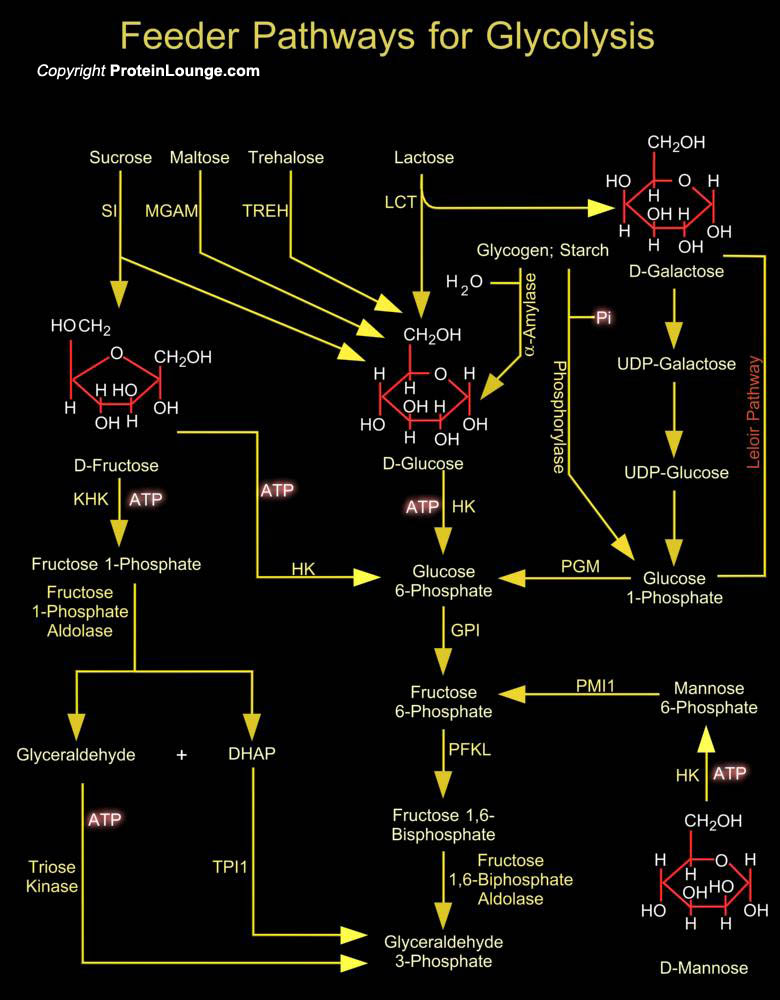
Many carbohydrates besides Glucose (or D-Glucose) meet their catabolic fate in Glycolysis, after being transformed into one of the Glycolytic intermediates. The most significant are the storage polysaccharides Glycogen and Starch; the disaccharides Maltose, Lactose, Trehalose and Sucrose; and the monosaccharides Fructose (or D-Fructose), Mannose (or D-Mannose) and Galactose (or D-Galactose) (Ref.1). Glycogen in animal tissues and in microorganisms; and Starch in plants, are mobilized for use within the same cell by a phosphorolytic reaction catalyzed by Phosphorylase (that is Glycogen Phosphorylase in animals and in microorganisms or Starch Phosphorylase in plants). These enzymes catalyze an attack by Pi (Inorganic Phosphate) on the (Alpha1-4) glycosidic linkage[..]
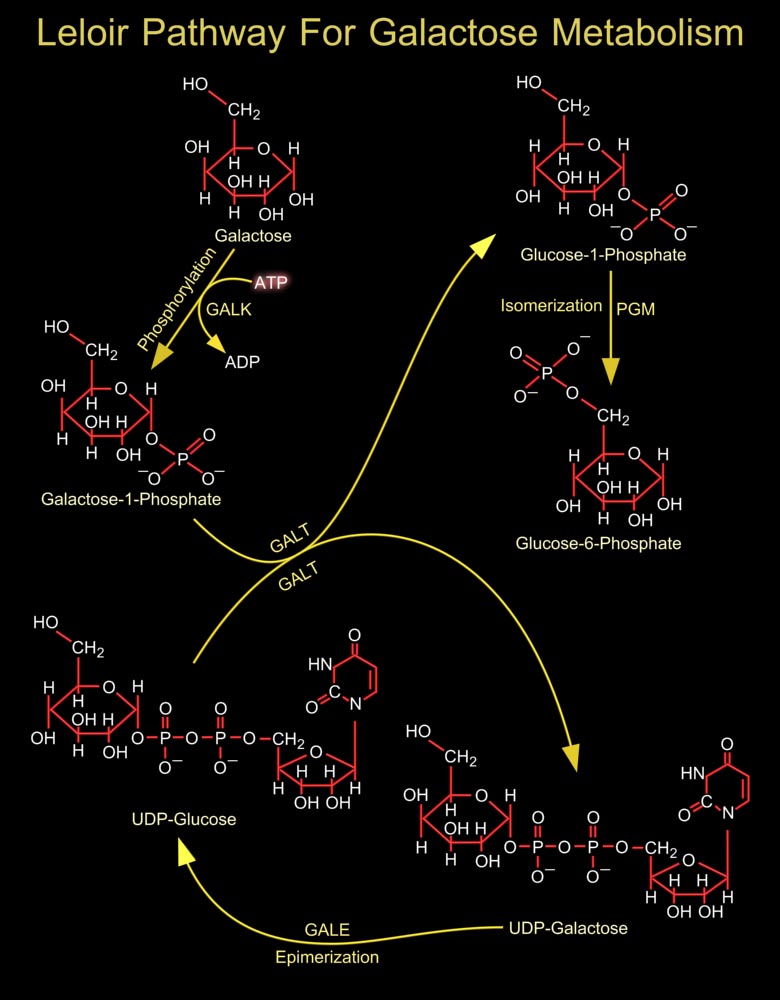
In most organisms, the conversion of Galactose to the more metabolically useful Glucose-1-Phosphate is accomplished by the action of four enzymes that constitute the Leloir pathway. In the first step of this pathway, Beta-D-Galactose is epimerized to Alpha-D-Galactose by GALM (Galactose Mutarotase/Aldose 1-Epimerase) (Ref.1). The active site of GALM is positioned in a rather open cleft with the hydroxyl groups of Galactose lying within hydrogen bonding distance to a number of side chains, the reaction catalyzed by human GALM proceeds via Glu-307 (Glutamic acid-307) and His-176 (Histidine-176). Till now no diseases have been attributed to mutations in human GALM. The next step involves the ATP-dependent phosphorylation of Alpha-D-Galactose by GalK (Galactokinase) to[..]
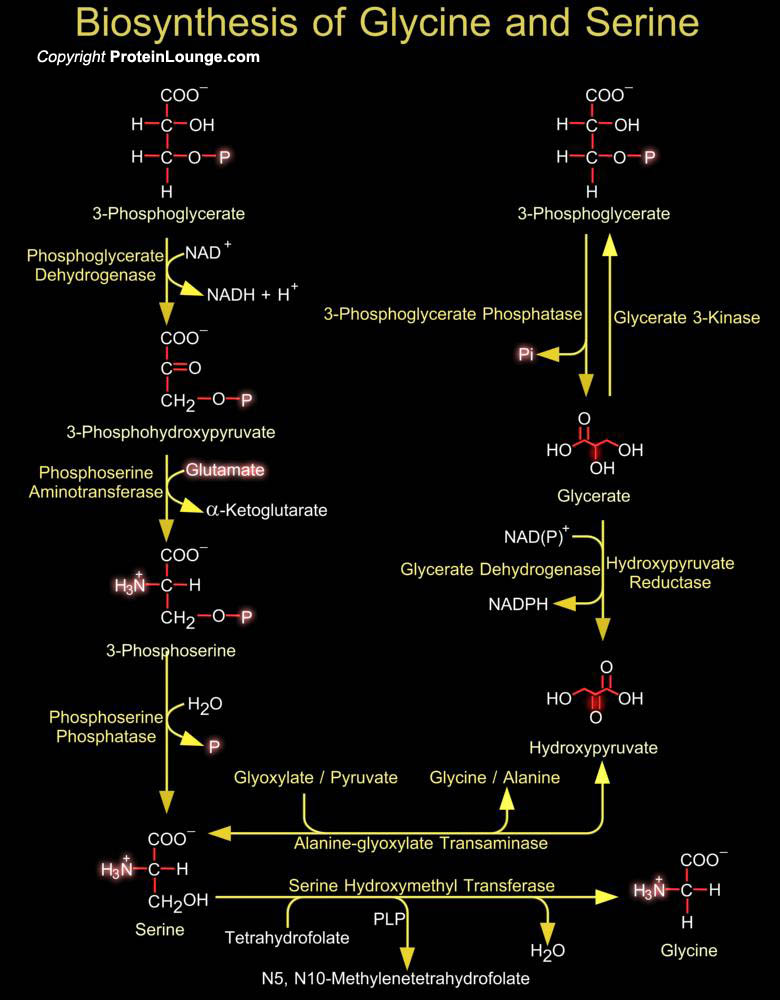
Intermediates in energy production pathways such as glycolysis and the Krebs cycle are commonly the starting point for the biosynthesis of amino acids. The biosynthesis of serine and glycine constitute a major metabolic pathway that plays a central role in the formation of other amino acids, nucleic acids and phospholipids. Serine an ɑ-amino acid used in the biosynthesis of proteins can be synthesized in the human body under normal physiological circumstances, making it a nonessential amino acid. It contains an α-amino group a carboxyl group, and a side chain consisting of a hydroxymethyl group, classifying it as a polar amino acid. Glycine is also an amino acid that with a single hydrogen atom as its side chain. It is one of the proteinogenic amino acids and[..]
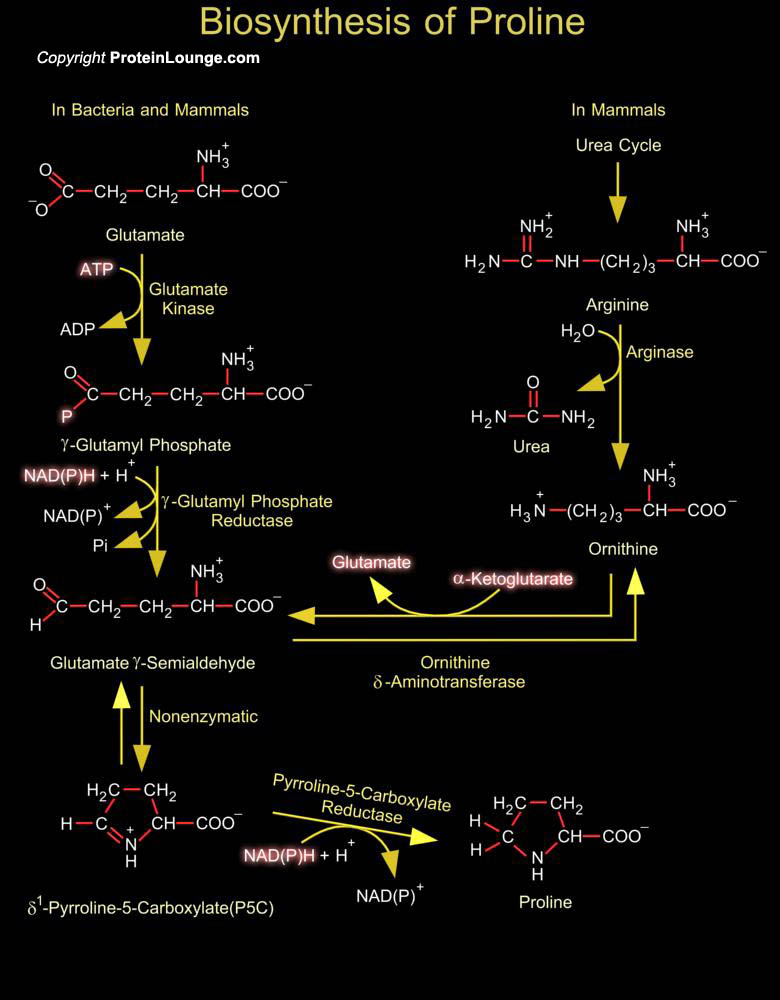
Proline is unique in having a cyclic structure with its side chain connected to the amino group to create a secondary amine. As a consequence of its cyclic structure, Proline constrains the structure of proteins where it occurs, disrupting Alpha-helices. Isomerization between the cis and trans forms of Proline in proteins is isomerized by peptidyl-prolyl isomerases like the cyclophilins that contribute to protein folding and are components of signal transduction pathways (Ref.1). Proline is derived from glutamate. Its biosynthesis begins with the ATP-driven phosphorylation and reduction of the carboxyl side chain of glutamate. The Gamma-carboxylate group of glutamate is activated by phosphorylation with ATP to from a Gamma-Glutamyl Phosphate intermediate. The[..]
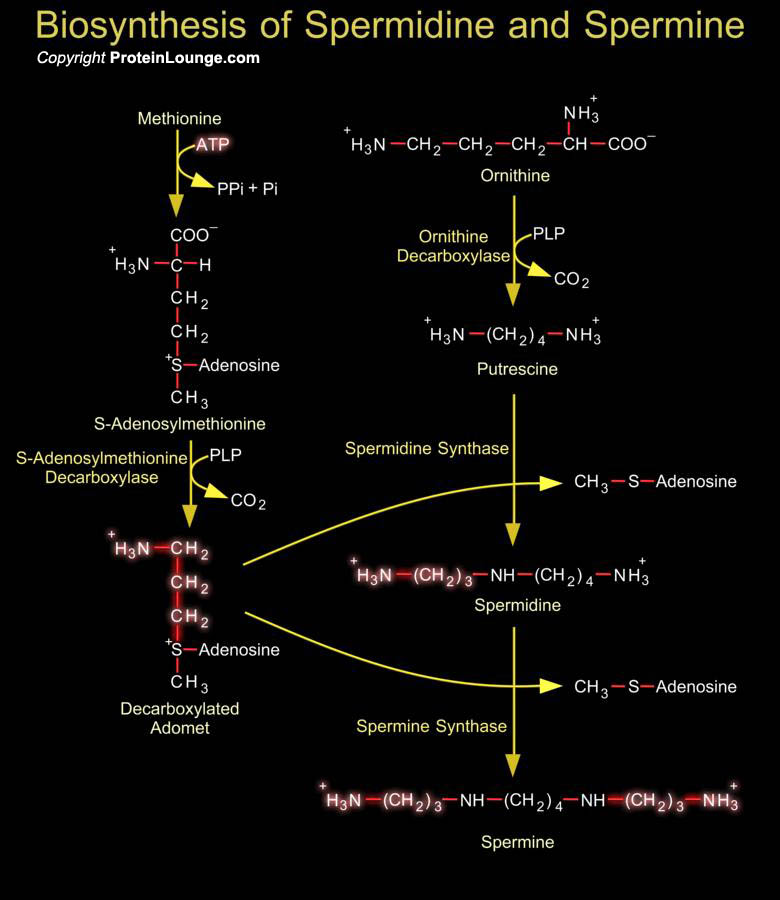
The polyamines Spermidine and Spermine are naturally occurring ubiquitous polycations involved in the regulation of cell growth, differentiation, apoptosis and progression through the cell cycle. (Ref.1). Cellular polyamine levels are regulated by multiple pathways such as synthesis from amino acid precursors, cellular uptake mechanisms that salvage polyamines from diet and intestinal microorganisms, as well as stepwise degradation and efflux (Ref.2). Putrescine and S-Adenosylmethionine are the obligate precursors for Spermidine and Spermine. In mammals Putrescine is formed by decarboxylation of Ornithine by the action of Ornithine Decarboxylase. Ornithine, one of the starting materials for polyamine biosynthesis, is derived from the amino acid Arginine as part of[..]
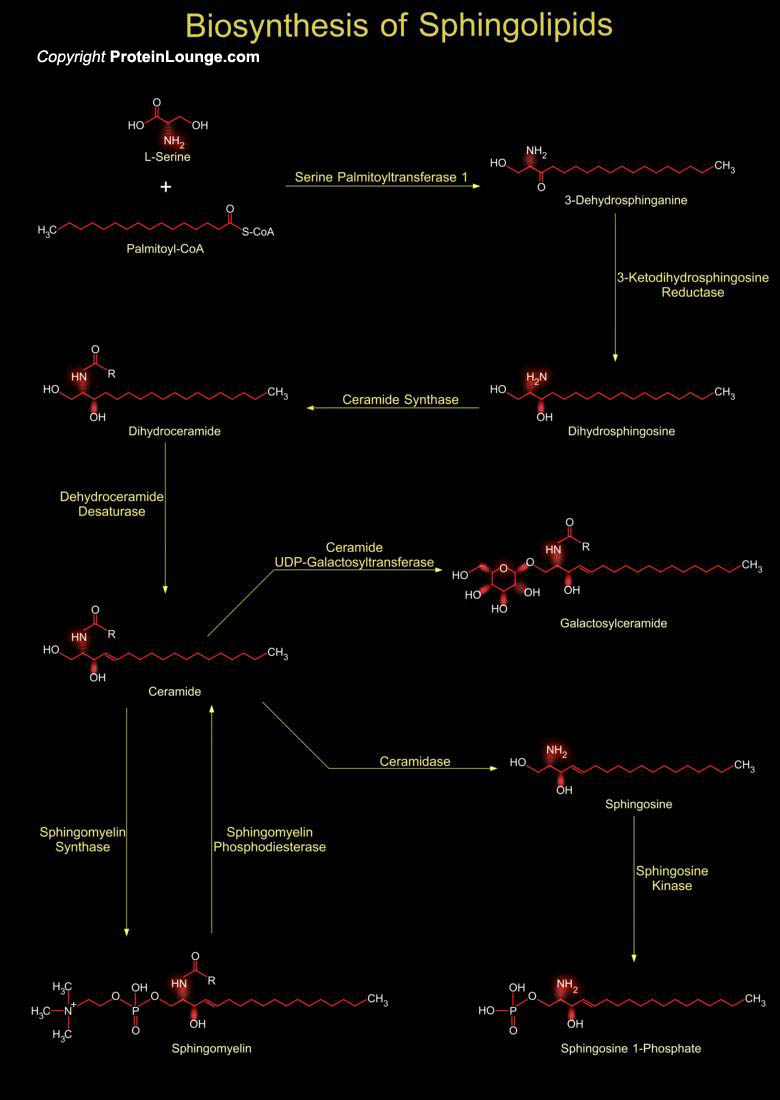
Sphingolipids structural components of the cell membrane play a key role in the regulation of several cellular processes. Cell signaling related to sphingolipids induces apoptosis, cell cycle arrest and cell growth. There are multiple bioactive sphingolipids metabolites which include ceramide, sphingosine-1-phosphate, sphingosine, and sphingomyelin which may act as secondary messengers in cellular signal transduction pathways. Sphingolipids are abundant components of the myelin sheath which protects and insulates cells of the central nervous system. Sphingolipids are also found in plant cells and yeast and are derivatives of the base sphingosine (phytosphingosine in plants). Sphingolipids are a class of lipids with two non-polar tails (sphingosine and fatty[..]
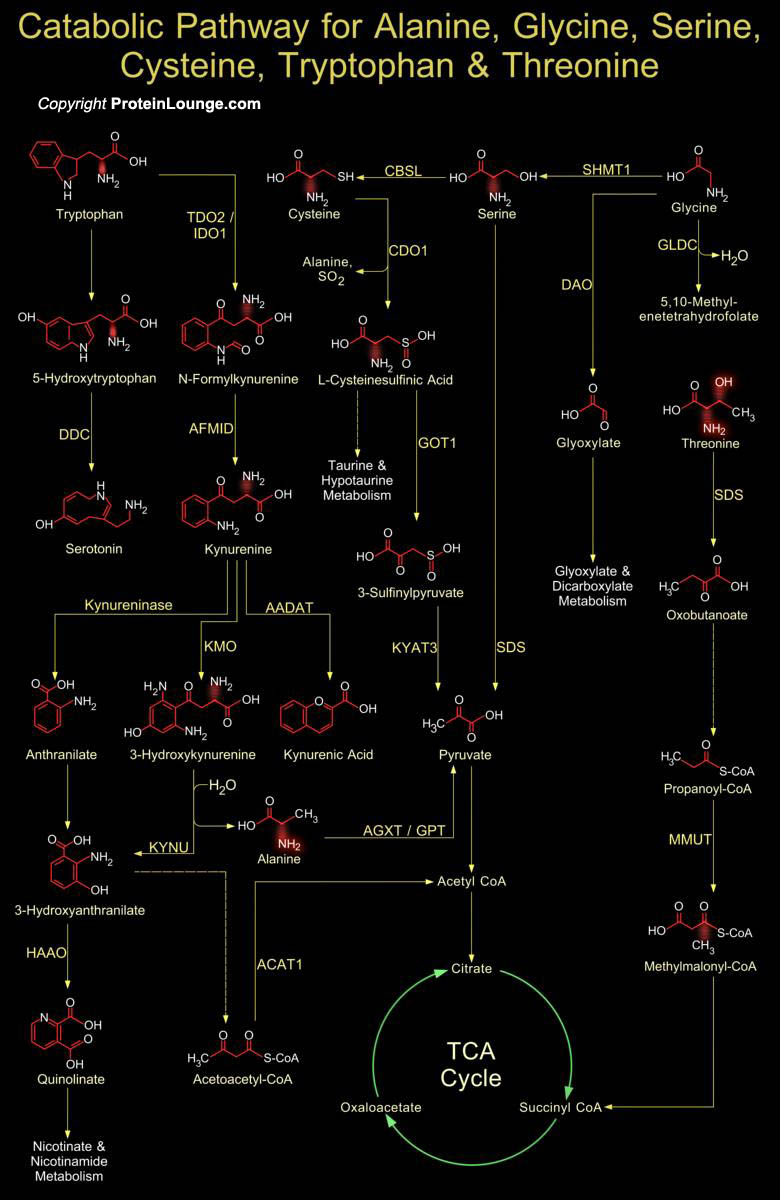
Five amino acids, Alanine, Cysteine, Glycine, Serine and Threonine are broken down to yield Pyruvate. Tryptophan is included in this group since one of its breakdown products is Alanine, which is transaminated to Pyruvate. Alanine is an important in inter tissue nitrogen transporter and is an important part of the Glucose-Alanine cycle. The alanine catabolic pathway involves a simple aminotransferase reaction that directly produces pyruvate. The transamination is carried out by GPT (Glutamic--Pyruvic Transaminase). Post pyruvate production, based on metabolic requirements of the cell, pyruvate can either be oxidized by the PDH complex to acetyl-CoA and diverted into the TCA cycle or it can be diverted into the gluconeogenic pathway to release glucose[..]
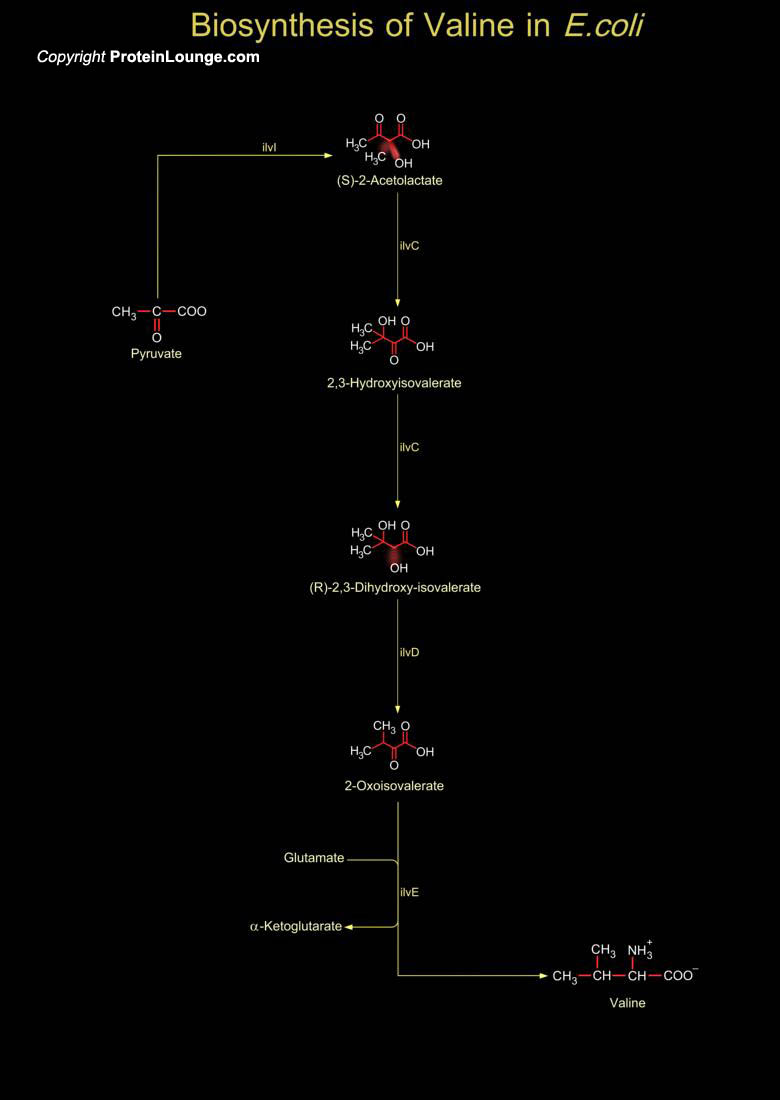
Valine is a hydrophobic amino acid, it often forms the helical structures within interior proteins. Valine is an essential amino acids and must be obtained in the diet in human but not synthesized in human. It is generally biosynthesized by plants, algae, fungi, bacteria and archaea. Important sources of valine include soy flour, cottage cheese, fish, meats, and vegetables. Valine is incorporated into proteins and enzymes at the molar rate of 6.9 percent when compared to the other amino acids. In Escherichia coli (E.coli), Valine is biosynthesized from two Pyruvate molecules where two molecules of Pyruvate are converted to Alpha-Acetolactate by the enzyme ilvI (acetolactate synthase/acetohydroxybutanoate synthase, catalytic subunit) yielding a molecule of CO2.[..]
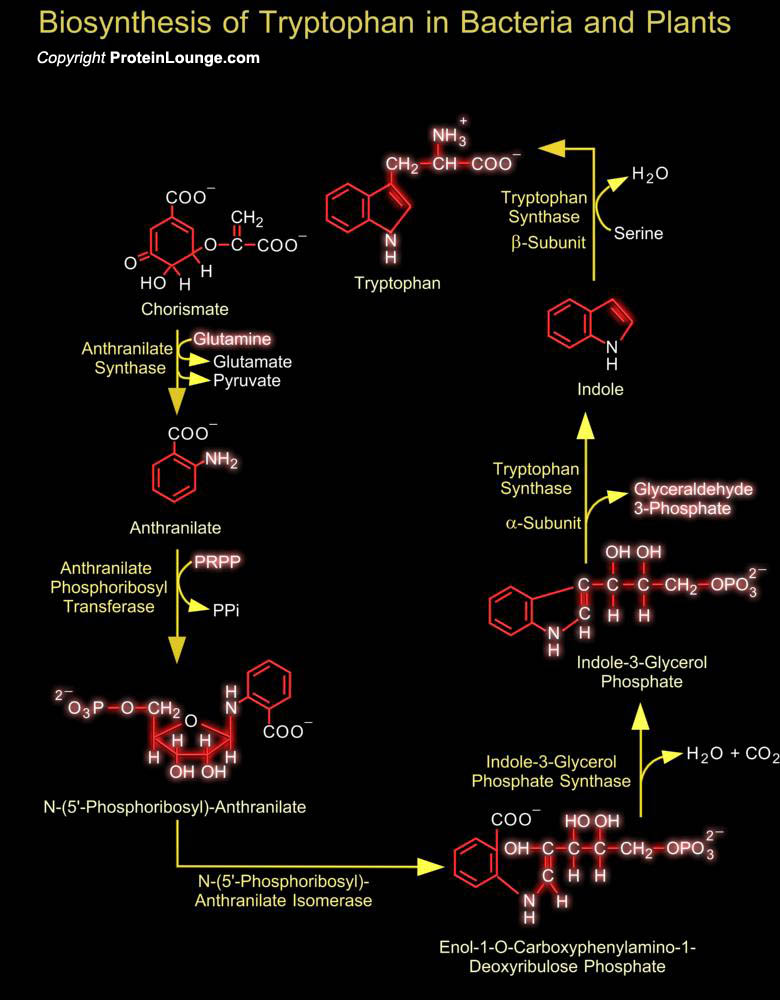
Tryptophan is an amino acid essential to survival. Amino acids serve as building blocks for proteins, as well as serving as starting points for the synthesis of vitamins and many other crucial cellular molecules. While most plants and microorganisms can produce all the amino acids they need, tryptophan is one of eight amino acids that cannot be produced by animals. Humans and animals do not themselves have the biosynthetic machinery to synthesize tryptophan but rely on dietary intake from bacteria and plants that do produce it. Tryptophan is the least abundant of the essential amino acids. However, it is also one of the most crucial, as it is involved not only in protein synthesis, but also in the formulation of niacin and the neurotransmitter serotonin. Serotonin is[..]
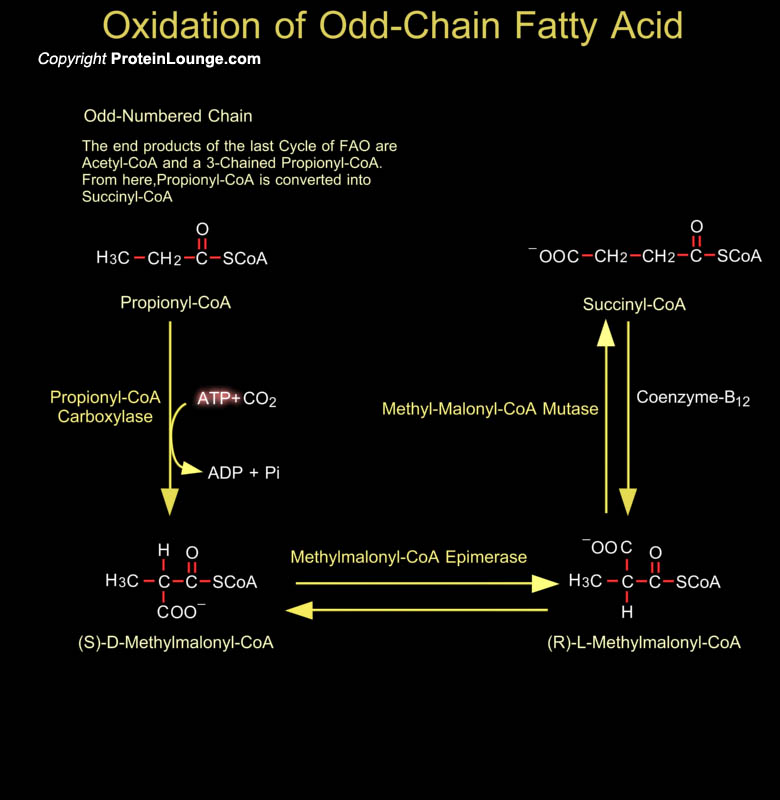
Most fatty acids have even number of carbon atoms and are therefore completely converted to Acetyl-CoA. Some plants and marine organisms, however, synthesize fatty acids with an odd number of carbon atoms. The final round of Beta-Oxidation of these fatty acids forms Propionyl-CoA, which is converted to Succinyl-CoA for entry into the Citric Acid Cycle. Propionyl-CoA can be derived from breakdown of certain amino acids (Isoleucine, Valine and Methionine), but in mammalian cells these pathways exist in mitochondria. Bacteria in the ruminant animal digestive system provide a source of propionate which eventually gets incorporated as odd-numbered fatty acids in milk fats, and this is the major source for human metabolism. The conversion of[..]
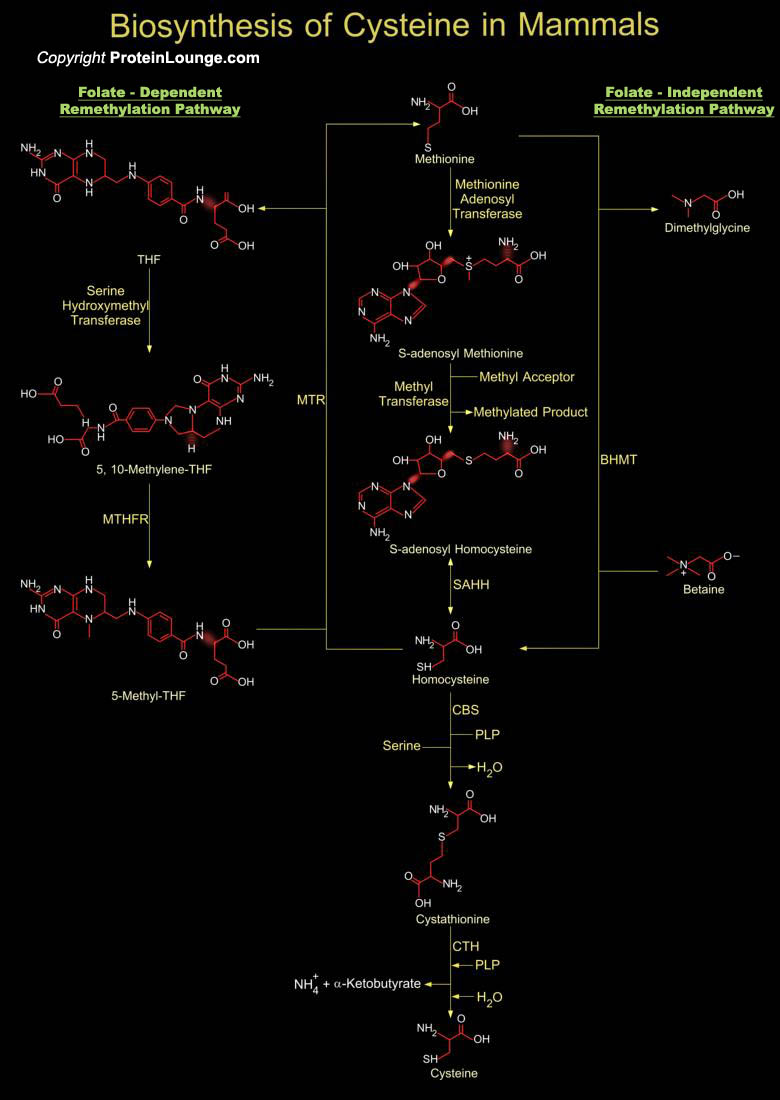
Cysteine, a sulfur-containing amino acid, is indispensable for the survival of virtually all living organisms, from bacteria to higher eukaryotes. This amino acid is implicated in several processes, including the stability, structure, regulation of catalytic activity, and post-translational modification of various proteins. Due to the ability of its thiol group to undergo redox reactions, Cysteine forms the basic building block of all thiol antioxidants, acting as a direct antioxidant and also as a precursor for the biosynthesis of glutathione, trypanothione, or ovothiol. In addition, cysteine is also essential for the synthesis of biomolecules, including coenzyme A, hypotaurine, taurine, and ubiquitous iron-sulphur (Fe-S) clusters, which are involved in electron[..]
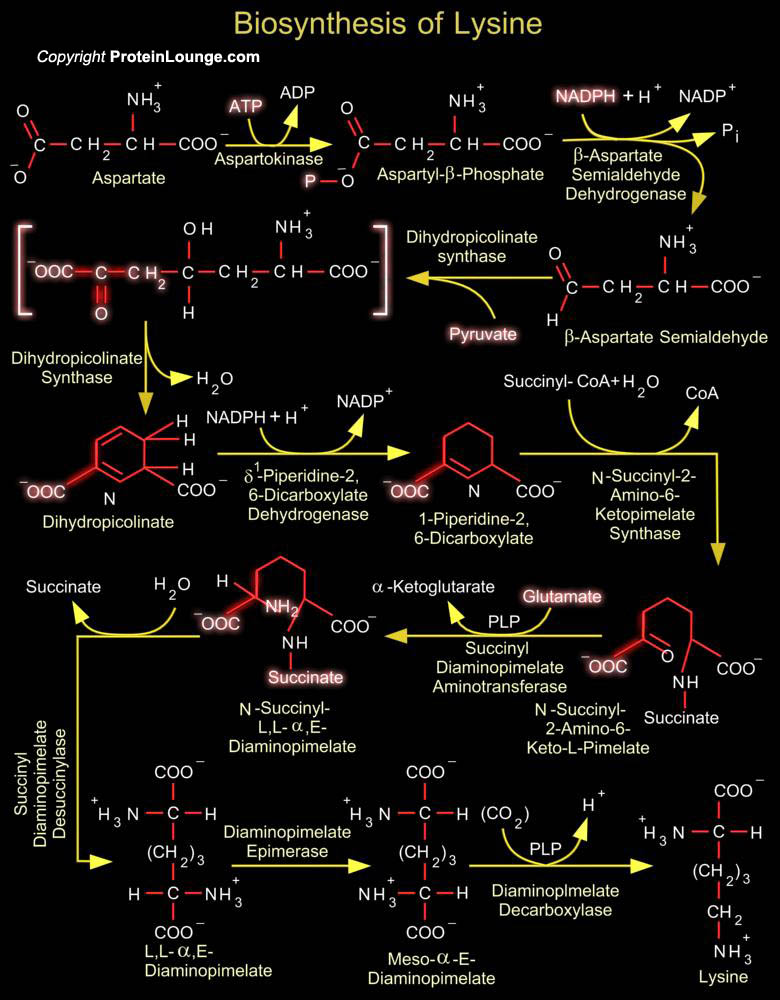
The amino acid L-lysine is synthesized by plants and microorganisms by two different pathways, one proceeding via Diaminopimelate and the other via Alpha-Aminoadipate. Humans, however, cannot synthesise the compound and so it is an essential part of their diet. The Diaminopimelate pathway operates in bacteria, lower fungi and green plants. This pathway is the source of the lysine and Diaminopimelate incorporated into bacterial cell wall peptidoglycan and therefore there has been extensive investigation of its enzymes with a view to the development of new antibacterial agents. In Euglenoids and higher fungi lysine biosynthesis occurs via the Homocitrate-Aminoadipate pathway. An intermediate on this pathway (Alpha-Aminoadipic acid) is necessary for Beta-lactam antibiotic[..]

