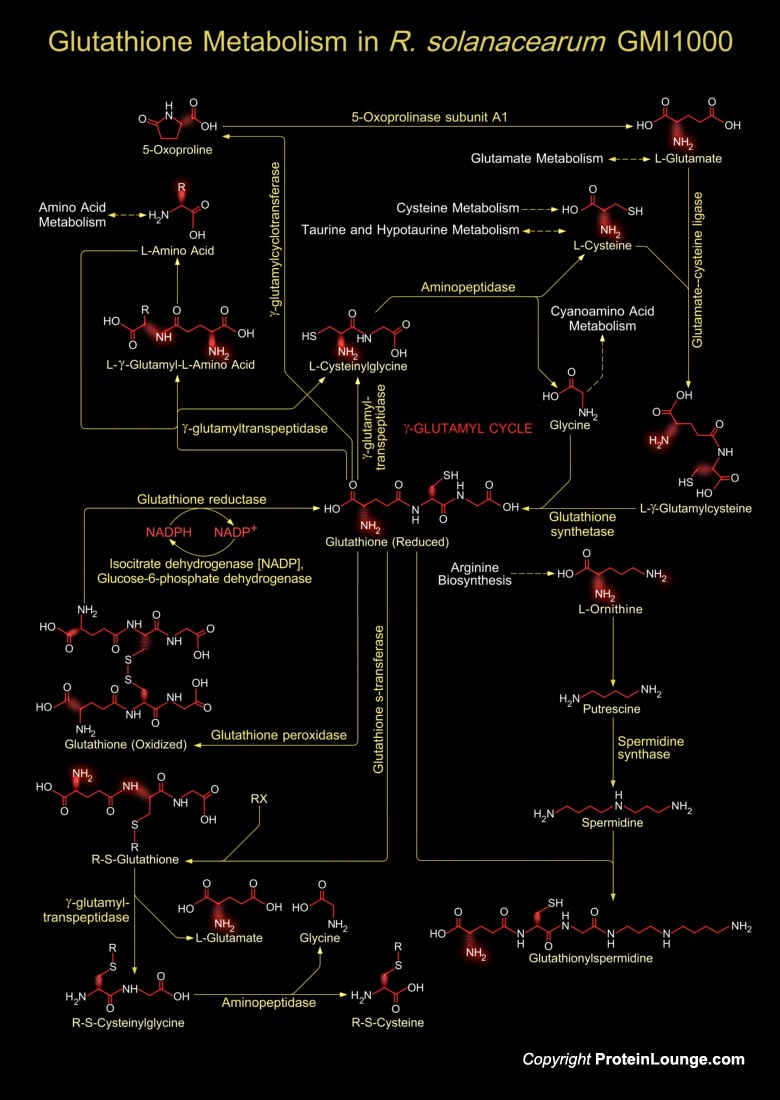
Ralstonia solanacearum is a Gram negative β-proteobacteria. It is an aerobic non-spore-forming, Gram-negative, plant pathogen. R. solanacearum is soil-borne and motile with a polar flagellar tuft. It colonises the xylem, causing bacterial wilt in a very wide range of potential host plants (Ref.1). Although R. solanacearum is considered a plant pathogen, it mainly behaves as a soil bacterium of saprophytic life with an extremely versatile lifestyle, which allows the bacteria to survive in the soil for long periods in the absence of its host plant his phytopathogen invades the vascular tissue in a systemic way and has a wide range of hosts with arge geographic distribution and diverse pathogenic behavior. R. solanacearum can live for long periods in[..]
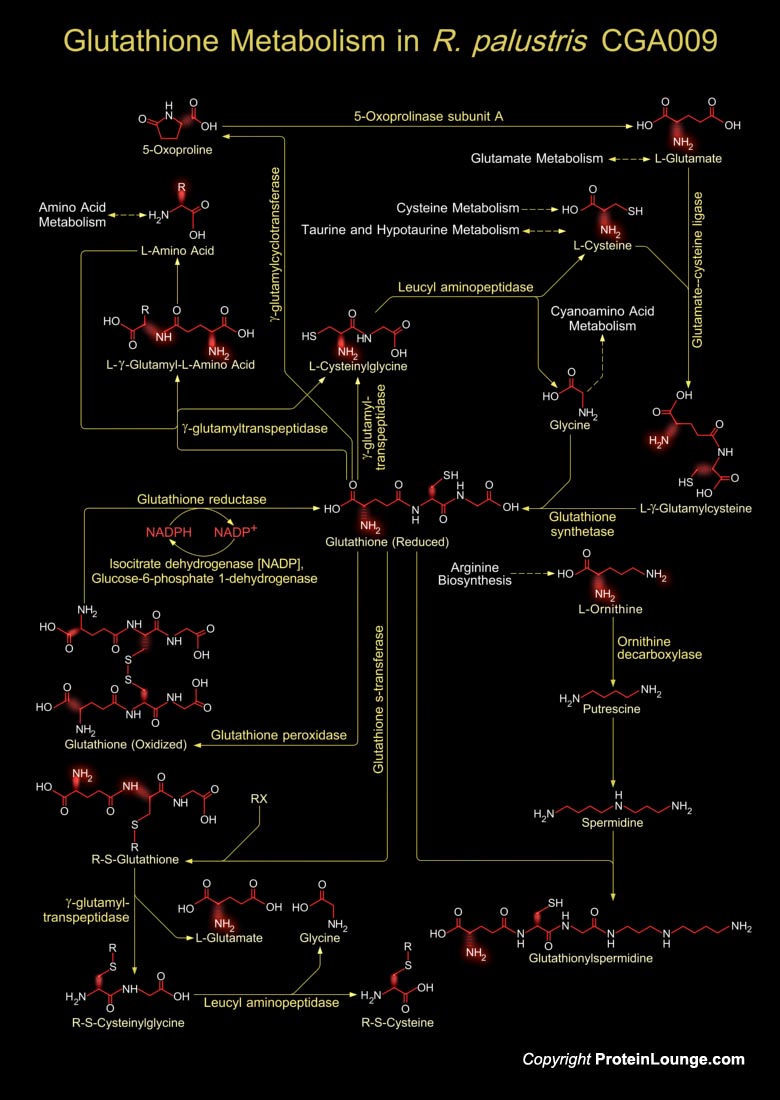
Rhodopseudomonas palustris is a rod-shaped, Gram-negative purple nonsulfur bacterium. It is an alphaproteobacterium that serves as a model organism for studies of photophosphorylation, regulation of nitrogen fixation, production of hydrogen as a biofuel, and anaerobic degradation of aromatic compounds. This bacterium is able to transition between anaerobic photoautotrophic growth, anaerobic photoheterotrophic growth, and aerobic heterotrophic growth. It is found in terrestrial soil and water environments and grows phototrophically under anaerobic conditions. Under such conditions, it uses light as a source of energy and either organic compounds or carbon dioxide as carbon sources. R. palustris also grows aerobically as a heterotroph, and some strains grow anaerobically[..]
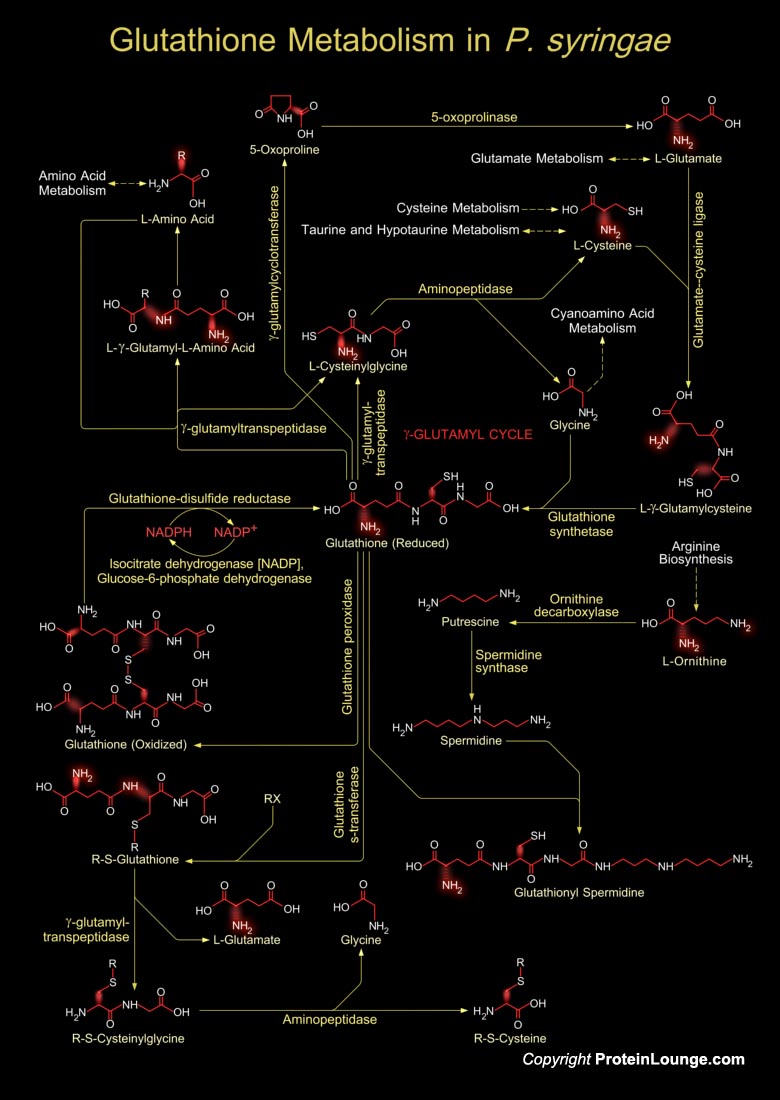
Pseudomonas syringae is one of the best studied plant pathogen with more than 50 pathovars. Each pathovar is known to infect a characteristic group of host plant species (Ref.1). P. syringae is a Gram-negative bacterium and its different strains are known for their diverse interactions with plants. Among these strains, P. syringae pv. tabaci is a non-host pathogen of Arabidopsis thaliana (Ref.2). P. syringae survives saprophytically on the plant surface, and after the entering plant tissues through wounds or natural openings (such as stomata), it reproduces endophytically in the apoplastic space (Ref.3). P.syringae has evolved two principal virulence strategies which are a) suppression of host immunity b) creation of an aqueous apoplast (Ref.1). To survive both in the[..]
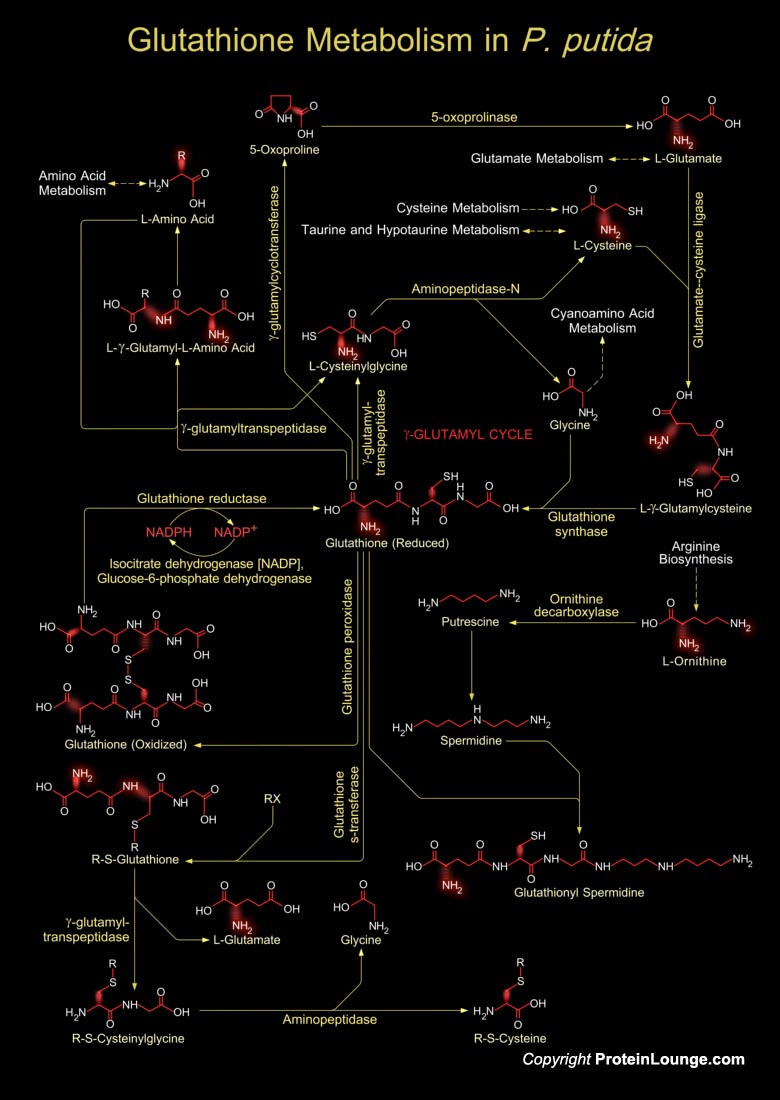
Pseudomonas putida is a Gram-negative, rod-shaped, saprotrophic soil bacterium (Ref.1). It has been reported as opportunistic human pathogens capable of causing nosocomial infections. P. putida exhibits an amazing ability to metabolize a wide range of biogenic and xenobiotic compounds (Ref.2). As a frequent inhabitant of sites polluted with toxic chemicals, P.putida can tolerate high levels of endogenous and exogenous oxidative stress. It is equipped with the enzymatic machinery needed to catabolism both natural and recalcitrant aromatic compounds besides sugars and organic acids. Thiols play several roles in bacteria like maintaining the redox balance, quenching ROS and nitrogen reactive species, and detoxifying other toxins and stress-inducing factors. In[..]
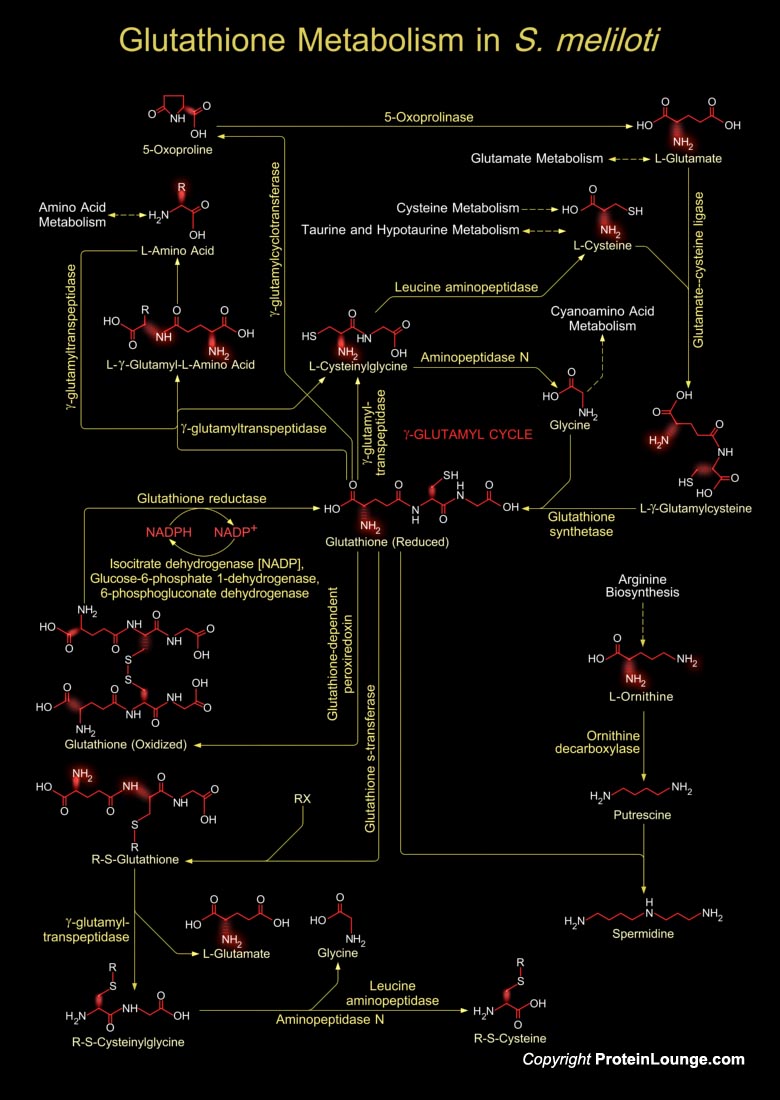
Glutathione is a sulfhydryl (-SH) antioxidant, antitoxin, and enzyme cofactor. It is ubiquitous in animals, plants, and microorganisms, and being water soluble is found mainly in the cell cytosol and other aqueous phases of the living system. Glutathione is a tripeptide composed of Glutamate, Cysteine and Glycine that has numerous important functions within cells. Glutathione is homeostatically controlled, both inside the cell and outside. It often attains millimolar levels inside cells, which makes it one of the most highly concentrated intracellular antioxidants. Glutathione exists in two forms. The antioxidant "reduced Glutathione" tripeptide is conventionally called Glutathione and abbreviated Gsh; the oxidized form is a sulfur-sulfur linked compound,[..]
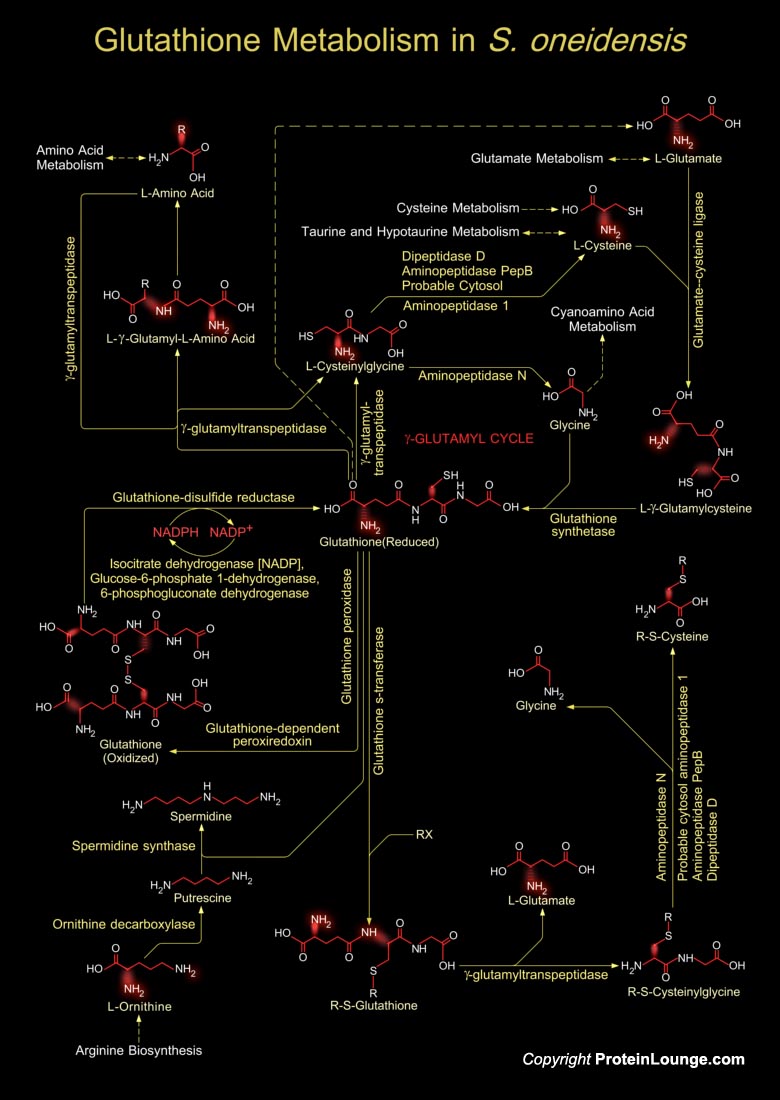
Shewanella oneidensis is a facultative aerobic Gram-negative bacterium. It uses oxygen as the terminal electron acceptor during aerobic respiration, but during anaerobic conditions, S. oneidensis undertakes respiration by reducing alternative terminal electron acceptors such as oxidized metals, fumarate, nitrate etc. The microbe can directly reduce both uranium and chromium from the dissolved liquid state. Such abilities facilitate the removal of dilute metal pollutants in both contained-storage and natural sites. Additionally S. oneidensis immobilizes toxic metals through the formation of insoluble metal sulfides. Thus, the bacterium is an important model organism for bioremediation studies because of its diverse respiratory capabilities, conferred in part by[..]
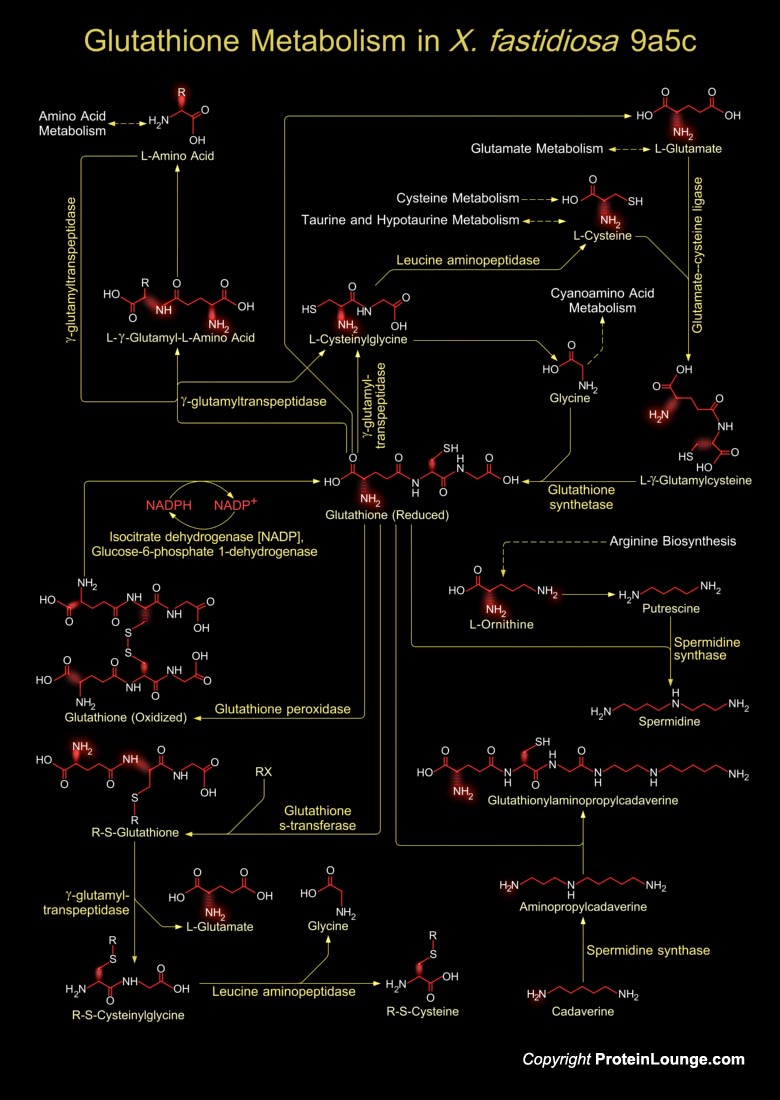
Xylella fastidiosa is a Gram-negative, fastidious, xylem-limited bacterium that causes a range of economically important plant diseases. It causes citrus variegated chlorosis-a serious disease of orange trees. It is responsible for pathogenicity and virulence involving toxins, antibiotics and ion sequestration systems. Glutathione is a tripeptide present in Xylella sp., which is composed of Glutamate, Cysteine and Glycine, and has numerous important functions within cells. The tripeptide is the thiol compound; present in the highest concentration in all types of cells (Ref.1 & 2).Glutathione metabolism in X. fastidiosa involves both the synthesis of Glutathione and its catabolism. Glutathione biosynthesis starts from an L-Amino acid, which in presence of the enzyme[..]
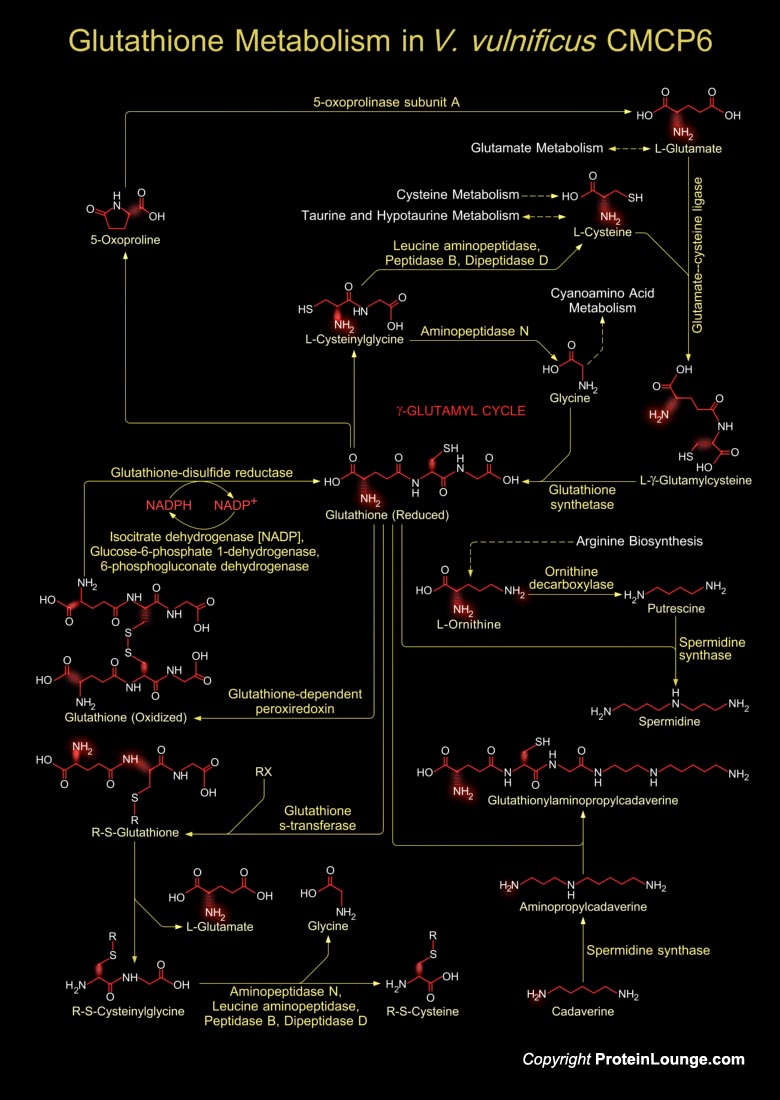
Vibrio vulnificus is an etiologic agent for severe human infection acquired through wounds or contaminated seafood. This is a lactose-fermenting, halophilic, Gram-negative, opportunistic pathogen, is found in estuarine environments and is associated with various marine species such as plankton, shellfish (Oysters, Clams, and Crabs), and finfish (Ref.1). V. vulnificus belong to the Gamma-group of Proteobacteria, and it shares morphological and biochemical characteristics with other human Vibrio pathogens, including Vibrio cholerae and Vibrio parahaemolyticus. It is divided into three biotypes according to its different biochemical and biological properties. V. vulnificus CMCP6 is a new strain of this bacterium, classified as Biotype-3 and is the sole cause of the[..]
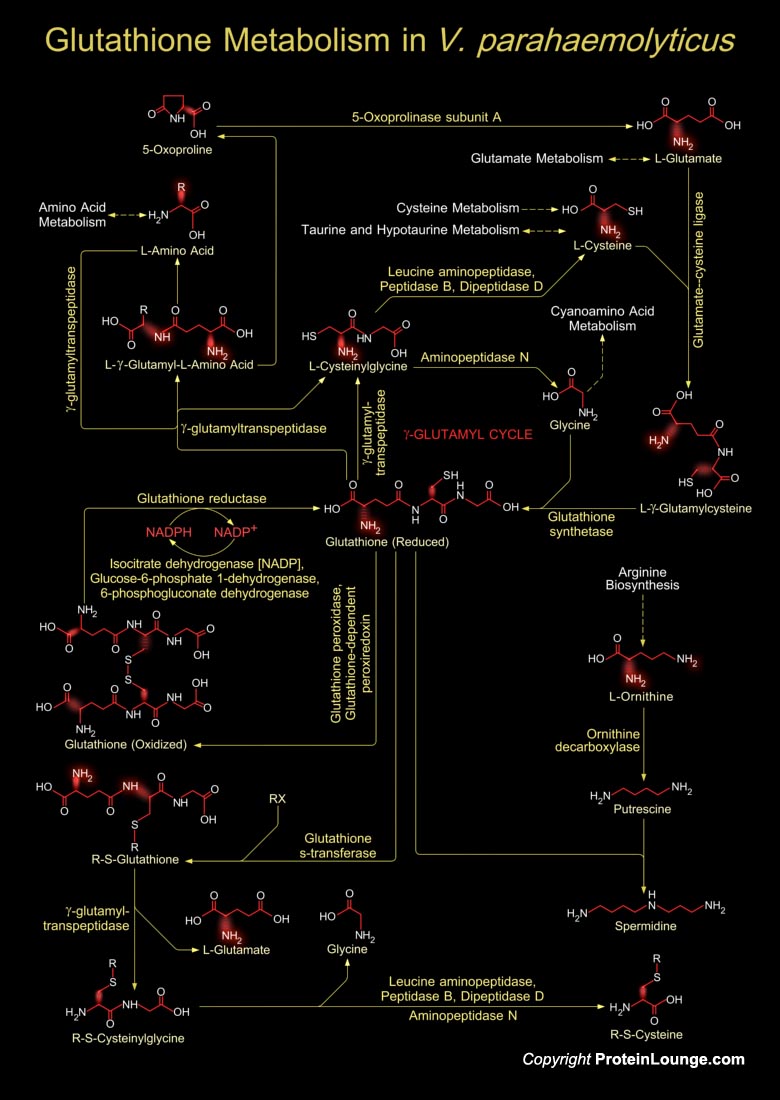
Vibrio parahaemolyticus, a Gram-negative marine bacterium, is a worldwide cause of food-borne gastroenteritis. The organism is phylogenetically close to V. cholerae, the causative agent of cholera. This universal marine pathogen is used as a bacterial model to clarify the various physiological phenomena of its native and host environments (Ref.1 & 2).Glutathione in V. cholerae is a tripeptide composed of Glutamate, Cysteine and Glycine, and has numerous important functions within the bacterial cell. This tripeptide is specifically a thiol compound, present in the highest concentration in all types of cells. During Glutathione metabolism, the laevorotatory amino acids present in the bacterium are used as precursors for ultimately synthesizing Glutathione, the[..]
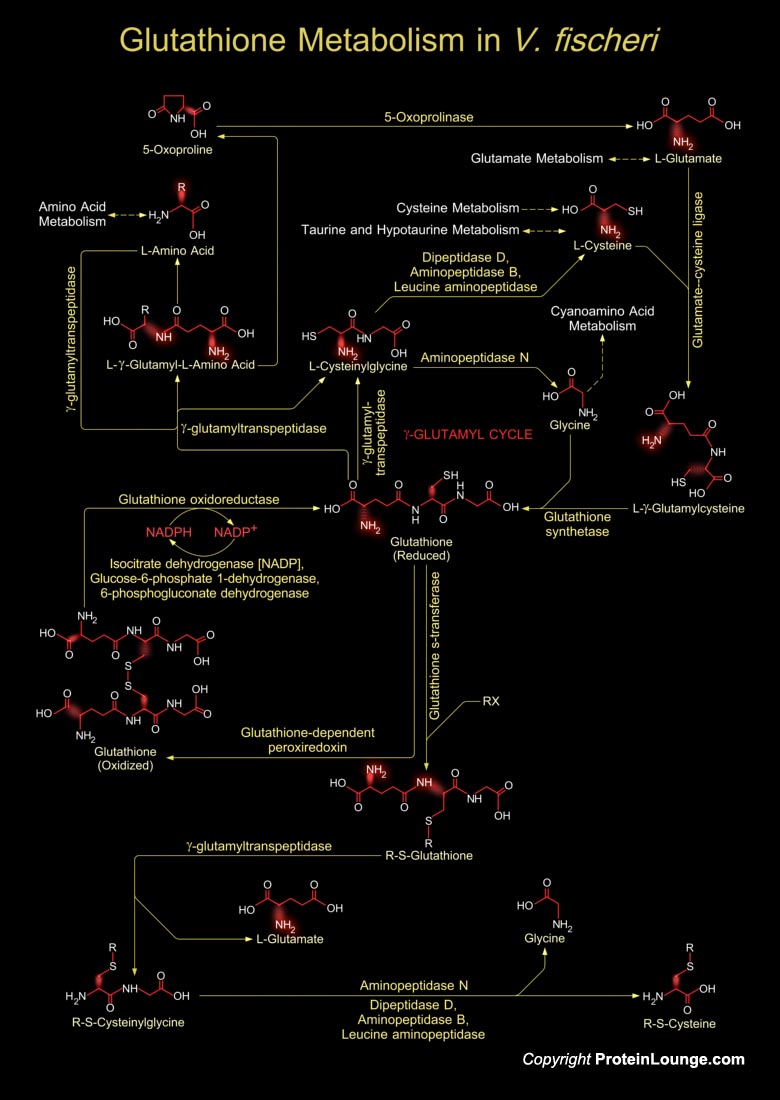
Vibrio fischeri is a Gram-negative heterotrophic bacterium, belonging to the Vibrionaceae, a large family within the Gamma-proteobacteria, consisting of many species that are characterized by both cooperative and pathogenic interactions with animal tissue. V. fischeri has a worldwide distribution, principally in temperate and subtropical waters, where it occupies a variety of niches. In addition to being a light-organ symbiont of several species of squids and fishes, this bacterium occurs as a member of the enteric consortia of many marine animals, as a pathogen of certain invertebrates and as a 'free-living' saprophyte growing on dissolved and particulate organic matter. In locations where it forms light-organ symbiosis with animals, free-living V. fischeri[..]
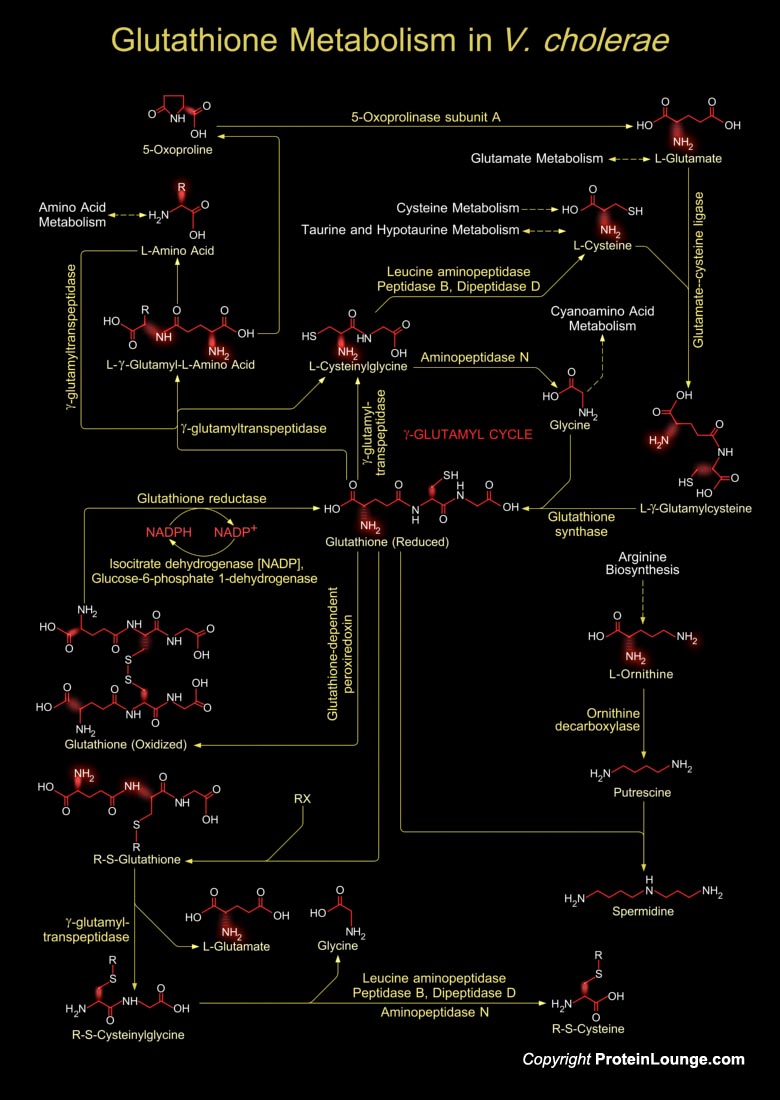
Vibrio cholerae is a facultative anaerobic, Gram-negative, crescent-shaped, motile rod like bacterium, and the causative infectious agent of the diarrheal disease, Cholera. It colonizes the mucosal surface of the human small intestine and secretes cholera toxin. The toxin stimulates secretion of water and electrolytes by the cells of the small intestine, leading to the severe watery diarrhoea that is characteristic of cholera. Unlike most bacterial agents of diarrhea, V. cholerae tends to survive in aquatic environments during periods between epidemics and can give rise to dramatic outbreaks of disease(Ref.1).Glutathione in V. cholerae is a tripeptide, composed of glutamate, cysteine and glycine, and has numerous important functions within the bacterial cell. This[..]
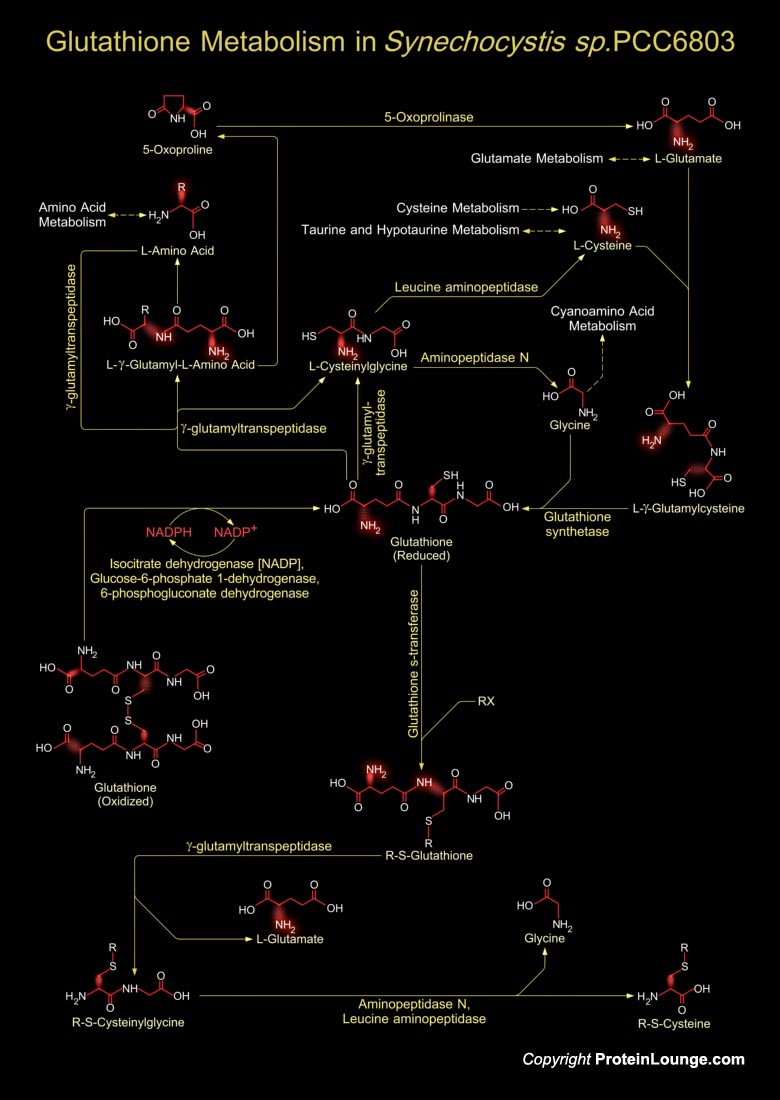
Synechocystiae are unicellular, photoautotrophic, facultative glucose-heterotrophic bacteria. They are oxygenic photosynthetic with two photosystems at their disposal, similar to those in algae and plants, and they can fix nitrogen. Synechocystis sp. PCC6803 can grow in the absence of photosynthesis if a suitable fixed-carbon source such as glucose is provided. The total length of Synechocystis sp. strain PCC6803 is 3,573,470 bp and it has developed into a Cyanobacterium model. Glutathione metabolism in Synechocystis sp. involves both the synthesis of Glutathione and its catabolism. Glutathione is a small molecule found in almost every cell. It cannot enter most cells directly and therefore must be made available inside the cell from its three constituent amino acids:[..]

