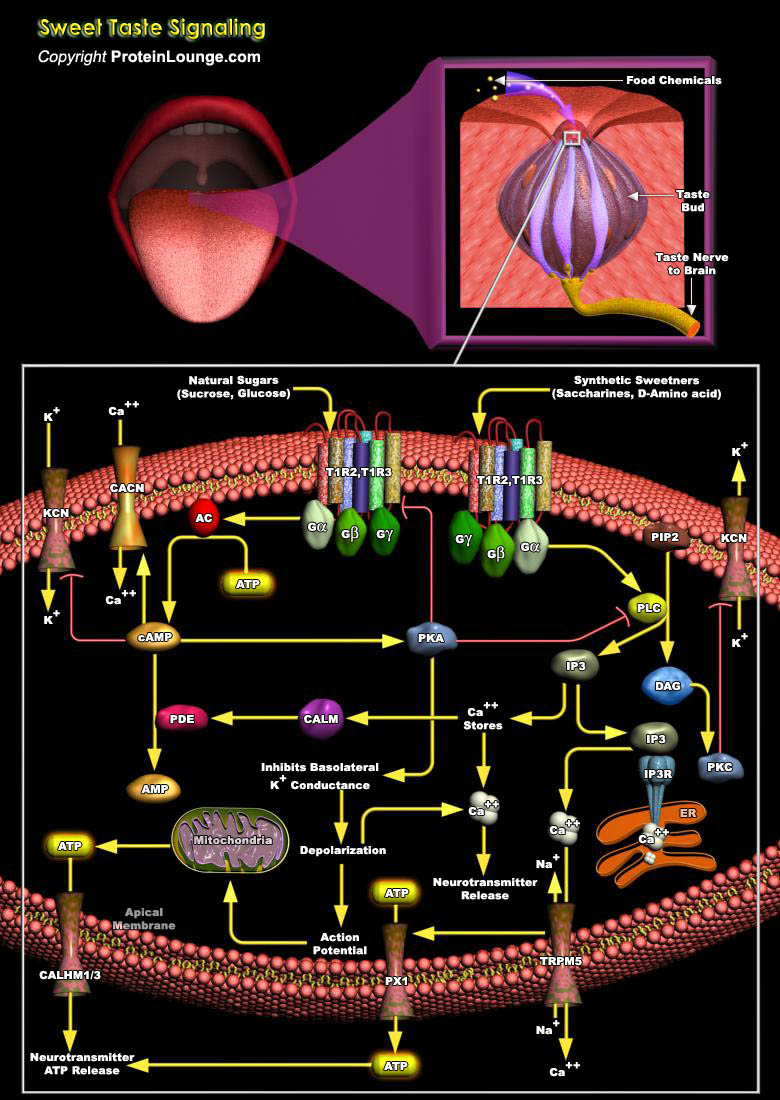
The sense of taste plays a critical role in the life and nutritional status of humans and other organisms. Human taste perception may be categorized according to four well known and widely accepted descriptors, sweet, bitter, salty, and sour (corresponding to particular taste qualities or modalities), and two more controversial qualities: fat and amino acid taste.Taste is a sensory modality involving the oral perception of food-derived chemicals that stimulate receptor cells within taste buds. Taste principally serves two functions: it enables the evaluation of foods for toxicity and nutrients while helping us decide what to ingest and it prepares the body to metabolize foods once they have been ingested. Taste percepts are elicited by molecules that stimulate the[..]
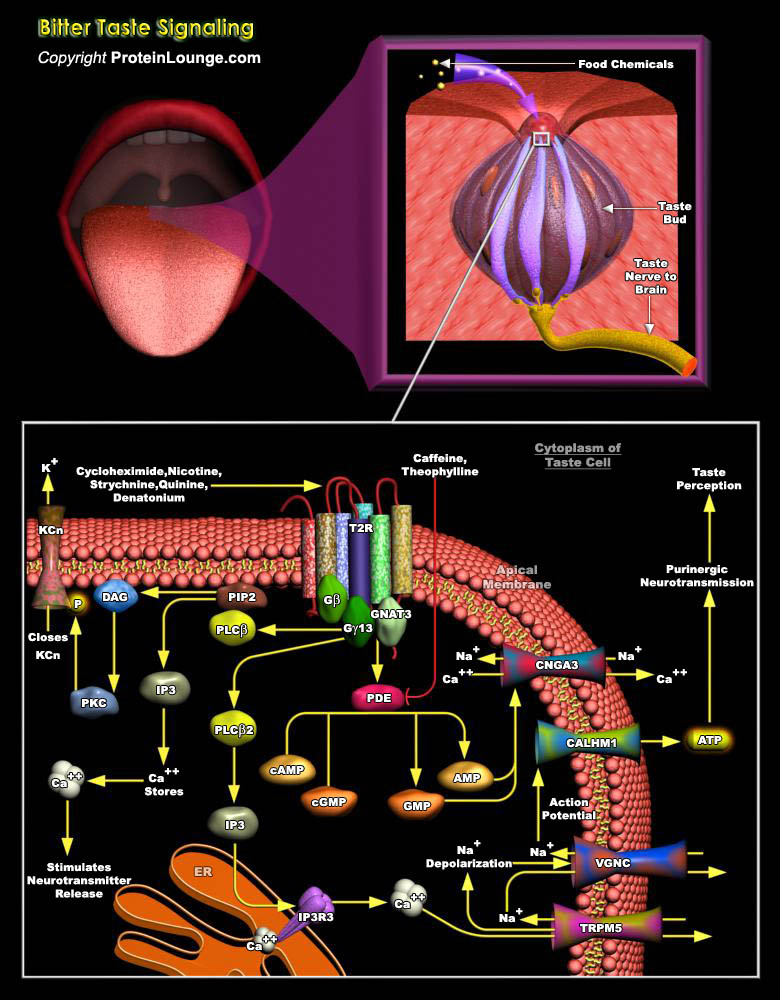
The sense of taste plays a critical role in the life and nutritional status of humans and other organisms. Human taste perception may be categorized according to four well known and widely accepted descriptors, sweet, bitter, salty, and sour (corresponding to particular taste qualities or modalities), and two more controversial qualities: fat and amino acid taste. In addition to these basic tastes, the taste buds also detect a meat-like taste known as umami in protein-rich food. The food chemicals that produce tastes set off different reactions from the taste buds with a main goal of sending signals along nerve fibers to the brain for interpretation. The perception of bitter taste is essential for its protective value, enabling humans to avoid potentially deadly plant[..]
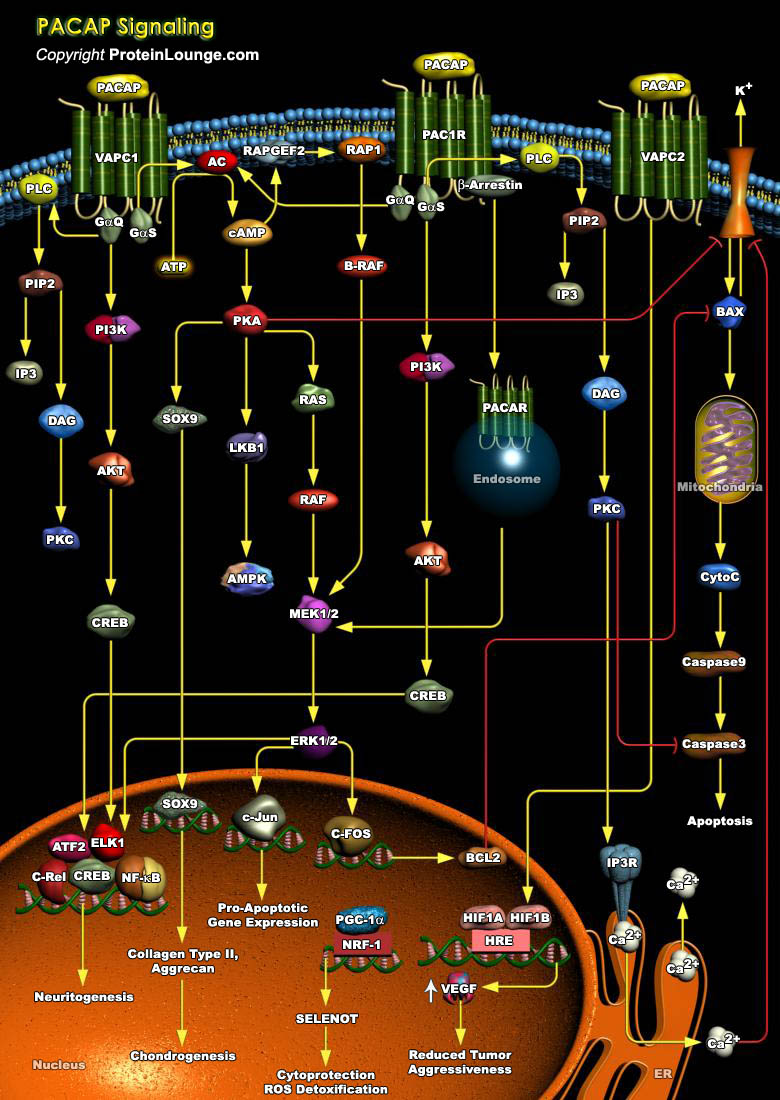
G Protein-coupled Receptors (GPCRs) based on sequence and structural similarities have been divided into five major classes: Rhodopsin (class A), Secretin (class B), Glutamate (class C), Adhesion, and Frizzled/Taste. A number of critical regulatory peptides, such as corticotropin-releasing hormone, calcitonin gene-related peptide, parathyroid hormone, glucagon, vasoactive intestinal peptide (VIP) and pituitary adenylate cyclase-activating polypeptide (PACAP) activate the Rhodopsin (class A) GPCRs. These regulatory peptides are important for neural development, calcium homeostasis, glucosemetabolism, circadian rhythm, thermoregulation, inflammation,feeding behaviour, pain modulation, as well as stress andrelated endocrine responses. ADCYAP1 (Adenylate Cyclase Activating[..]
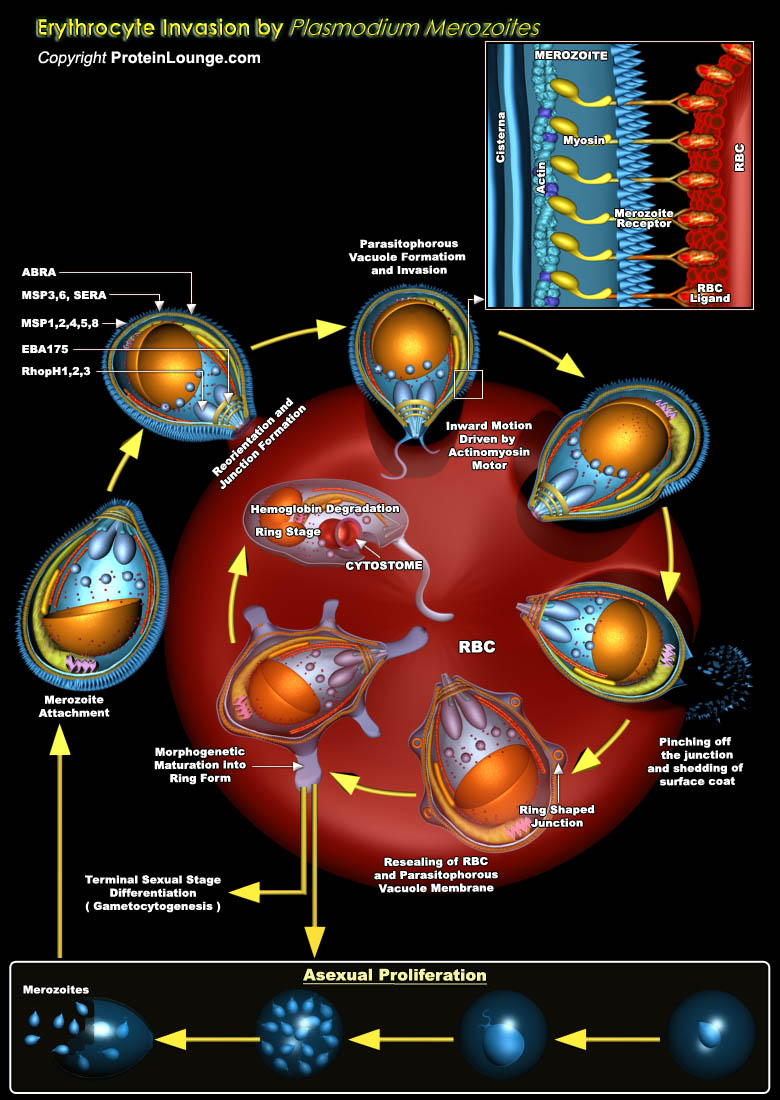
Malaria is caused by infection with four species of the intracellular protozoan parasite Plasmodium that are transmitted by Anopheles mosquitoes. Most severe form of malaria in human is caused by Plasmodium falciparum, with above 300 million people affected by malaria each year and about 2 million deaths (Ref.1). Life cycle of Plasmodium in human starts with a bite from an infected mosquito and an initial phase of infection and replication in liver cells: the exo-erythrocytic phase, where merozoites are formed. These merozoites then enter the bloodstream beginning the asexual bloodstage lifecycle of this parasite to known as erythrocytic phase where they may proliferate indefinitely in red blood cells (RBCs) (Ref.2). Merozoite first invades the host RBC through a[..]
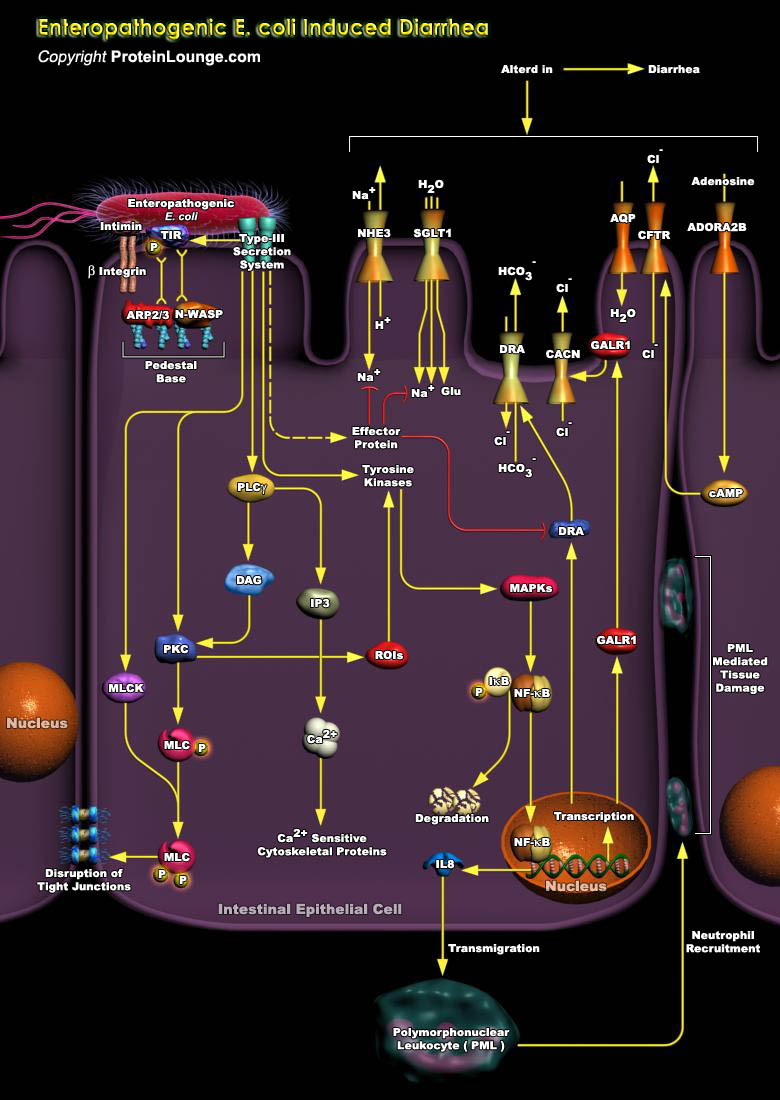
Pathogenic microbes subvert normal host-cell processes to create a specialized niche, which enhances their survival. A common and recurring target of pathogens is the host cell's cytoskeleton, which is utilized by these microbes for purposes that include attachment, entry into cells, movement within and between cells, vacuole formation and remodeling, and avoidance of phagocytosis. One such subversive microbe is a gram-negative pathogen EPEC (Enteropathogenic Escherichia coli) that causes chronic, watery diarrhea in humans, primarily young children and infants. Central to EPEC-mediated disease is its colonization of the intestinal epithelium. After initial adherence, EPEC causes the localized effacement of microvilli and intimately attaches to the host cell surface,[..]
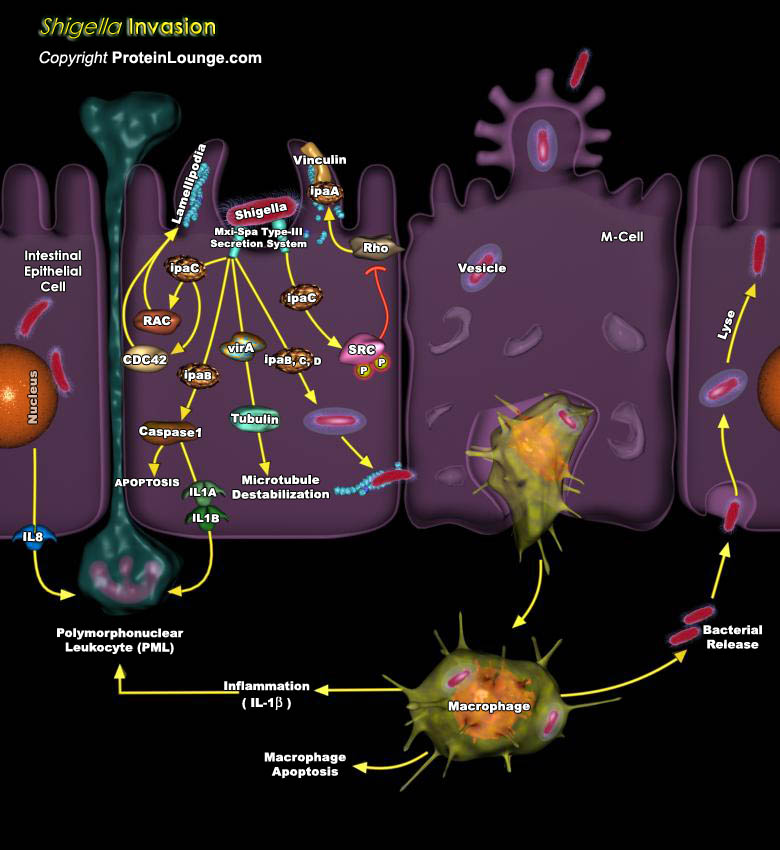
Bacterial pathogens possess highly specialized adaptive processes that enable their penetration of the host intestinal epithelium and cause disease. Once bound to the epithelial surface, bacteria may colonize and establish a permanent residence in the gut. Some Gram-negative pathogenic bacteria have acquired sophisticated 'molecular syringes', such as Type-III or Type-IV secretion systems, which are multisubunit molecular machines that span the bacterial and host membranes and translocate effectors directly into host cells. Shigella flexneri is a gram-negative facultative intracellular pathogen causing disease by invading the colonic mucosa. When Shigella reaches the colon, the bacteria are translocated through the epithelial barrier by way of the M-Cells of[..]
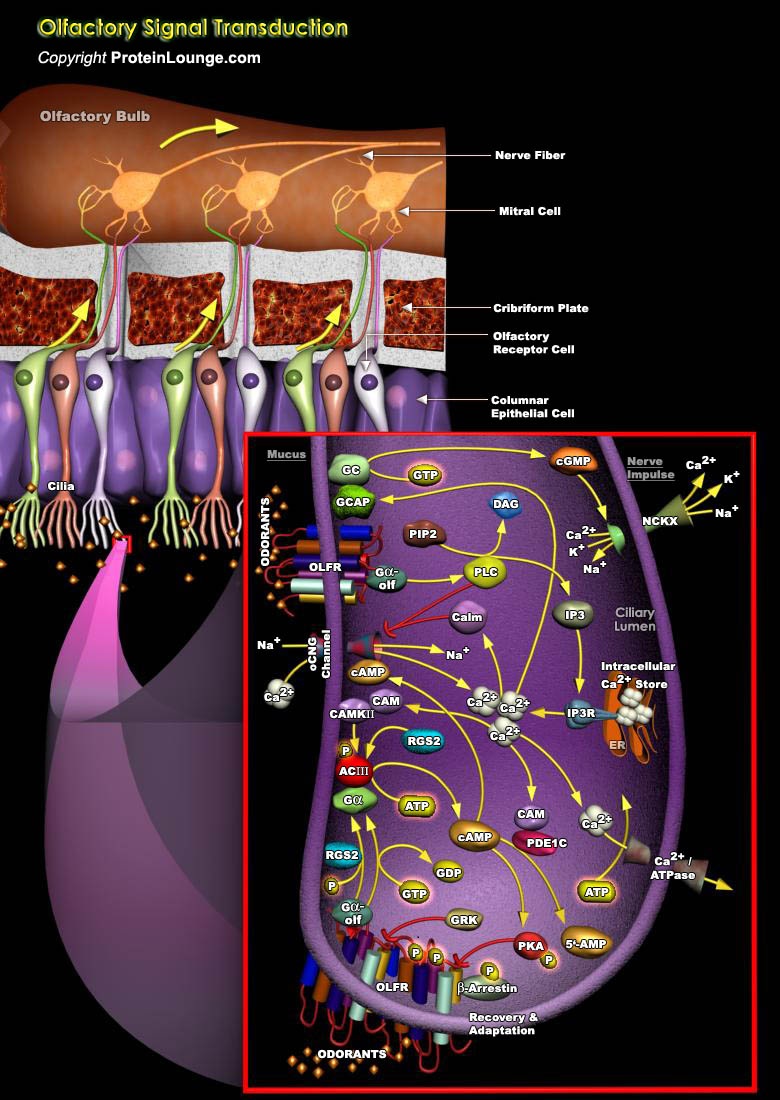
The olfactory system is a very efficient biological setup capable of odor information processing with neural signals. The mammalian olfactory system can recognize and discriminate a large number of different odorant molecules. The detection of chemically distinct odorants presumably results from the association of odorous ligands with specific receptors on OSNs (Olfactory Sensory Neurons). As a chemical sensor, the olfactory system detects food and influences social and sexual behavior. Activation occurs when odiferous molecules come in contact with specialized processes known as the olfactory vesicles. Within the nasal cavity, the turbinates or nasal conchae serve to direct the inspired air toward the olfactory epithelium in the upper posterior region. This area[..]
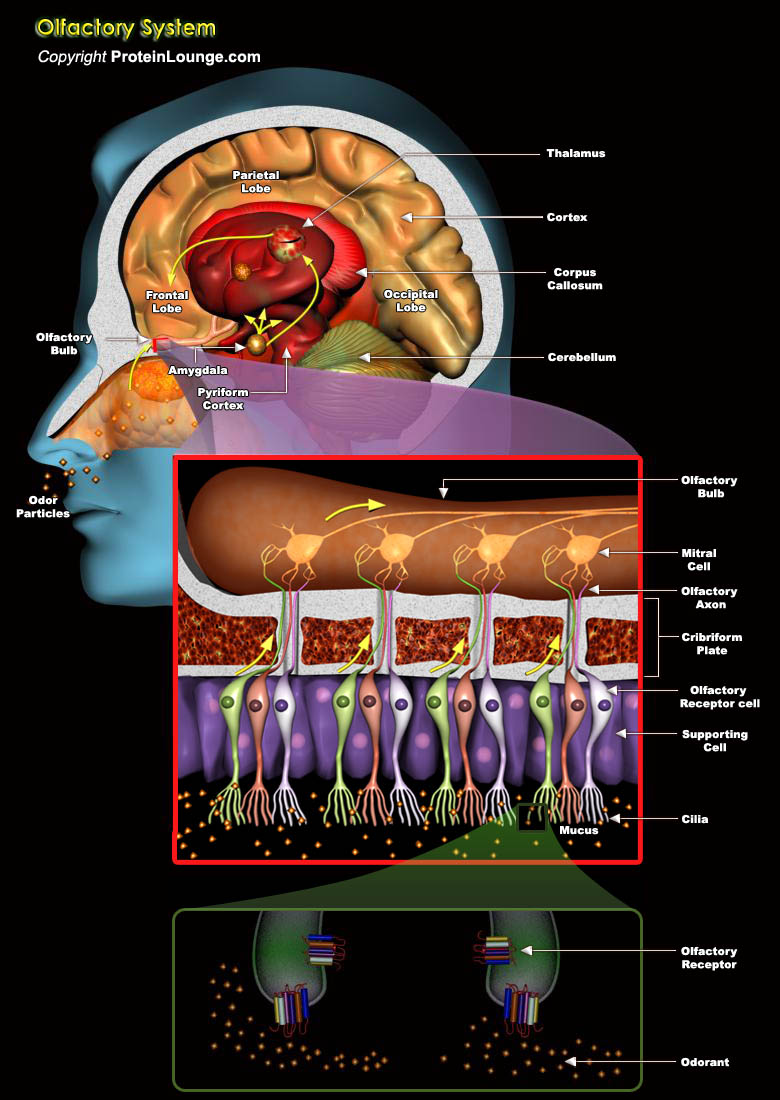
Our chemical senses, taste and smell, are our oldest senses. Long before organisms could hear or see, they needed to find nutrients, had to avoid toxic substances, and they were attracted by signaling molecules that were released by their mating partners. Right from the beginning of evolution, the task of our chemical senses was not to merely identify the chemical compounds encountered by the organism, but to directly initiate behavioral responses. This sense of smell has long remained the most enigmatic of our senses. The basic principles for recognizing and remembering about 10,000 different odors were not understood. To address the problem of olfactory perception at a molecular level, this year's Nobel Laureates Richard Axel (Howard Hughes Medical Institute,[..]
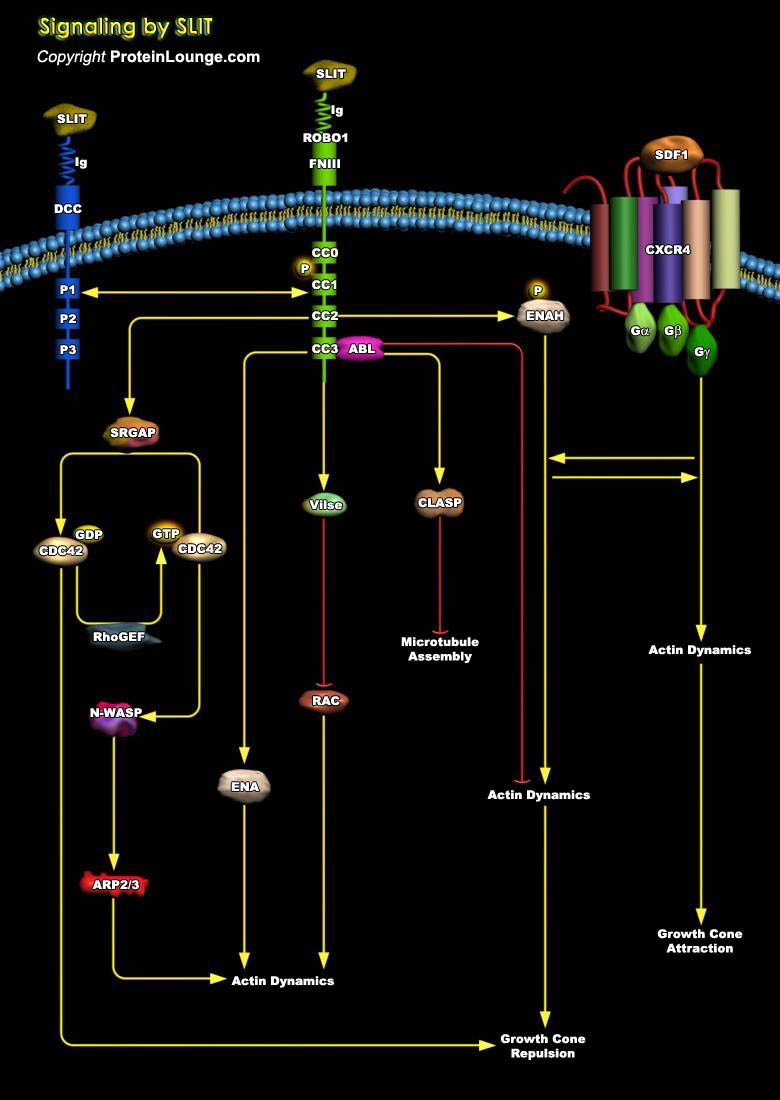
Neuronal growth cones in the developing nervous system are guided to their targets by attractive and repulsive guidance molecules, which include members of the Netrin, Semaphorin, Ephrin, and Slit protein families. Slit is a ~200 KD secretory protein originally shown to be expressed by neurons and glial cells. Slit was originally identified in Drosophila as an extracellular cue to guide axon pathfinding, to promote axon branching, and to control neuronal migration (Ref.1). Mammalian Slit consists of three members, Slit1, Slit2, and Slit3. All are expressed in the nervous system but Slit1 is specifically expressed in brain, whereas Slit2 and Slit3 are expressed in brain as well as kidney, lung, heart, spleen, and lymph nodes. A prototypical Slit protein contains an[..]
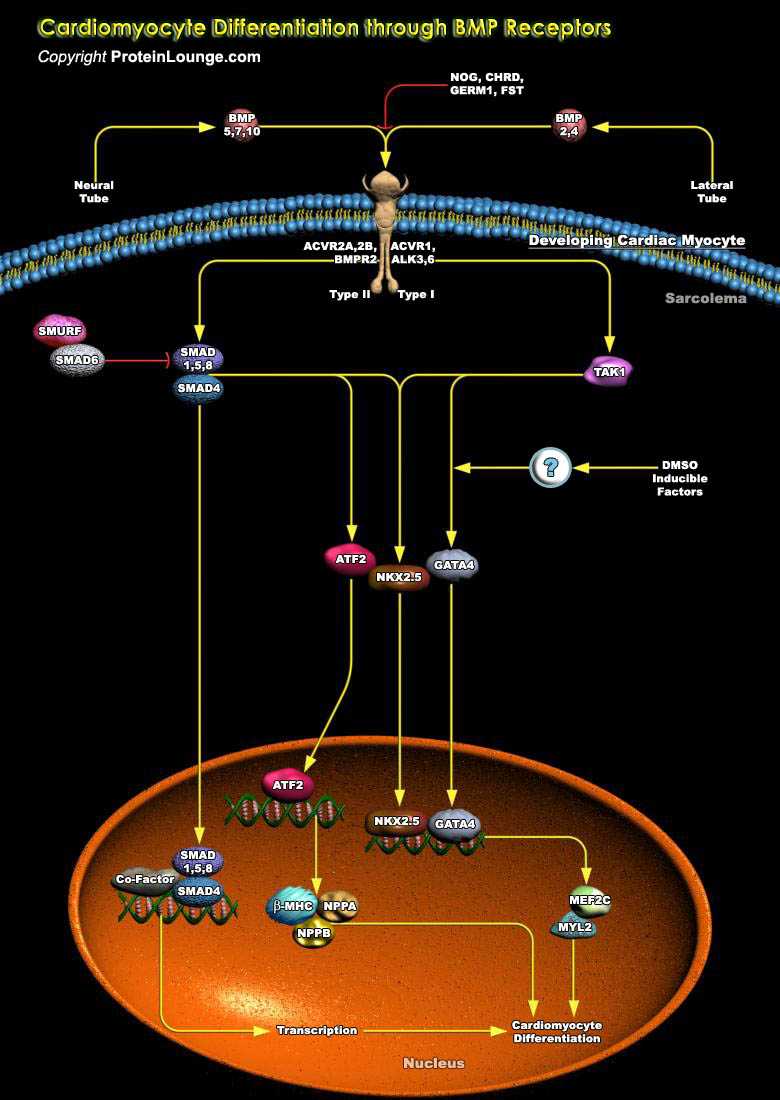
BMP (Bone Morphogenetic Protein) Receptors are essential, beyond the egg cylinder stage, for myocyte-dependent functions and signals in cardiac organogenesis. ALK3 (Activin Receptor-Like Kinase-3) is specifically required at mid-gestation for normal development of the trabeculae, compact myocardium, interventricular septum and endocardial cushion. The invariable defects in myocardium results from congenital deletion of ALK3 and this provide strong support for its assessment as a candidate gene in human congenital heart disease. BMPs like BMP2, BMP4 and BMP5, BMP7, BMP10, bind to Serine/threonine kinase receptors, Type-I (ALK3 and ALK6) and Type-II, BMPR2 (Bone Morphogenetic Protein Receptor Type-II), respectively, and form a heteromeric signaling complex acting in[..]
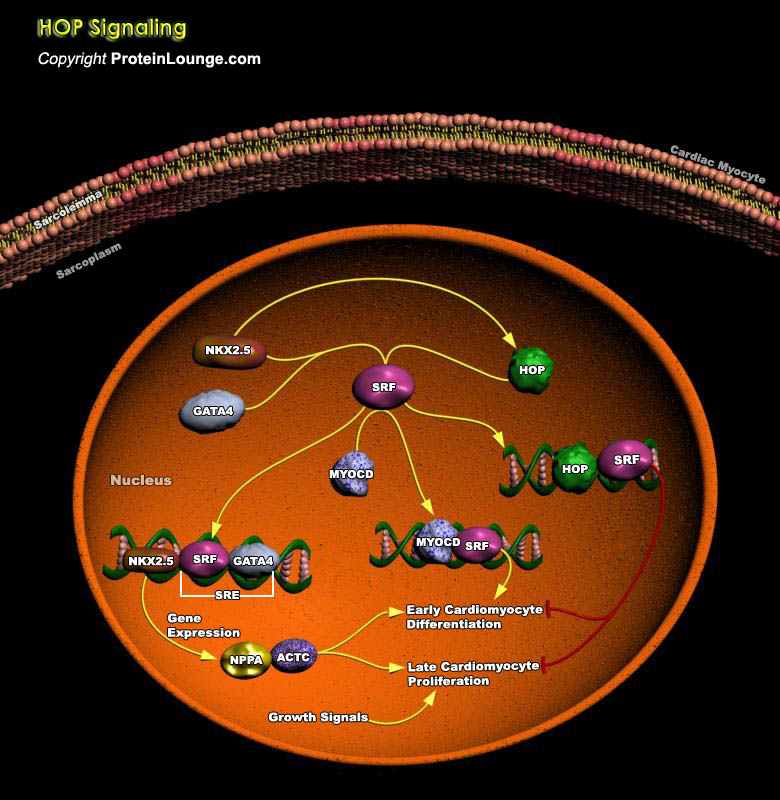
Cardiac myocyte proliferation and their differentiation early in development are dependent on the coordinate expression and action of SRF (Serum Response Factor), GATA4 (GATA Binding Protein-4) and the homeodomain factor NKX2.5 (NK2 Transcription Factor Related Locus-5). All three of these factors are expressed in developing cardiomyocytes and induce expression of cardiac genes. HOP (Homeodomain-Only Protein) physically interacts with SRF and inhibits activation of SRF-dependent transcription by inhibiting SRF binding to DNA. HOP gene encodes the smallest known homeodomain protein. HOP acts to modulate SRF-dependent transcription and cardiac-specific gene expression in the absence of intrinsic DNA binding capability. Although HOP is a homeodomain protein, it lacks a[..]
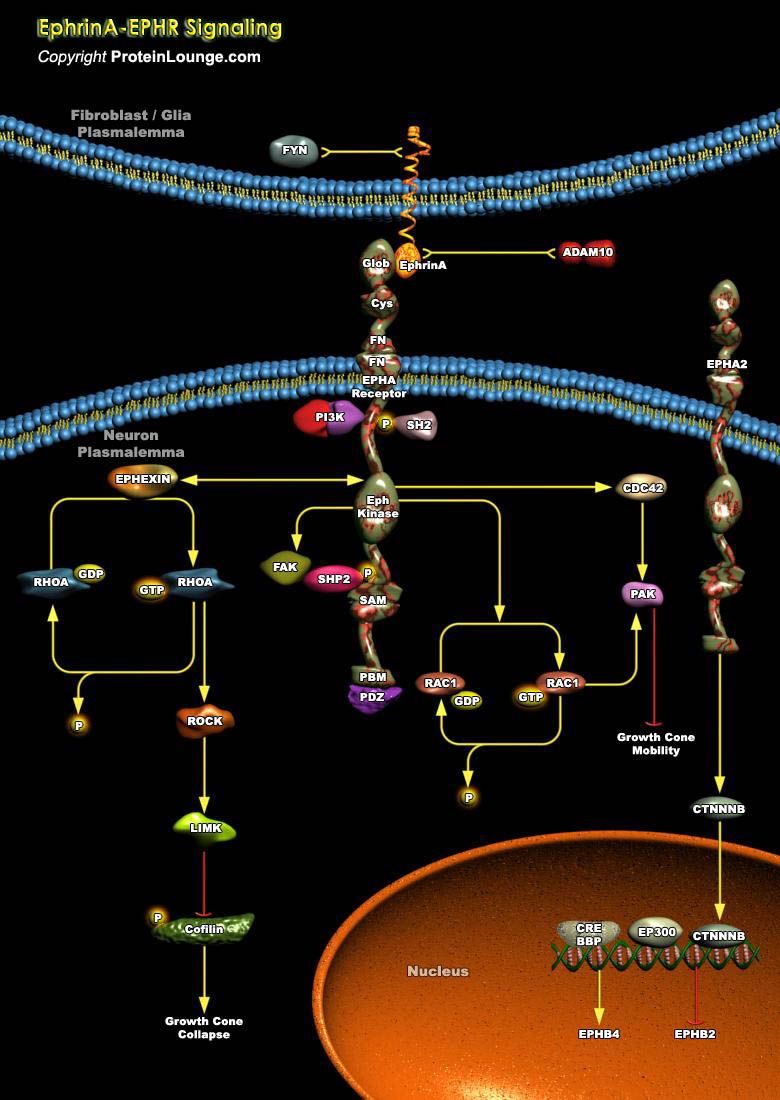
Neuronal growth cones in the developing nervous system are guided to their targets by attractive and repulsive guidance molecules, which include members of the netrin, semaphorin, ephrin, and Slit protein families. The Eph family forms the largest group of RTKs (Receptor Tyrosine Kinases) comprising 14 members in mammals that play critical roles in diverse biological processes during development as well as in the mature animal. They are activated by membrane-bound ligands called Ephrins, which are classified into two subclasses based on their modes of membrane anchorage. The EphrinA ligands are tethered to the plasma membrane by GPI (Glycosylphosphatidylinositol) anchor and prefer to bind to EphA Receptors. The EphrinB ligands (EphrinB1–B3), which possess a[..]

