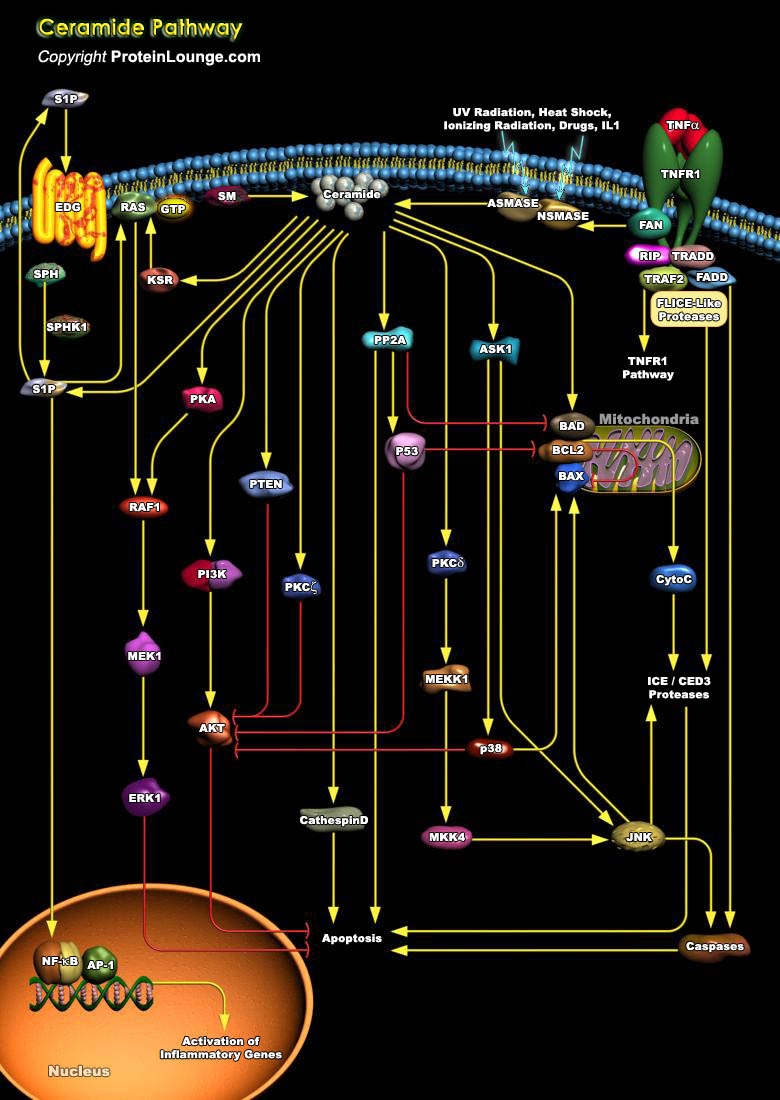
The SM (Sphingomyelin) pathway is an evolutionarily conserved stress response system linking diverse environmental stresses (Ultraviolet, Heat Shock, Oxidative Stress, and Ionizing Radiation) to cellular effector pathways. Ceramide is the second messenger in this system and can be generated either by hydrolysis of SM through SM-specific PLC (Phospholipase-C) termed SMases (Sphingomyelinases) or by de novo synthesis through the enzyme Ceramide Synthase . There are two classes of SMase, acidic (A-Smase) and neutral (N-Smase). Stress stimuli such as, TNF-Alpha (Tumor Necrosis Factor-Alpha), lipopolysaccharide, and some chemotherapy drugs such as doxorubicin also mediate apoptosis by generation of the lipid second messenger, Ceramide. A specific interaction between the FAN[..]

The ability of multicellular organisms to maintain cellular homeostasis is critically dependent on a balance between cell survival and cell death (apoptosis). The responsiveness of individual cells to death signals varies greatly depending on the presence of continuous survival cues from the extracellular environment. The perturbation of normal cell survival mechanisms, leading to an increase in cell survival or cell death plays an important role in the development of a number of disease states, including cancer. BAD (BCL2 Associated Death Promoter) is a pro-apoptotic critical regulatory component of the intrinsic cell death machinery that exerts its death-promoting effect upon heterodimerization with the antiapoptotic proteins of the BCL2 family defined by conserved[..]
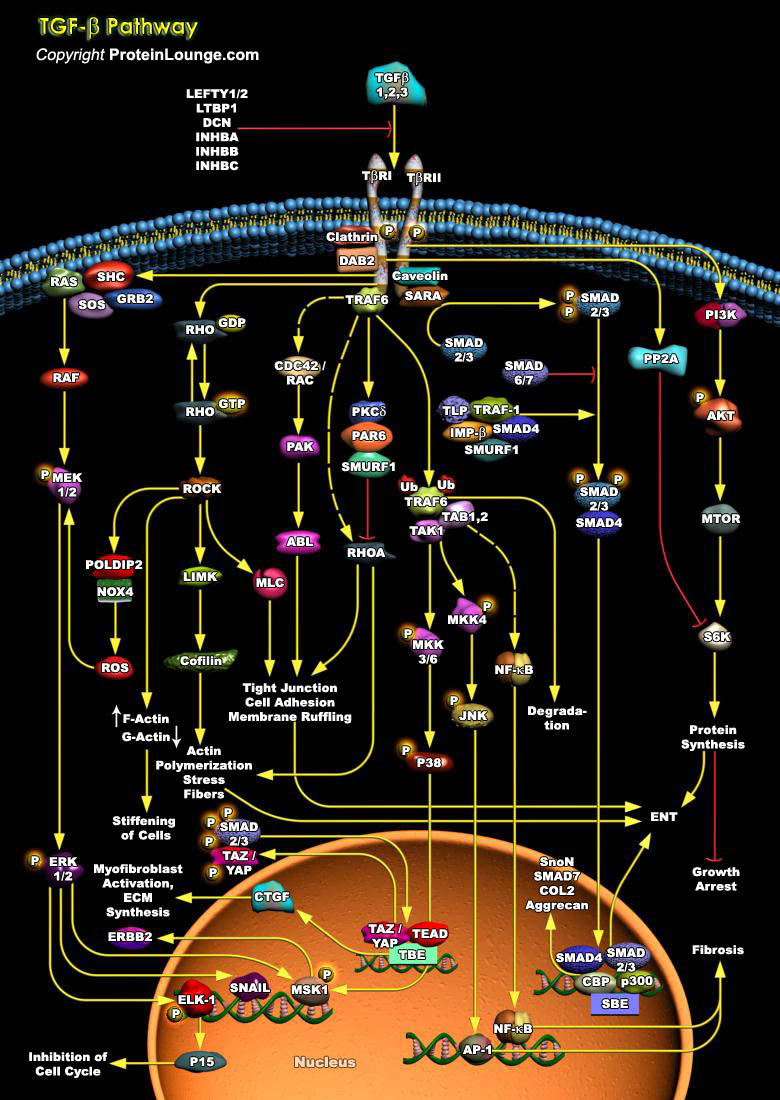
TGFB (Transforming growth factor-beta) is a multifunctional cytokine that regulates a wide variety of cellular functions, including cell growth, cellular differentiation, apoptosis, and wound healing. TGF-b signals are transmitted through two transmembrane serine/threonine kinase receptors TGFBR1 and TGFBR2 [Ref.1]. Initiation of the TGFB signaling cascade occurs upon ligand binding to TGFB receptor TGFBR2 and subsequent TGFBR1– TGFBR2 heterotetrameric complex formation. TGFBR2 is a constitutively active receptor kinase and phosphorylates Ser/Thr residues in the cytoplasmic GS domain of TGFBR1, which turns on the kinase activity of TGFBR1. Upon Activation, TGFBR1 transmits its signal to the various intracellular SMAD-dependent and SMAD independent signaling[..]
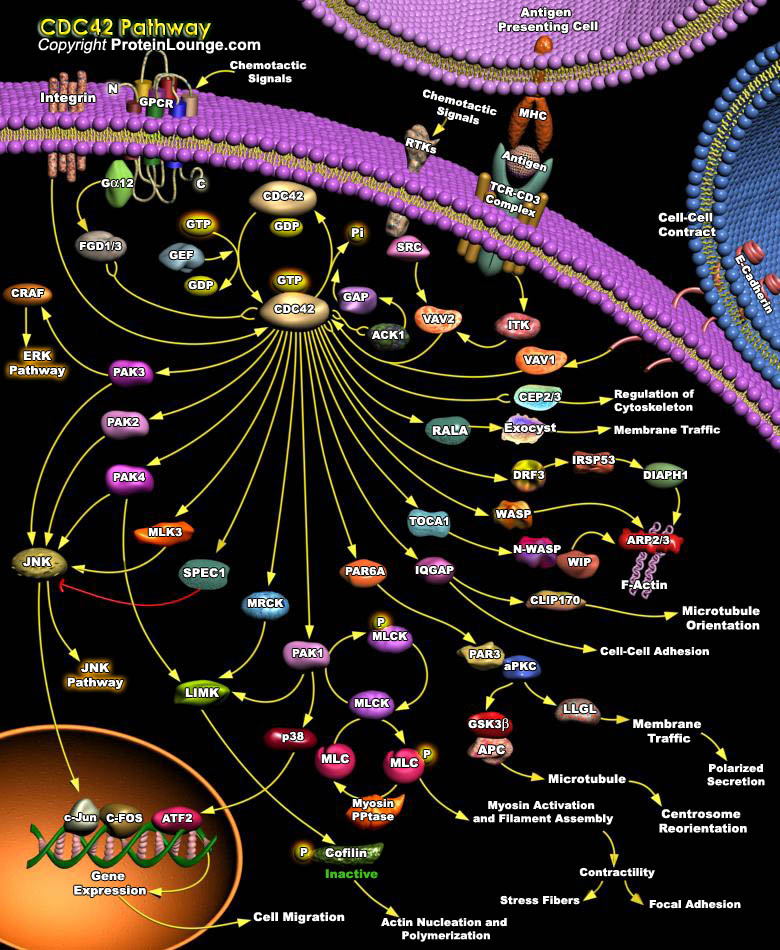
Dynamic regulation of the actin cytoskeleton underpins a multitude of cellular processes, from cell movement and polarization1,2 to cell division.3 The actin cytoskeleton and the proteins involved in its regulation are also fundamentally linked to endocytosis and membrane trafficking.4,5 Members of the Rho family of small GTPases have emerged as important overseers of the actin cytoskeleton and a number of Rho family members and their downstream effector proteins have been linked to specific actin pathways. Among the Rho family actin regulators is Cdc42 is a small GTPase responsible for a large number of eukaryotic cell signaling pathways(Ref.1).CDC42 acts downstream of cell surface receptors to regulate the formation of different F-actin-containing structures. It[..]
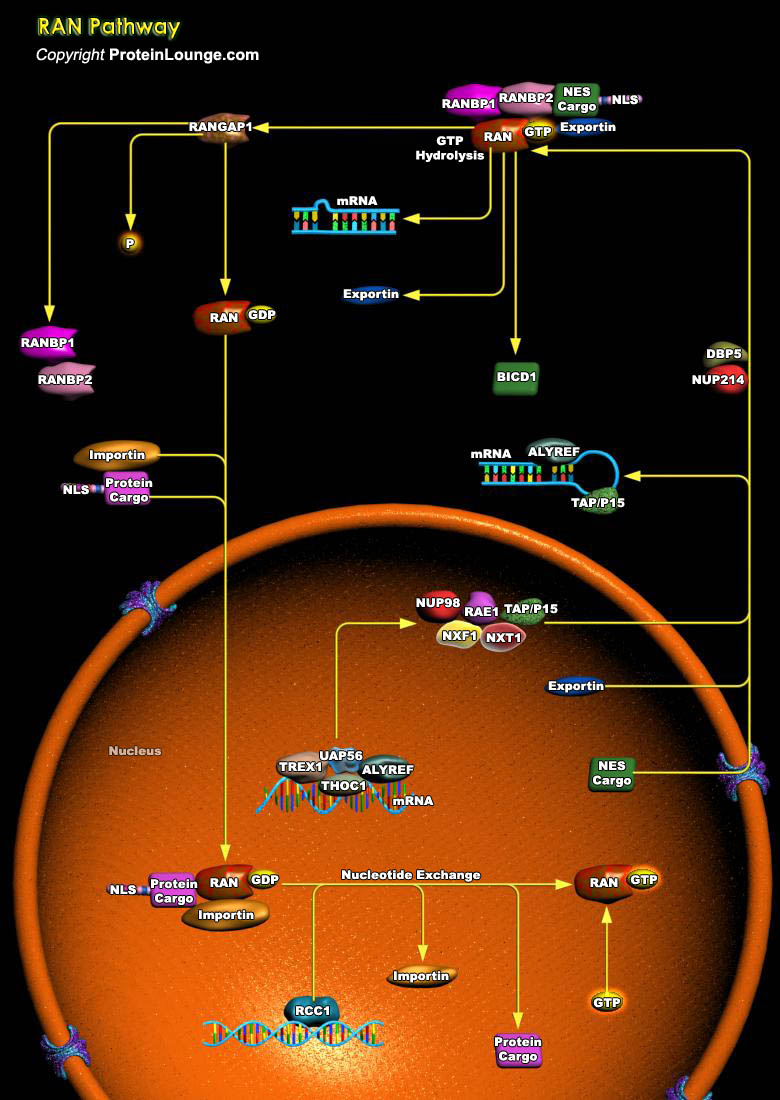
Ran is a member of the Ras family of small GTPases. The Ran subgroup is represented by its lone member, Ran, that is distinguished from Ras GTPases by its lipid modification and atypical subcellular localization. Unlike most other Ras-related proteins, Ran is not modified to bind to cell membranes. Instead, Ran protein is localized throughout the cell, where it is concentrated primarily in the nucleus (Ref.1). Ran is regulated by a cytosolic RanGAP1 (Ran GTPase–Activating Protein-1) and by a RanGEF (Chromatin-Bound Guanine Nucleotide Exchange Factor). The distribution of RanGTP provides important spatial information that directs cellular activities during different parts of the cell cycle. During interphase, the localization of RanGEF and RanGAP1 predicts that[..]
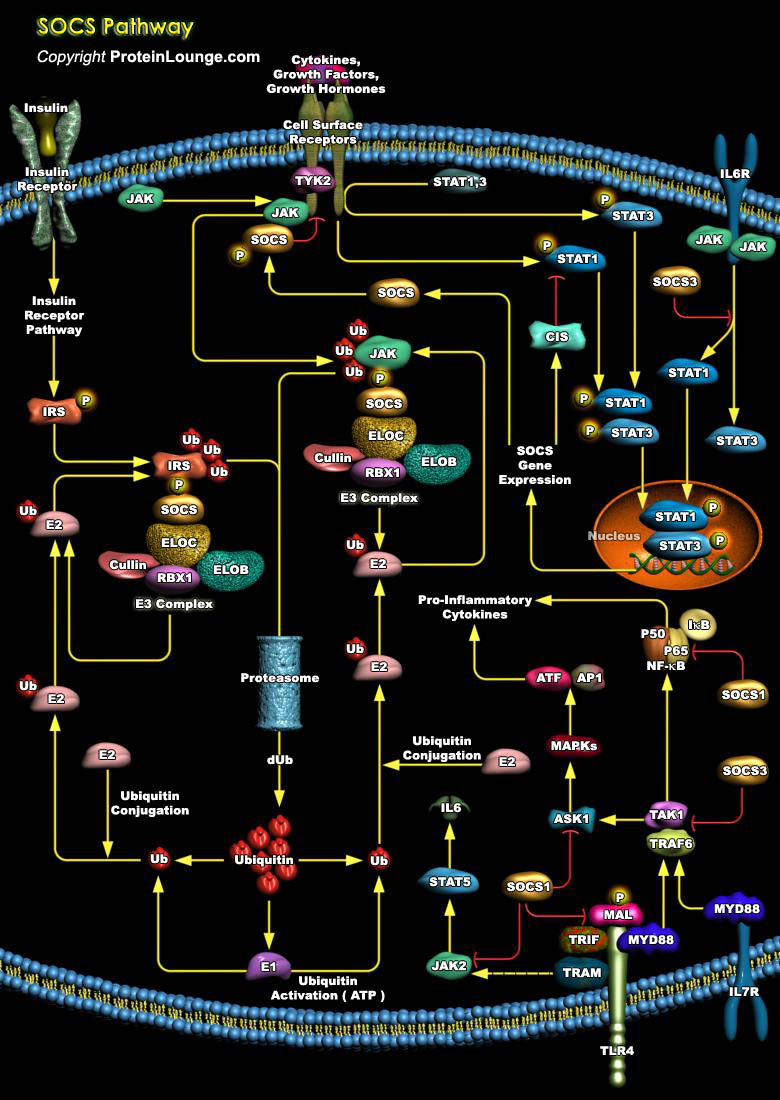
Our immune system is largely controlled by the action of pleiotropic cytokines and growth factors, small secreted proteins, which bind to receptors on the surface of immune cells to initiate an appropriate physiological response. The cellular response to cytokines and growth factors is predominantly executed by intracellular proteins known as the Janus kinases (JAKs) and the signal transducers and activators of transcriptions (STATs). These kinases activated upon ligand binding causes the phosphorylation of tyrosine residues within the receptor intracellular region and phosphorylation of other signalling proteins recruited to the receptor complex like signal transducers and activators of transcriptions (STATs). Once phosphorylated, STATs translocate to the[..]
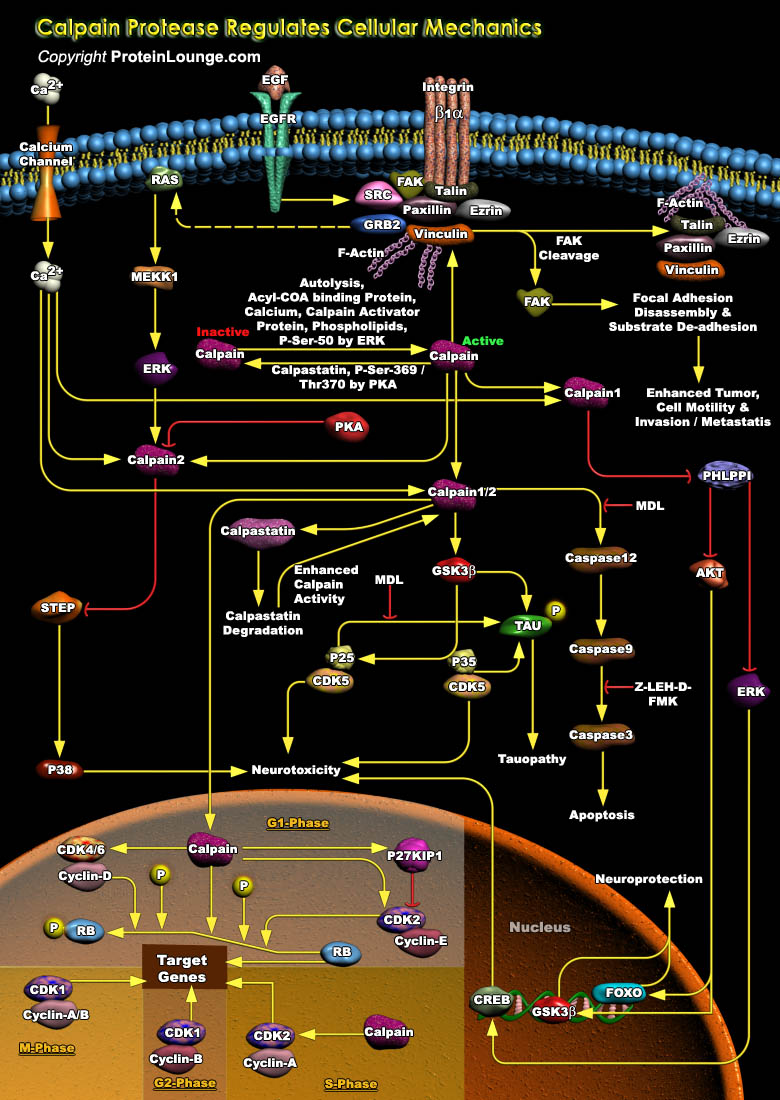
Calpain is an evolutionary old family of soluble, neutral, calcium-dependent proteases, which have the unique property of using protein cleavage to modify the activity/function of their substrate proteins. There are two major calpain isoforms in the brain, calpain-1 and calpain-2. Calpain-1 and Calpain-2 exhibit opposite functions in both synaptic plasticity and neurodegeneration. Calpain-1 activation is required for the induction of long-term potentiation (LTP) and is generally neuroprotective, while calpain-2 activation limits the extent of potentiation and is neurodegenerative. Calpains release the link between the integrin-dependent FA complex and the actin cytoskeleton by proteolysis of talin, which allows proper cell migration. The activity of Calpains is tightly[..]
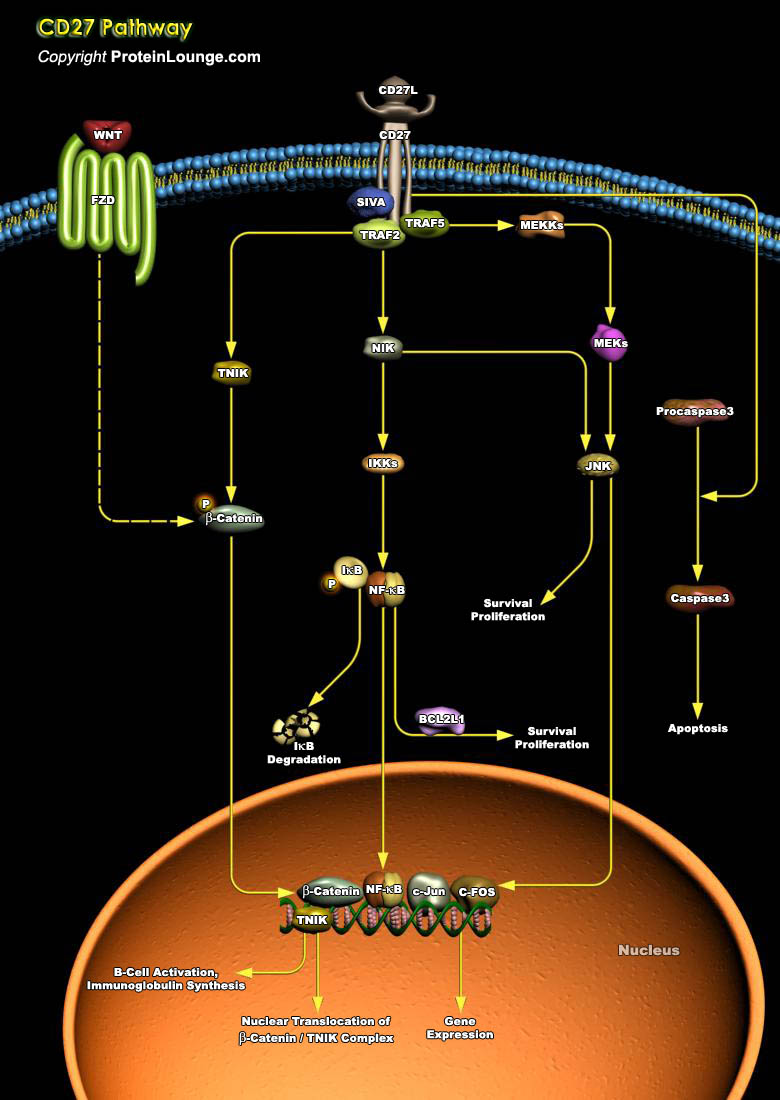
Members of the TNFR (Tumor Necrosis Factor Receptor) superfamily are important for cell growth and survival. CD27 is a member of the TNFR superfamily, which includes TNFR types I and II, NGFR (Nerve Growth Factor Receptor), CD30 (associated with Hodgkin lymphoma), Fas/Apo1 (CD95), CD40, 4–1BB, and OX40. These receptors are known to play a very important role in cell growth and differentiation, as well as apoptosis or programmed cell death. In addition to providing costimulatory signals for cell proliferation, ligation of both TNFR1 and Fas can result in programmed cell death or apoptosis. The underlying mechanism requires an intact 80-aa stretch present in the cytoplasmic tails of both TNFR1 and Fas, termed the DD (Death Domain) (Ref.1). CD27 is a disulfide-linked[..]
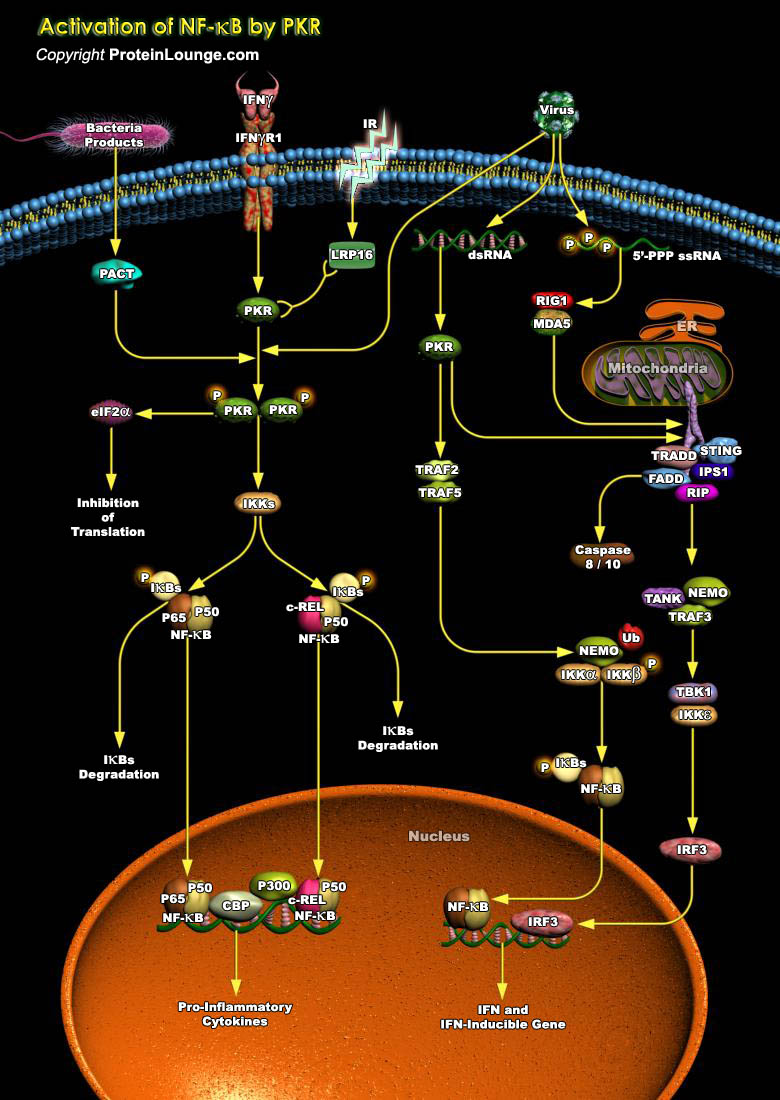
PKR (Protein Kinase-R) is a 68-kDa serine–threonine kinase that appears to play a primary role in mediating the antiviral activities of infected cells. PKR mediates apoptosis induced by many different stimuli, such as LPS (Lipopolysaccharides), TNF-Alpha (Tumour Necrosis Factor-Alpha), viral infection, or serum starvation. Viral infection leads to the increased expression and secretion of the cytokine IFN-Gamma (Interferon- Gamma) from host cells. IFN-gamma induces the dsRNA (Double-Stranded RNA)-dependent PKR. dsRNA, produced during viral replication, is an active component of a viral infection that stimulates antiviral responses in infected cells (Ref.1). The PKR protein is enzymatically inactive unless it is activated by binding to dsRNA. PKR activates as a kinase[..]
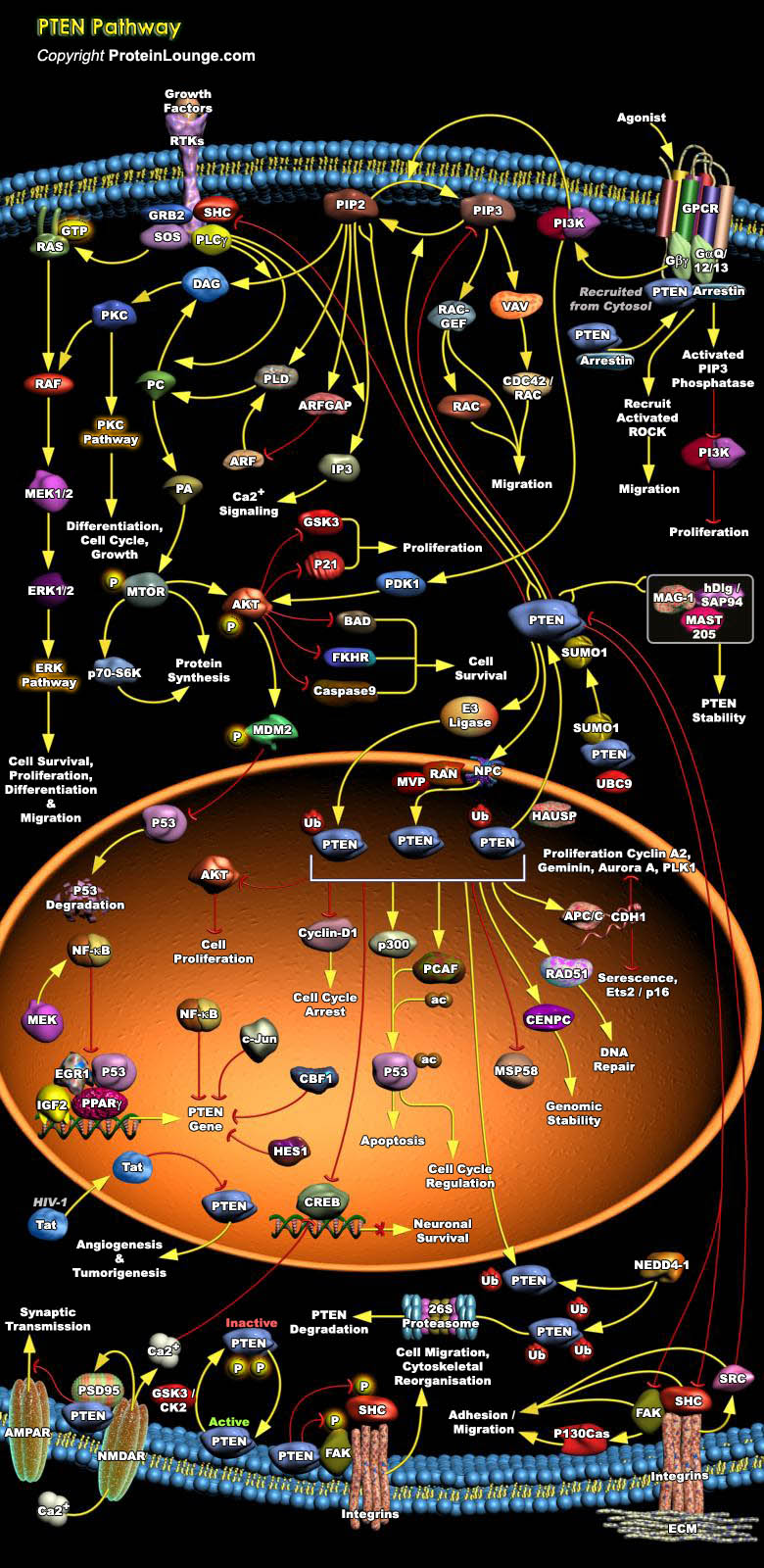
Phosphatase and tensin homolog deleted from chromosome 10 (PTEN) is a tumor suppressor protein, that is linked to AKT signaling, critically controlling proliferation, survival, migration, energy metabolism and cellular architecture in hematopoietic cells. PTEN is a dual-specificity protein phosphatase and an inositol phospholipid phosphatase that dephosphorylates phosphatidylinositol 3,4,5-trisphosphate (PIP3), thus producing phosphatidylinositol 4,5-bisphosphate (PIP2) and thereby negatively regulating oncogenic and nononcogenic PI3K/AKT signaling. PTEN may have additional phosphatase-independent activities, as well as other functions in the nucleus (Ref.1 and 2). PTEN's tumor-suppressing function largely relies on the protein's phosphatase activity and subsequent[..]
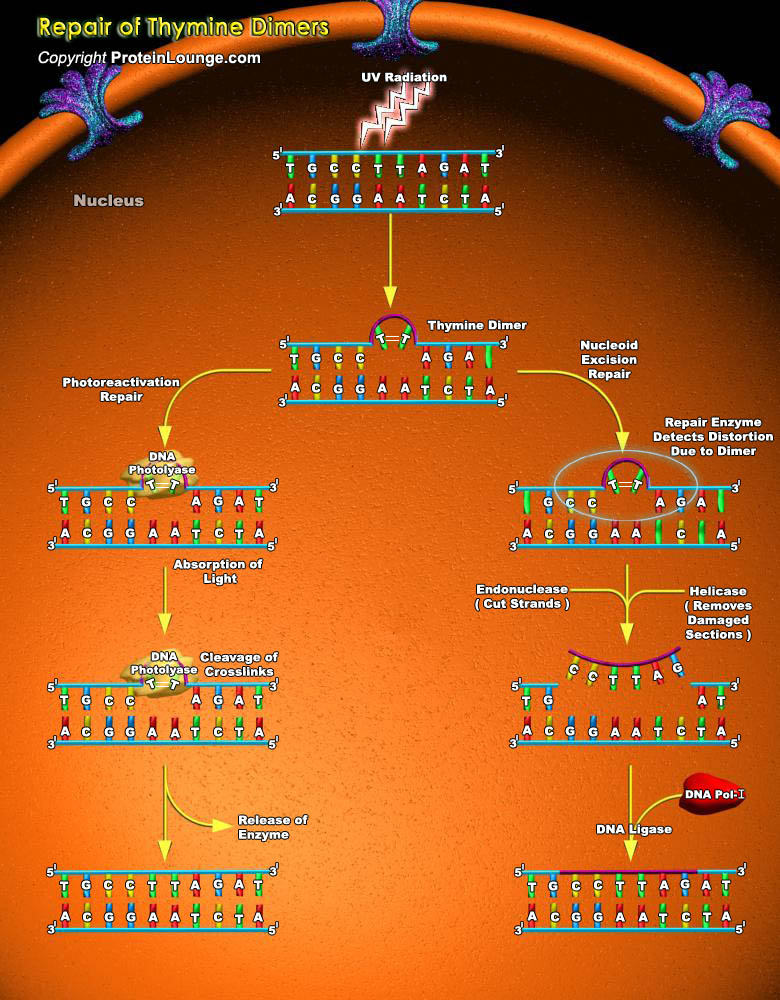
Irradiation of DNA by UV (Ultraviolet light) causes lesions, such as Cyclobutane-Pyrimidine Dimers or 6-4PPs (6-4 Pyrimidine Pyrimidone). The most common covalently linked adjoining pyrimidines are T-T (Thymine dimers), T-C (Thymine-Cytosine dimers) and C-C (Cytosine-Cytosine dimers). T-T dimers cause kinks in the DNA strand that prevent both replication and transcription of that part of the DNA. Because they block DNA replication (and therefore prevent cells from reproducing), T-T dimers and other forms of UV damage cannot be inherited, and thus do not constitute mutations. Such kinds of DNA damage are known as premutational lesions because they prevent both transcription and replication of the genes in which they are present and these lesions are fatal if they go[..]
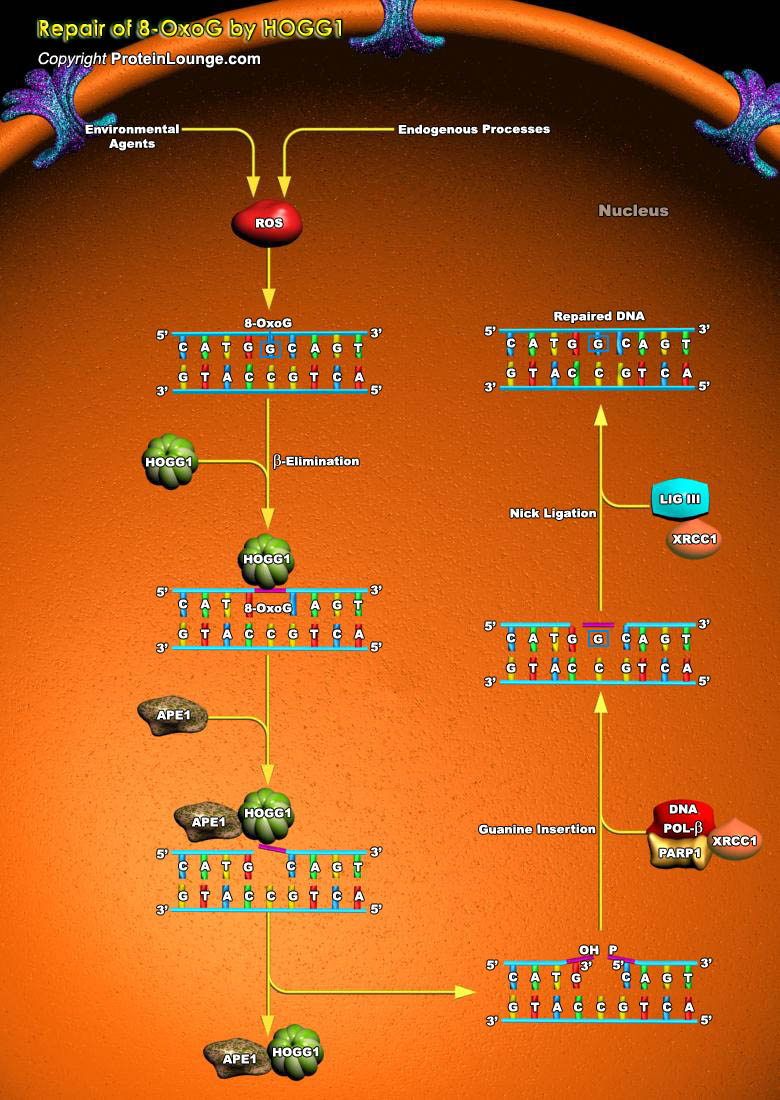
8-oxo-7,8-dihydroguanine (8-oxoG) is a pre-mutagenic DNA lesion that is formed by the oxidation of guanine. Reactive oxygen species (ROS) that is primarily responsible for the damage of guanine is produced as a result of several endogenous processes or environmental agents. ROS can cause damage to cellular macromolecules like proteins, lipids and nucleic acids. Proteins, proteins, lipids and RNA having oxidative damage are generally degraded and recycled, whereas DNA lesions like 8-oxoG need to be repaired to maintain genomic integrity (Ref.1 and 2). 8-oxoG is generally repaired in human through base excision repair (BER) pathway. The enzyme human 8-oxoG DNA glycosylase 1 (HOGG1) is the primary enzyme that initiates the repair of 8-oxoG. OGG1 initiated BER[..]

