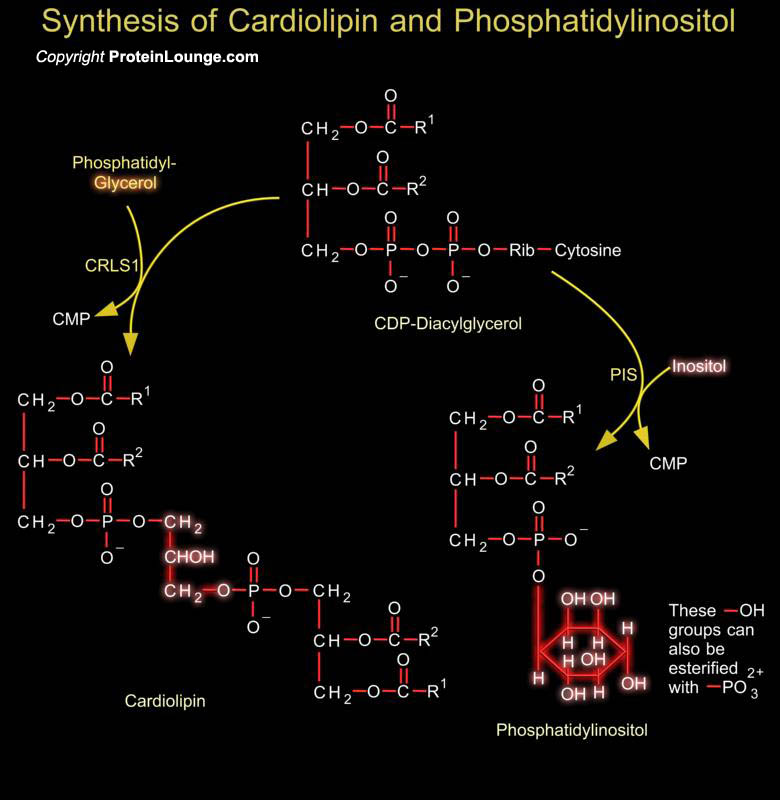
Phosphoglycerides such as phosphatidylethanolamine and phosphatidylserine are the major component of lipid bilayer membranes in which their detergent-like properties help to provide membranes the quality of self-assembly. Phosphatidylglycerol is a precursor in the synthesis of both cardiolipin and PI (Phosphatidylinositol). Cardiolipin is a unique dimeric phospholipid found in the heart and most mammalian tissues. This phospholipid is the only phospholipid localized primarily in the mitochondrial membrane of mammalian cells. In cardiolipin, two phosphatidylglycerol units are joined together. Phosphatidic acid is activated to CDP-DAG (CDP-Diacylglycerol), which is the precursor to PI and cardiolipin. PI is the precursor of PI Phosphates, which have one of more[..]
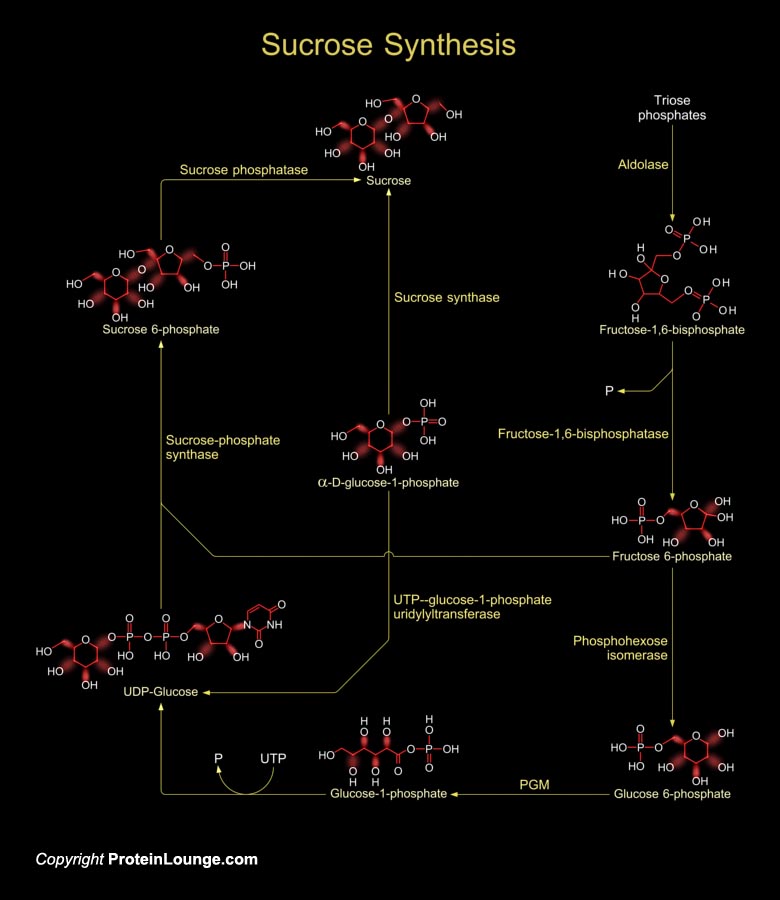
Sucrose is produced by plants, algae and cyanobacteria as an end product of photosynthesis. It is the primary sugar transported from the source tissues to sink tissues by the phloem in most plants. In other non-photosynthetic tissues, sucrose serve as raw material for many metabolic pathways (Ref.1).Sucrose synthesis starts in the cytosol where two triose-phosphates produce one Fructose-1,6-bisphosphate by the enzyme aldolase. Fructose-1,6-bisphosphate is converted to fructose-6-phosphate by the enzyme Fructose-1,6-bisphosphatase. Then the enzyme phosphohexose isomerase converts fructose-6 phosphate to glucose-6-phosphate. Glucose-6-phosphate then forms glucose-1-phosphate in presence of the enzyme phosphoglucomutase (PGM). The glucose-1-phosphate is converted to[..]
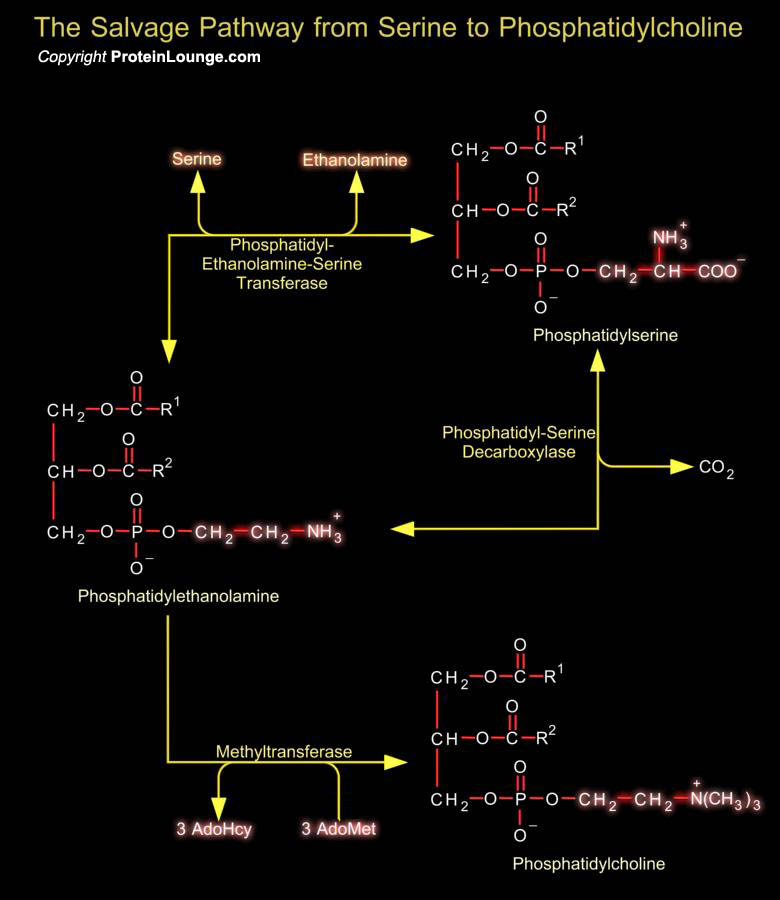
Phosphatidylserine (or 1, 2-Diacyl-sn-Glycero-3-Phospho-L-Serine) is the only amino acid-containing glycerophospholipid in animal cells. Although it is distributed widely among animals, plants and microorganisms, it is usually less than 10% of the total phospholipids, the greatest concentration being in myelin from brain tissue. It is an acidic (anionic) phospholipid with three ionizable groups, i.e. the phosphate moiety, the amino group and the carboxyl function. As with other acidic lipids, it exists in nature in salt form.In bacteria and other prokaryotic organisms, Phosphatidylserine is synthesized by a mechanism comparable to that of other phospholipids, i.e. by reaction of L-Serine with CDP-DAG (CDP-Diacylglycerol).In contrast in animal tissues, there are two[..]
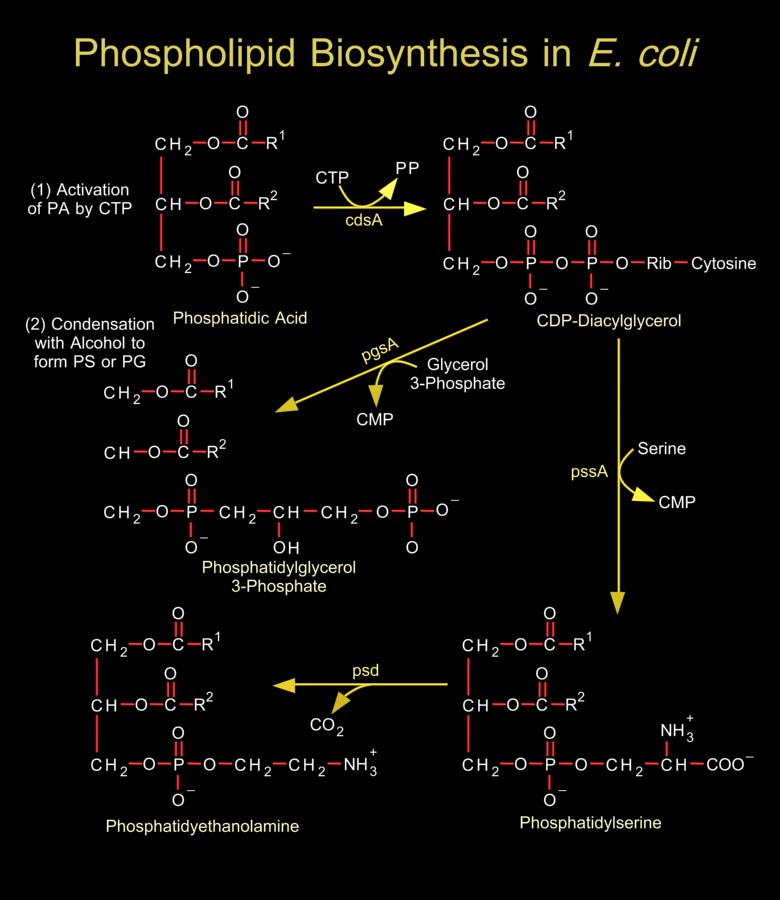
Biological membranes are composed of hundreds of distinct proteins and phospholipids. Phospholipids are diacylglycerol derivatives with a hydrophilic, zwitter ionic, often charged headgroup at position C3 of the glycerol backbone. The properties of phospholipids give lipid bilayer membranes their self-organizing structure. Phospholipids are usually composed of two fatty acid chains esterified to two of the carbons of glycerol phosphate, the phosphate being esterified to a hydroxyl group of another hydrophilic compound, such as choline, ethanolamine or serine.In general, E. coli and most related gram-negative bacteria do not contain phosphatidylcholine, phosphatidylinositol, sphingolipids, glycolipids, plasmalogens, or sterols, which are characteristic of[..]
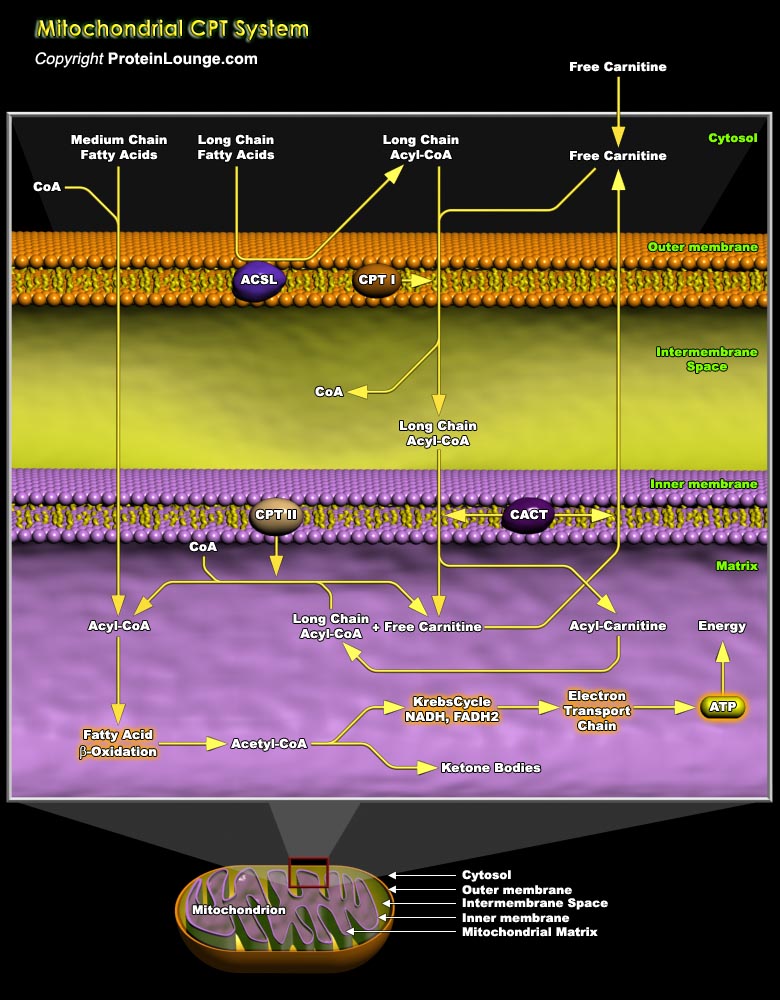
Mitochondrial fatty acid oxidation (FAO) and its key rate-limiting enzyme, the carnitine palmitoyltransferase (CPT) system, are important for energy homeostasis in situations like fasting or during exercise. They also regulate host immune responses and the deficiency or over-activation of CPT may cause energy metabolism disorder that may lead to many diseases starting from inflammatory disorders to cancer. Fatty acid catabolism occurs mostly in mitochondria through the beta-oxidation pathway (Ref.1 and 2). Long-chain fatty acids (LCFA), major component of fatty acids, that are important source of energy for heart, liver and muscles, cannot enter the mitochondria by simple diffusion unlike short or medium chain fatty acids. LCFA are activated and converted to[..]
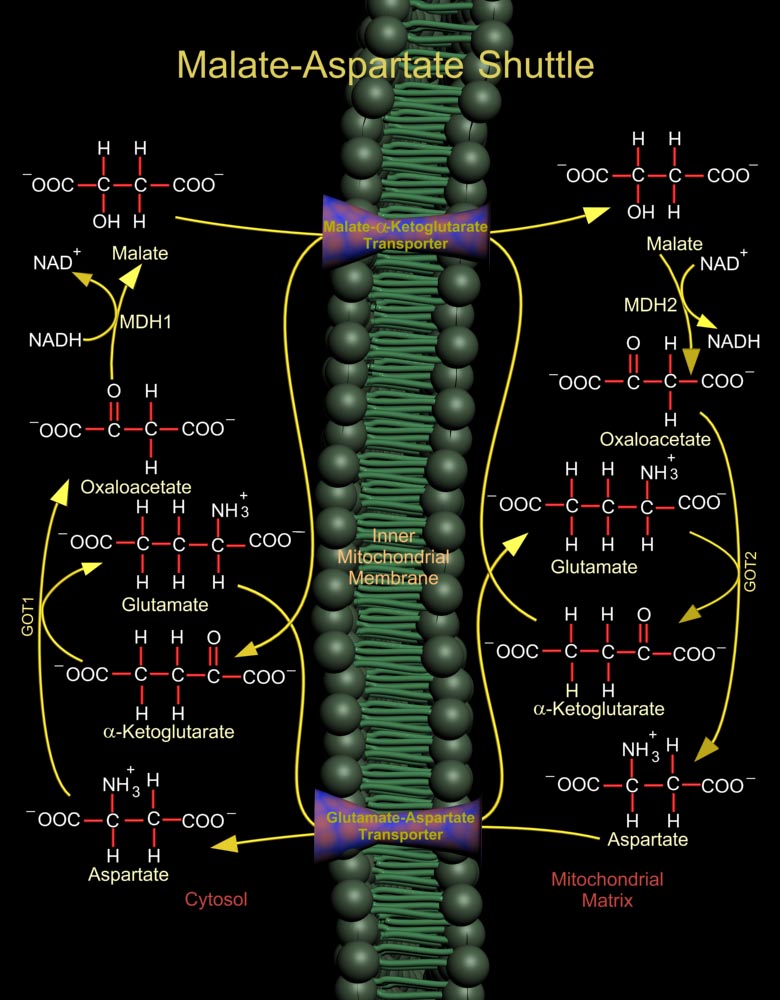
The Malate (L-Malic Acid)-Asp (Aspartate or L-Aspartate or Aspartic Acid) Shuttle of mammalian systems is more complex but more energy efficient. Mitochondrial NAD+ (Nicotinamide Adenine Dinucleotide) is reduced by cytosolic NADH (Nicotinamide Adenine Dinucleotide, Reduced) through the intermediate reduction and subsequent regeneration of OAA (Oxaloacetate) (Ref.1). In the cytosol, the shuttle converts OAA to Malate using the enzyme MDH1 (Malate Dehydrogenase-Cytoplasmic), and at the same time re-oxidizes NADH to NAD+, making it available for Glucose oxidation. Malate is then shuttled into the mitochondria by a Malate-Alpha-Ketoglutarate Transporter. In the mitochondria, the reverse reaction takes place and Malate is again converted to OAA by the enzyme MDH2 (Malate[..]
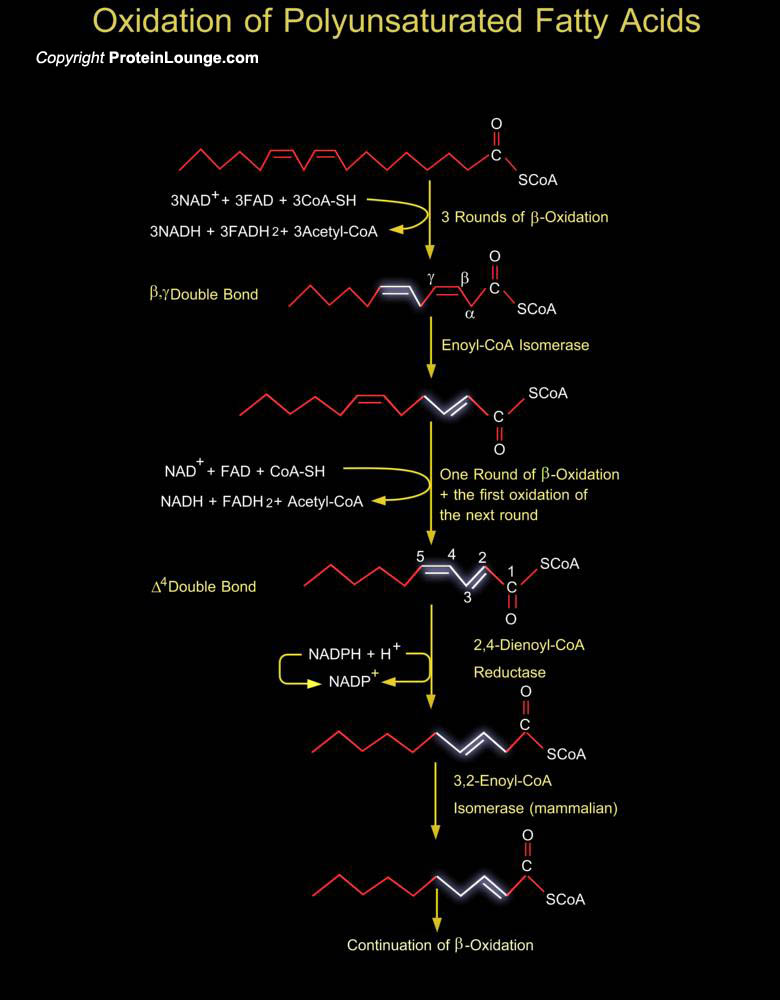
Unsaturated fatty acids are Beta-oxidized in the normal pathway as far as possible. However, polyunsaturation or unsaturation at odd positions produces a molecule that the major pathways cannot utilize as a substrate. To solve this problem, a number of enzymes exist in the peroxisome to convert these molecules to appropriate substrates which can be shuttled into the normal pathway. Peroxisomes are capable of oxidizing a variety of substrates including polyunsaturated Enoyl-CoA esters. The Beta-Oxidation of unsaturated Enoyl-CoA esters in peroxisomes, and also in mitochondria, is not just chain-shortening but also involves the metabolizing of pre-existing carbon-to-carbon double bonds. In addition to the enzymes of the Beta-Oxidation spiral itself, this metabolism[..]
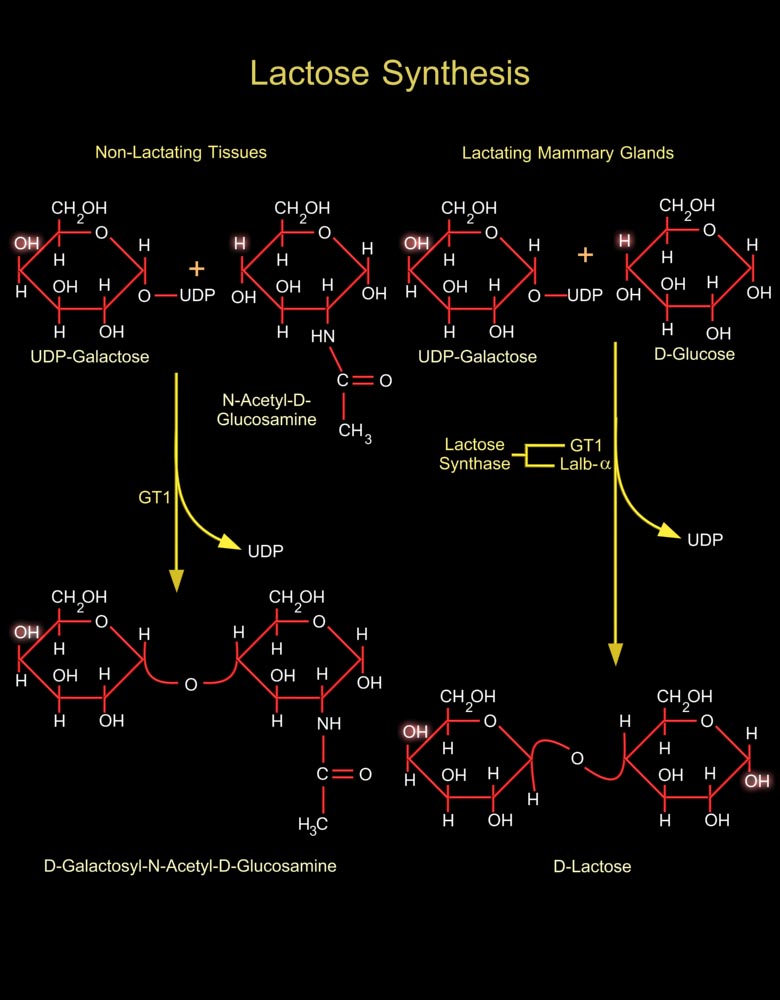
Lactose (or D-Lactose) is a disaccharide composed of Galactose (or Galactose) and Glucose (or D-Glucose). It occurs only in milk. It is the primary carbohydrate in milk of most species. Lactose is synthesized in mammary glands. Two Glucose molecules are required for each Lactose molecule synthesized. One Glucose is converted to UDP-Glucose (Uridine Diphosphate-Glucose), which in turn is converted to one UDP-Galactose (Uridine Diphosphate-Galactose) by GALE (UDP-Galactose-4-Epimerase) (Ref.1). Another Glucose molecule is used for Lactose synthesis without modification. Glucose passes across the Golgi apparatus membrane into the Golgi apparatus lumen by GLUT1 (Glucose Transporter-1). The presence of GLUT1 on the Golgi apparatus membrane apparently is specific to the[..]
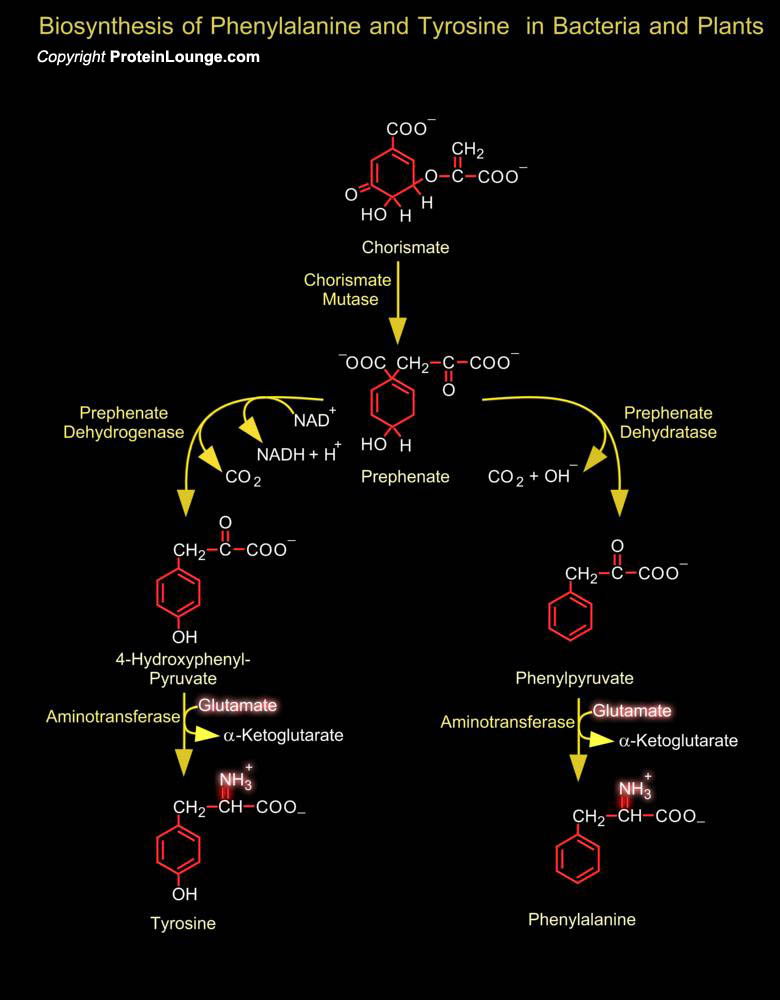
Chorismate is a branch point for different pathways leading to Phenylalanine and Tyrosine. A mutase reaction yields Prephenate, which undergoes either a DehydrataseDecarboxylase or a reductionDecarboxylase reaction (in some bacteria) resulting in aromatization, the oxo analogues of phenylalanine and tyrosine obtained in this way are then converted into the respective amino acids by transamination. In chloroplasts of higher plants, Prephenate is first transaminated to Arogenate, while the DehydrataseDecarboxylase reactions take place afterwards. Mammals obtain tyrosine from Phenylalanine by a hydroxylation reaction catalyzed by phenylalanine 4-monooxygenase, which uses Tetrahydrobioperin as a hydrogen donor. The reduced Tetrahydrobioperin is regenerated by NAD (P)H,[..]
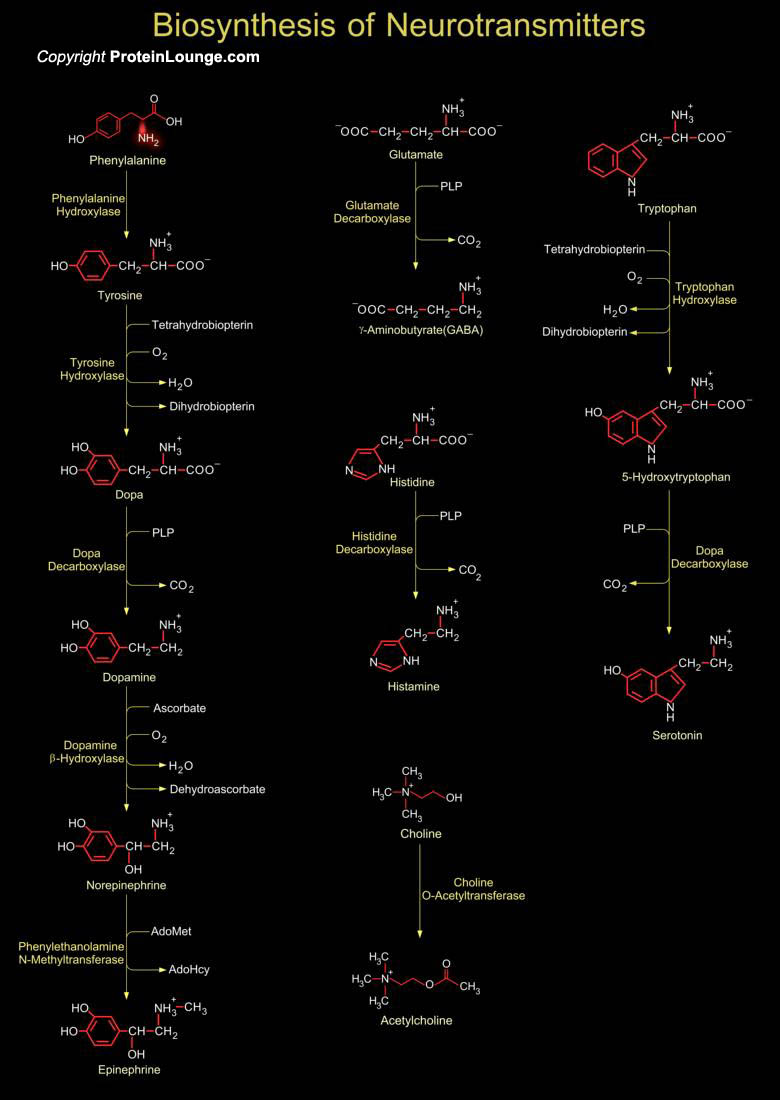
For neuronal signal transduction, the presynaptic cell synthesizes neurotransmitters that then traverse the synaptic cleft. Neurotransmitters are signaling molecules used by neurons to communicate across chemical synapses. The receptors for neurotransmitters include GPCRs (G-Protein Coupled Receptors) and ligand-gated ion channels. The biogenic amines are neurotransmitters derived from amino acids, and include Dopamine, Epinephrine, GABA, Histamine and Serotonin. For each of these, decarboxylation of an amino acid is a key step. Amino acid decarboxylases are PLP-Schiff base with the substrate so as to stabilize the C-alpha carbanion formed upon C-alpha-COO- bond cleavage. Formation of GABA and Histamine are one step processes. GABA (Gamma-aminobutyric acid) is[..]
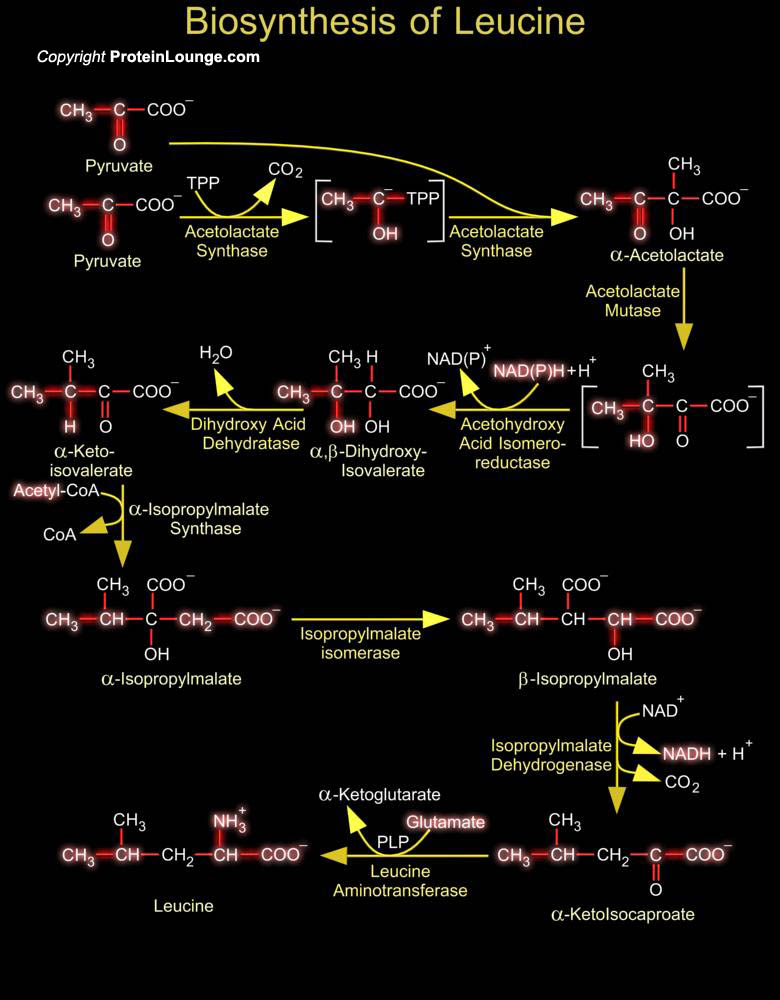
Isoleucine is essential amino acid, only synthesized in plants and bacteria, and required in the diet by animals. In proteins, the hydrophobic Isoleucine side-chain tends to reside with other hydrophobic residues in the interior of globular proteins or in transmembrane domains. Isoleucine biosynthesis begins with the common metabolic intermediate Pyruvate, the endpoint of glycolysis. The first step in Isoleucine biosynthesis requires TPP (Thiamine Pyrophosphate) to form a carbanion intermediate. This intermediate undergoes decarboxylation to produce a stabilized anion of Hydroxyethyl-TPP (HET-PP), which acts as a nucleophile on the Alpha-keto group of a second molecule of Pyruvate or Alpha-ketobutyrate, releasing TPP and Acetolactate or Acetohydroxybutyrate. Another[..]
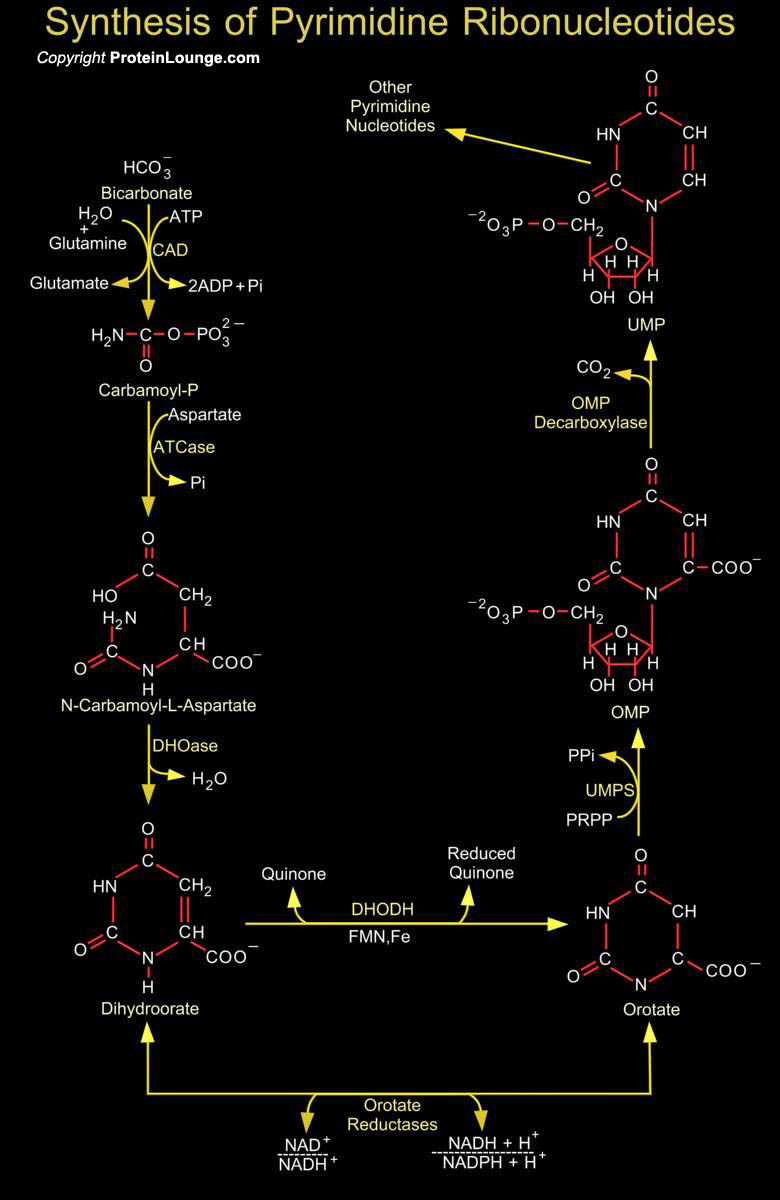
Synthesis of the Pyrimidines is less complex than that of the Purines, since the base is much simpler. Synthesis of Carbamoyl-P (Carbamoyl Phosphate) is the first reaction of Pyrimidine biosynthesis. Carbamoyl-P is formed from HCO3- (Bicarbonate) and the amide nitrogen of Glutamine by the cytosolic enzyme CPSase (Carbamoyl Phosphate Synthetase). This reaction consumes two molecules of ATP (Adenosine Monophosphate): One provides a phosphate group and the other energizes the reaction. Then condensation of Carbamoyl-P with Asp (Aspartate or L-Aspartate or Aspartic Acid) occurs to form CAA (N-Carbamoyl-L-Aspartate) which is catalyzed by ATCase (Aspartate Carbamoyltransferase). This reaction is the flux-generating step and occurs without need of ATP because Carbamoyl-P is[..]

