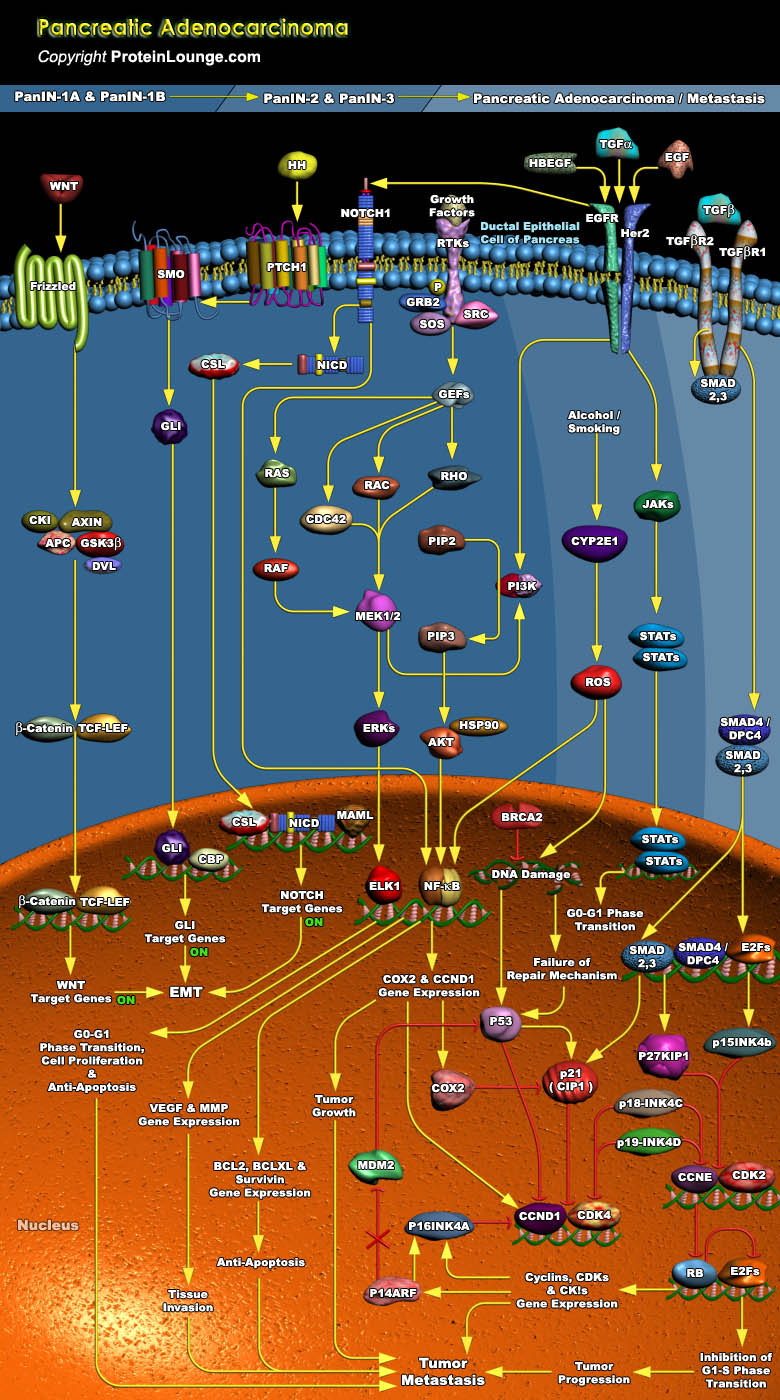
Pancreatic carcinoma is one of the most enigmatic and aggressive malignant diseases. Neoplasms of the pancreas encompass a wide spectrum of benign and malignant tumors. Pancreatic adenocarcinoma, the malignant neoplasm of the exocrine duct cells, accounts for more than ninety percent of all pancreatic tumors (Ref.1). Pancreatic ductal adenocarcinoma evolves from a progressive cascade of cellular, morphological and architectural changes from normal ductal epithelium through pre-neoplastic lesions termed PanIN (Pancreatic Intraepithelial Neoplasia). These PanIN lesions are in turn associated with somatic alterations in canonical oncogenes and tumor suppressor genes. Pancreatic cancer like many other tumors over-expresses the RTKs (Receptor Tyrosine Kinases), EGFR[..]
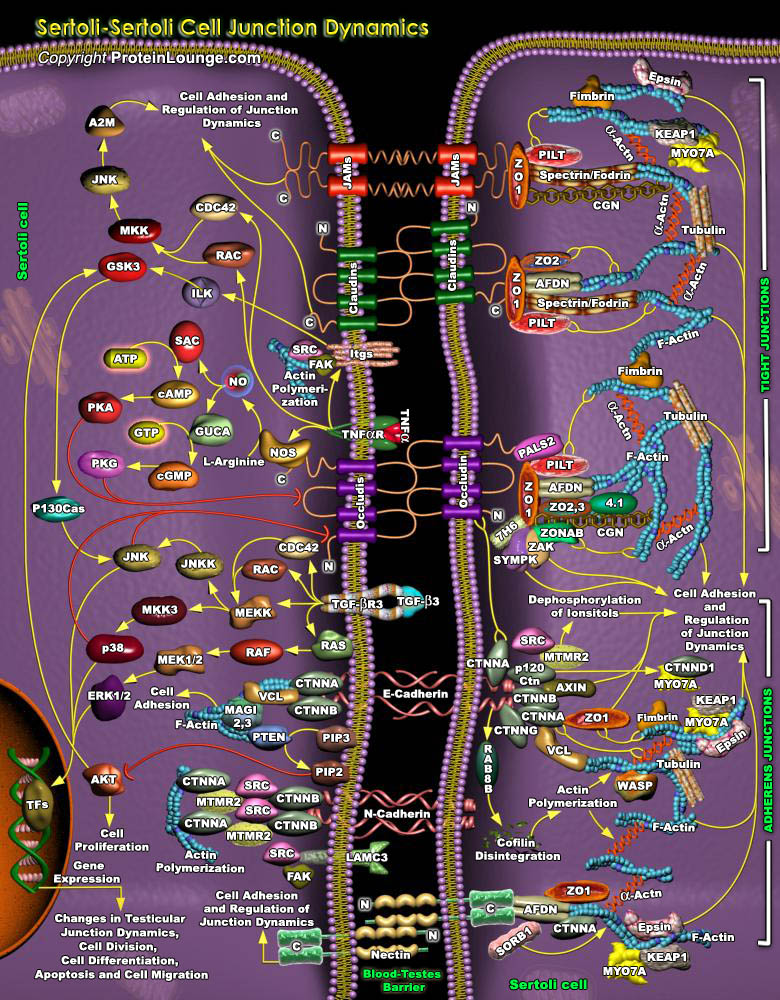
The Blood-Testes Barrier (abbreviated as BTB) acts as a physical barrier between the blood vessels and the seminiferous tubules of the testes. This barrier is formed by tight and adherens connections between the Sertoli cells, which are sustentacular cells (supporting cells) of the seminiferous tubules, and nourish the spermatogonia (Ref.1). In the testes, tight and adherens junctions are dynamically remodeled to allow the movement of post-meiotic germ cells across the seminiferous epithelium and the timely release of spermatids into the tubular lumen. Three main functions are ascribed to the blood-testes barrier; (i) creates a specialized environment; (ii) regulates the passage of molecules; and (iii) serves as an immunological barrier. When the blood-testes barrier[..]
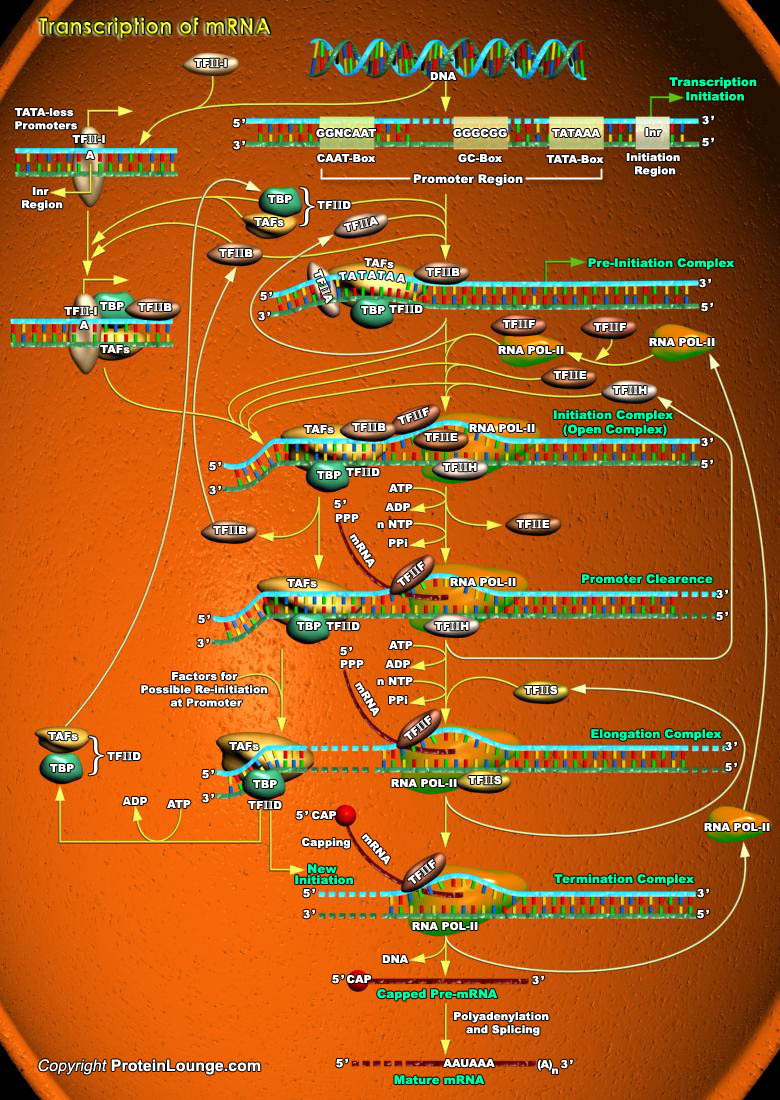
The process of DNA replication, gene transcription and protein translation combinedly known as central dogma of biology is mainly responsible for the expression and maintenance of every gene in an organism. Transcription is the process through which a DNA sequence is enzymatically copied by an RNA polymerase to produce a complementary RNA. Transcription can also be defined as a process that transcribes genetic information from DNA into RNA. In eukaryotes, it takes place in the nucleus, mitochondria and chloroplast. Transcription is performed by DNA-directed RNA Polymerases. Unlike DNA Polymerases, RNA Polymerases do not need a primer to start the reaction. While Bacteria contain only 1 RNA Polymerase, there are 3 different RNA polymerases in eukaryotic cells, which[..]
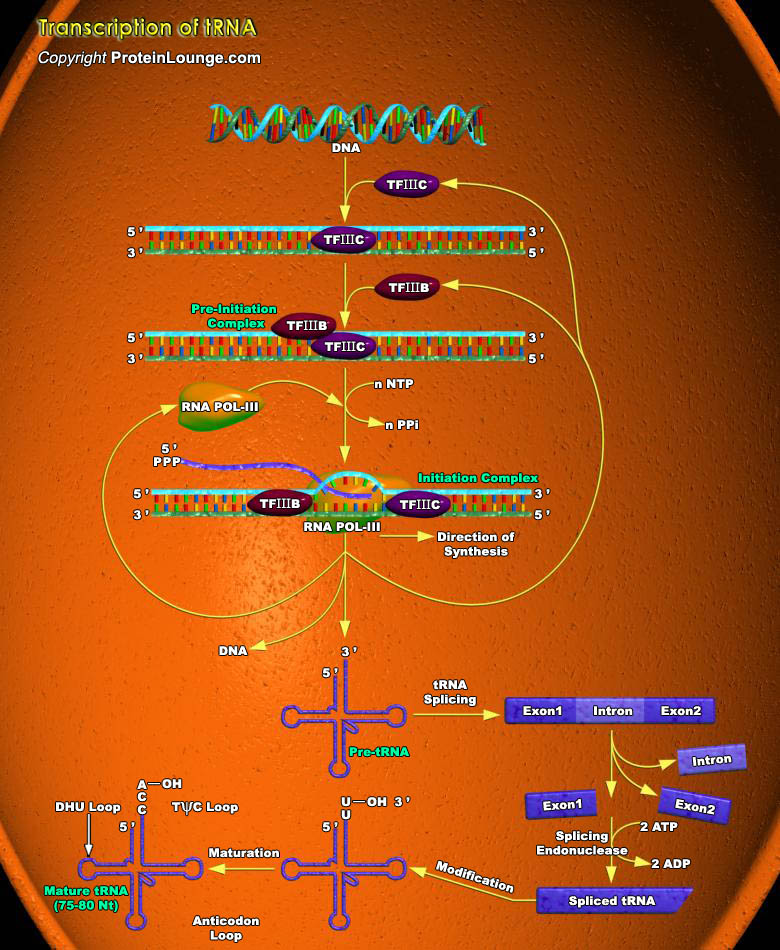
Transfer RNA (tRNA) is a small RNA chain (74-93 nucleotides) that converts information contained in mRNA into amino acid sequence. tRNA synthesis involves several steps, including initial transcript synthesis, tRNA precursors maturation and amino acid mediated charging of tRNA molecules. Maturation and charging of tRNA occurs both in cytoplasm as well as nucleus. In all organisms, tRNAs are transcribed in a pre-tRNA form that requires multiple processing steps before the mature tRNA is ready for use in translation (Ref.1).RNA polymerase III catalyses the synthesis of tRNAs in eukaryotic organisms. Many auxiliary factors work along with RNA POL III enzymes in regulating the transcription. They form a multi protein complex at tRNA genes that helps in polymerase[..]
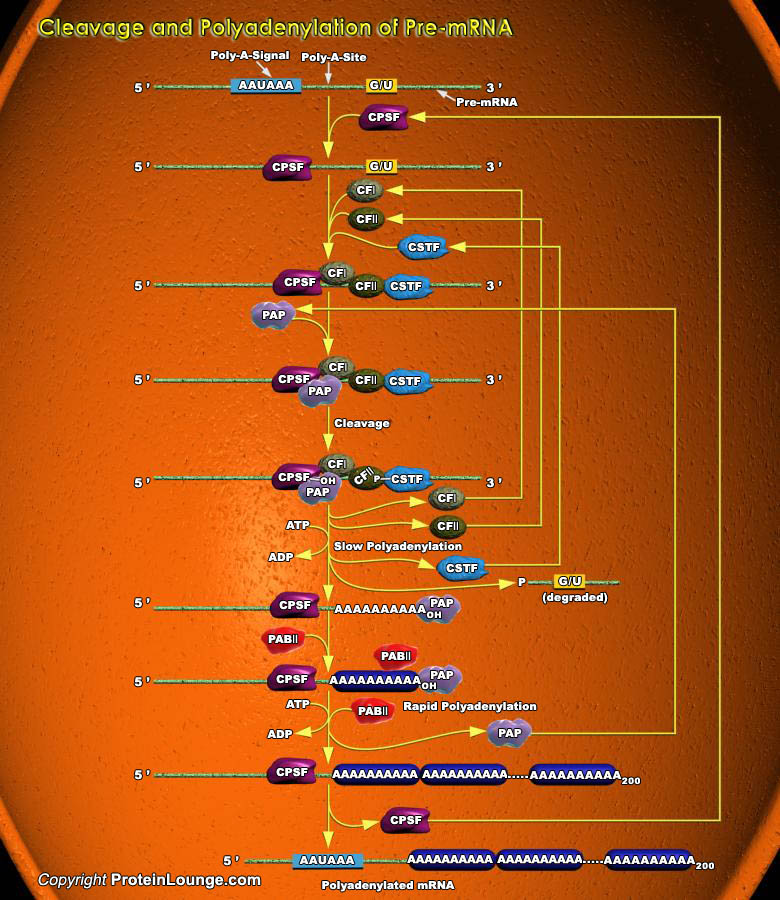
During the course of transcription, the initial RNA product synthesized by RNA POL-II (RNA Polymerase-II), called a Primary transcript undergoes several processing steps including Capping, Splicing and Polyadenylation, before a functional mRNA (messenger RNA) is produced. RNA Polymerase initiates transcription at the first nucleotide of the first exon of a gene. Shortly after transcription begins, the 5' end of the nascent RNA is capped with 7-Methyl-Guanylate.Transcription by RNA polymerase-II terminates at any one of the multiple sites approx. 0.5-2kb downstream from the 3' end of the last exon in the transcript. The 3' end of a functional mRNA then is generated by endonucleolytic cleavage at a specific sequence, the Poly-A site, located at the 3' end[..]
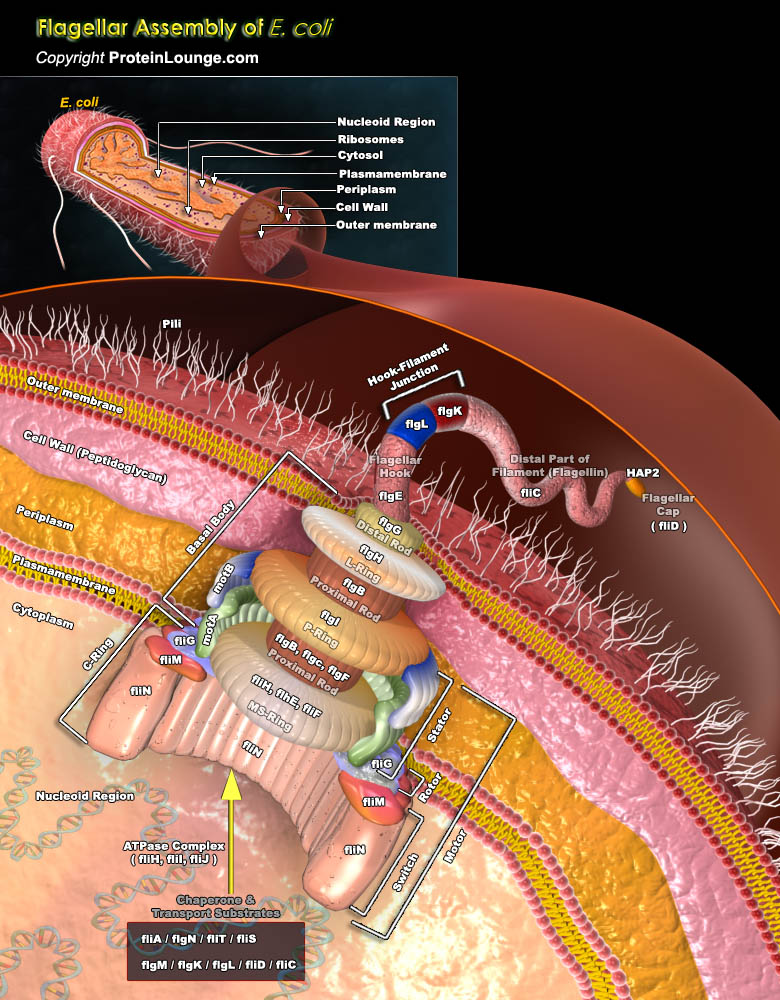
Flagella are hair like structures that help in locomotion of a cell or micro-organism. In gram negative bacteria like Escherichia coli (E.coli), flagella are an important system of protein transport besides being involved in the movement. Flagella can be present all over the surface, at one end or at both ends of the bacterium. Normally a flagellum consists of a long filament as a molecular screw, a hook that functions as a universal joint and a basal body which is a rotary motor. More than 60 genes are believed to be involved in flagellar formation and function (Ref.1).The rotary motor in flagella consist of several domains that include MS ring , the C ring, the switch complex, the rod, the export apparatus, L and P rings on the rod and the stator which is[..]
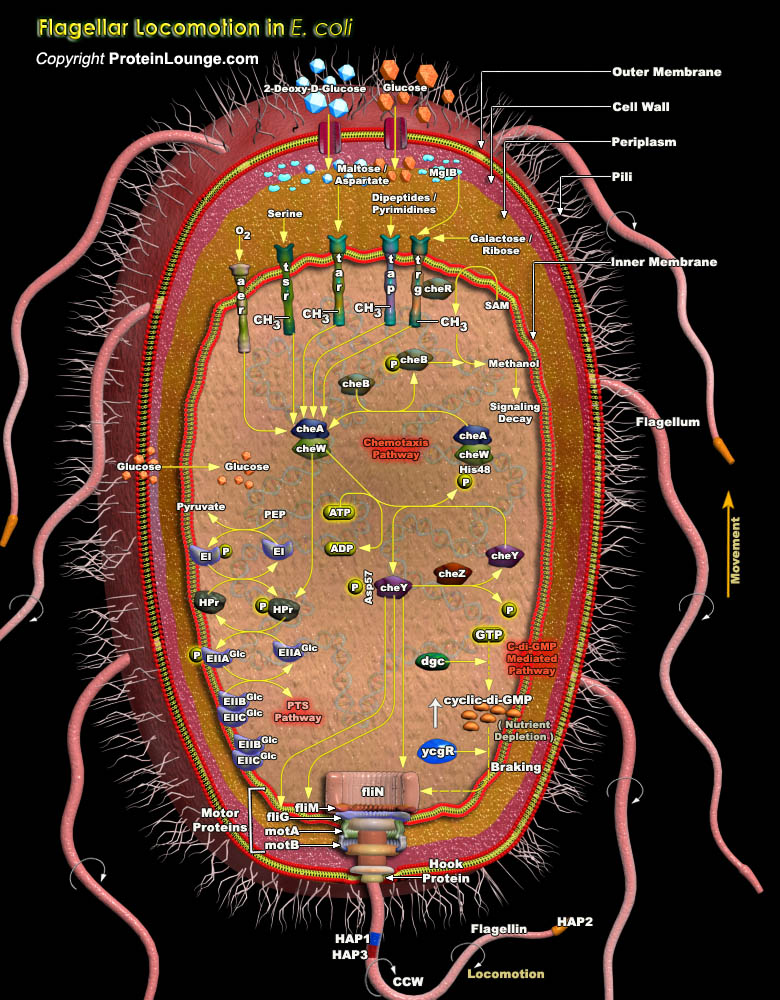
In an ever-changing environment, it is essential that organisms are able to sense these changes and to respond appropriately. Possible responses include alterations in gene expression and/or active movement towards or away from an environment. Most sensory pathways in eukaryotic organisms rely on serine, threonine or tyrosine protein kinases, whereas the most common sensory pathways in prokaryotes use a HAP (Histidine-Aspartate Phosphorelay) system. HAP systems have at least two components-a dimeric HPK (Histidine Protein Kinase) and a RR (Response Regulator). HAP systems are also found in many lower eukaryotes. Bacteria can sense a vast range of environmental signals, from the concentrations of nutrients and toxins to oxygen levels, pH, osmolarity and the intensity[..]

