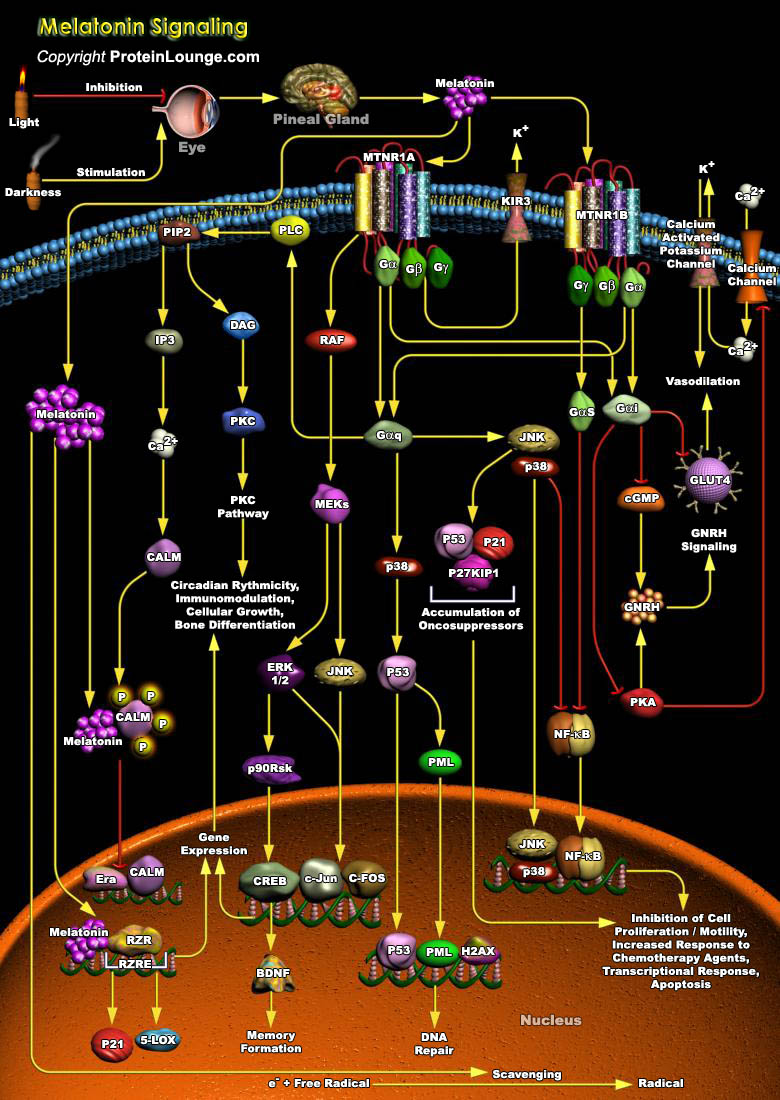
In mammals, the circadian system is comprised of three major components: the lateral eyes, the hypothalamic SCN (Suprachiasmatic Nucleus) and the pineal gland. The SCN harbours the endogenous oscillator that is entrained everyday to the ambient lighting conditions via retinal input. Among the many circadian rhythms in the body that are driven by SCN output, the synthesis of Melatonin (N-Acetyl-5 Methoxy-Tryptamine) in the pineal gland functions as a hormonal message encoding for the duration of darkness. Melatonin is a hormone secreted mainly by the pineal gland or epiphysis; it is also produced, but in much smaller quantities, by the retina. Melatonin is produced nocturnally, and is a neurochemical representation of time. Dissemination of this circadian[..]
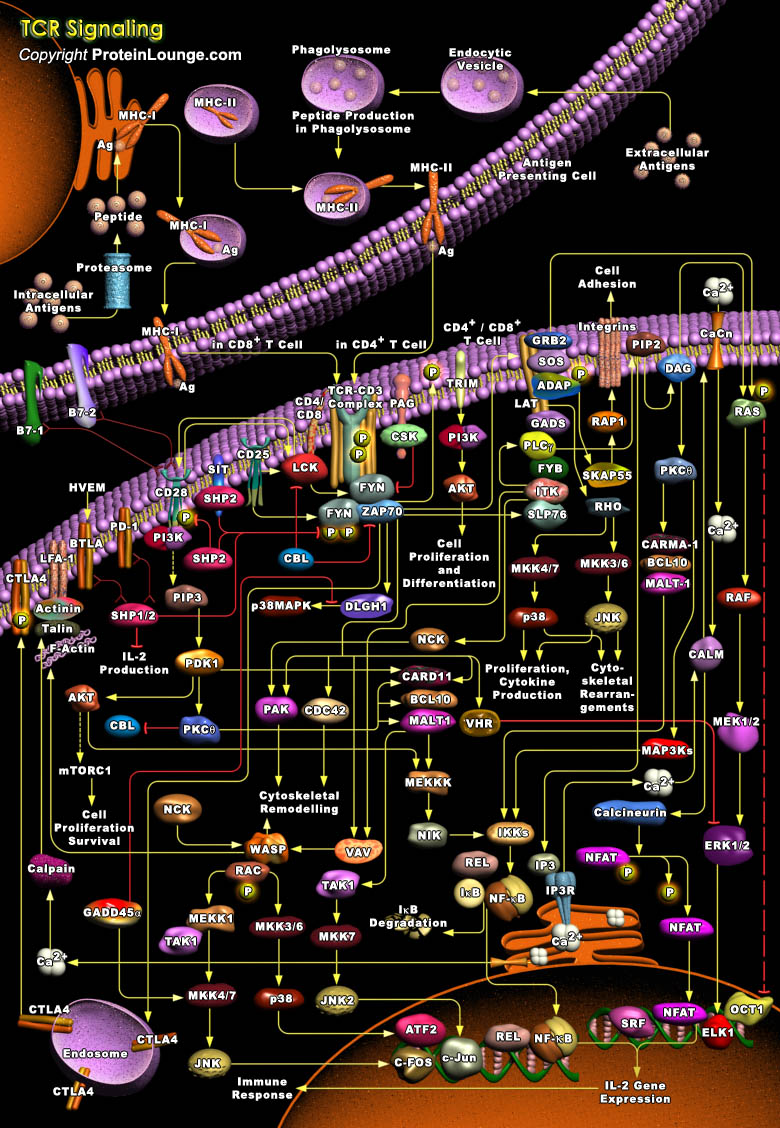
T-Cells are a subset of lymphocytes that play a large role in the immune response. The TCR (T-Cell Receptor) is a complex of integral membrane proteins that participates in the activation of T-Cells in response to the presentation of antigen. Stimulation of TCR is triggered by MHC (Major Histocompatibility Complex) molecules on Antigen Presenting Cells that present antigen peptides to TCR complexes and induce a series of intracellular signaling cascades. Engagement of the TCR initiates positive (signal-enhancing) and negative (signal-attenuating) cascades that ultimately result in cellular proliferation, differentiation, cytokine production, and/or activation-induced cell death. These signaling cascades regulate T-Cell development, homeostasis, activation, acquisition[..]
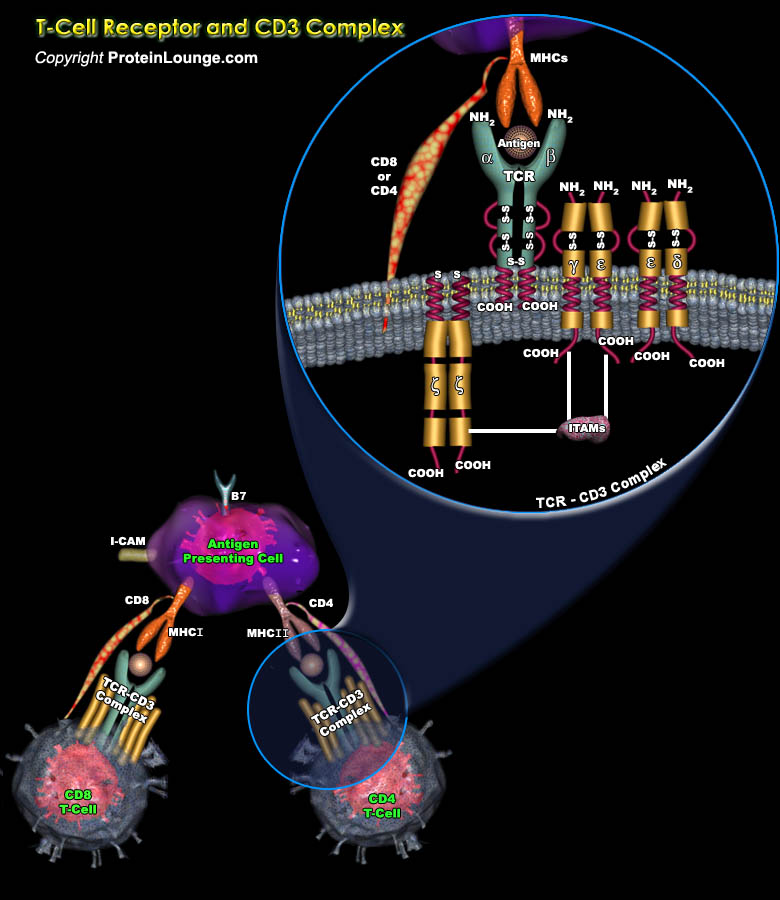
The lymphocyte population is mainly made up of the T-lymphocytes (Thymus-Derived Lymphocytes), B-lymphocytes (bone-marrow-derived), and the NK cells (Natural-Killer Cells). T-lymphocytes mediating the cellular immunity, along with B lymphocytes mediating humoral immunity, provide adaptive immunity, which work in close collaboration with the innate immune system. B-lymphocytes mature in the bone marrow itself, while the T-lymphocytes require the thymus to mature, before being deployed to the peripheral lymphoid organs for further antigen-mediated differentiation. These cells are responsible for antibody production, direct cell-mediated killing of virus-infected and tumor cells, and regulation of the immune response (Ref.1).T cells are key mediators of the adaptive[..]
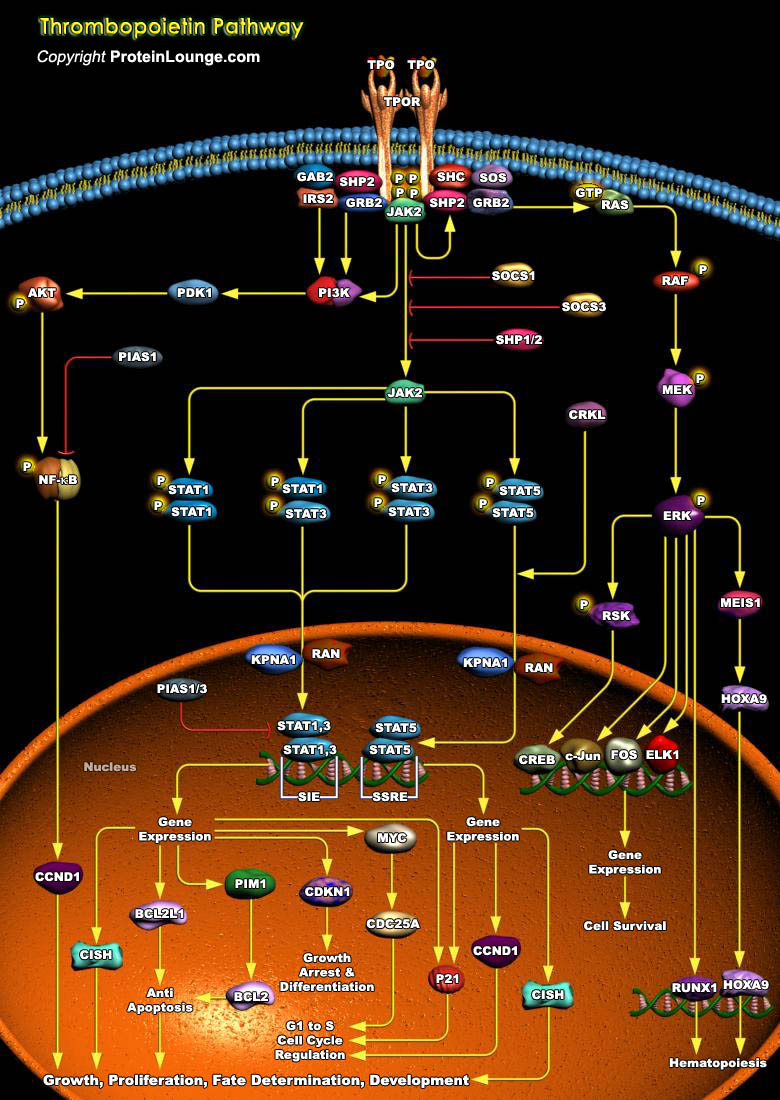
Throughout adult life, all blood cells are constantly regenerated from a small pool of hematopoietic stem cells. A single purified stem cell injected into a lethally irradiated host is sufficient to reconstitute all lineages, demonstrating pluripotency and an enormous regenerative potential. Genes encoding secreted growth factors, signal transduction molecules and nuclear proteins regulate this process. The helical cytokine family comprises structurally related secreted proteins that play an important role as hematopoietic growth factors. Their cognate receptors are single transmembrane proteins that constitute the so-called cytokine receptor superfamily. One such ligand-receptor pair is TPO (Thrombopoietin), also known as the c-MPL ligand or MGDF (Megakaryocyte Growth[..]
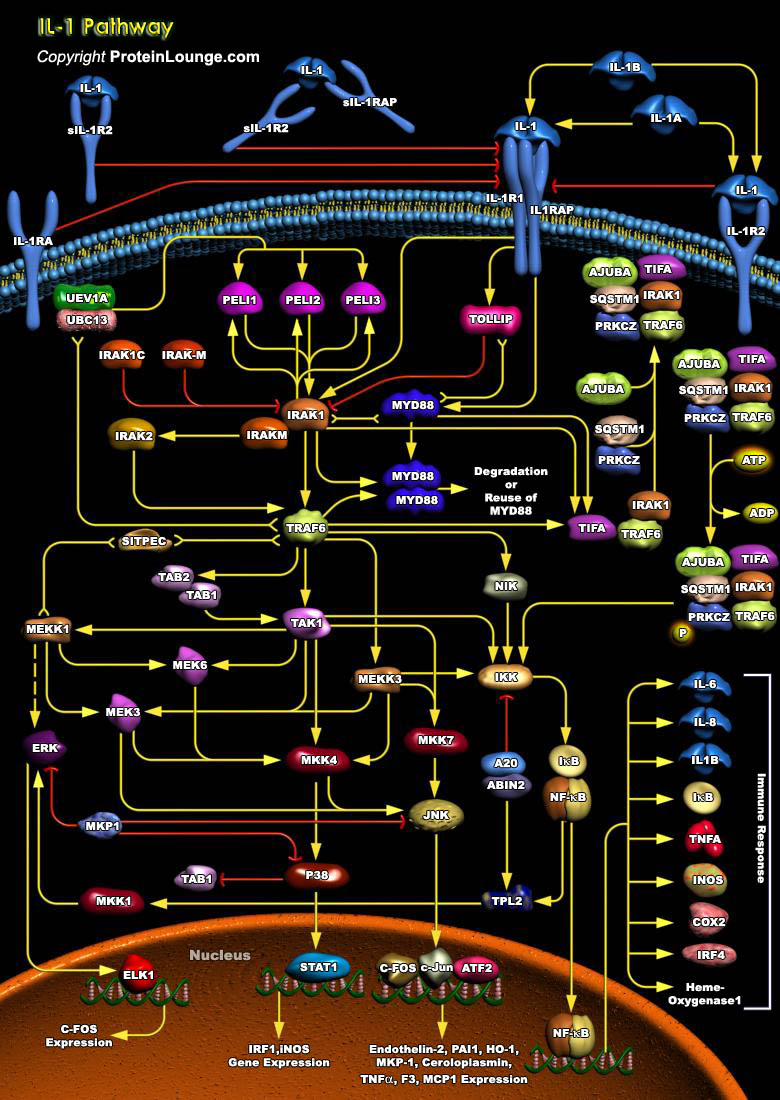
IL-1 (Interleukin-1) is a proinflammatory cytokine that stimulates a broad spectrum of immune and inflammatory responses. IL-1 is produced by activated macrophages, endothelia cells, B-Cells, and fibroblast cells. It induces inflammatory responses, edema, promotes the production of Prostaglandins, IL-2, and the growth of leukocytes (Ref.1). There are two forms of IL-1 encoded by distinct genes, IL-1 Alpha and IL-1 Beta. IL-1 Beta is produced as a 269 amino acid precursor that is cleaved by ICE (IL-1Beta Converting Enzyme) to the active IL-1 Beta form that is secreted. IL-1 induces cellular response through two subunits of its receptor, IL-1R1 (IL-1 Receptor1) and IL-1RAcP (IL-1 Receptor Accessory Protein). When it binds to its cell-surface receptor, IL-1 initiates a[..]
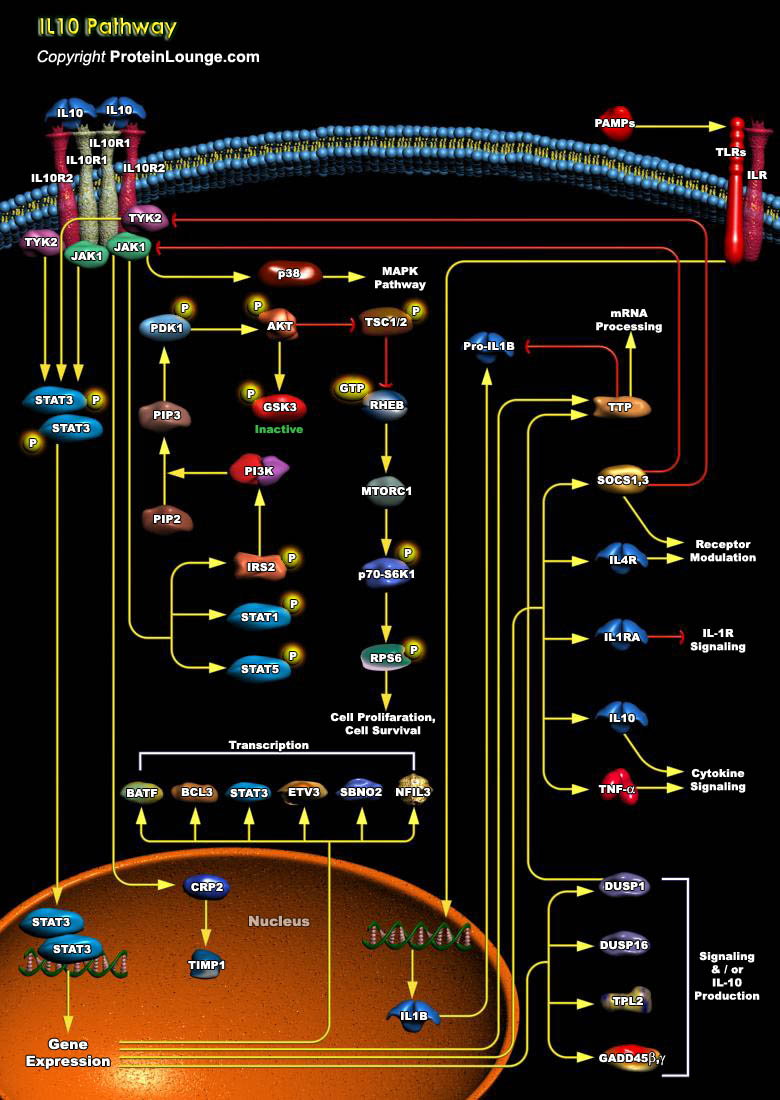
IL-10 (Interleukin-10) is a pleiotropic cytokine with important immunoregulatory functions whose actions influence activities of many of the cell-types in the immune system. It is a cytokine with potent anti-inflammatory properties, repressing the expression of inflammatory cytokines such as TNF-Alpha (Tumor Necrosis Factor-Alpha), IL-6 (Interleukin-6) and IL-1 (Interleukin-1) by activated macrophages (Ref.1). Functional IL-10R (IL-10 Receptor) complexes are tetramers consisting of two ligand-binding subunits (IL-10R-Alpha or IL-10R1) and two accessory signaling subunits (IL-10R-Beta or IL-10R2). Binding of IL-10 to the extracellular domain of IL-10R1 activates phosphorylation of the receptor-associated, JAK1 (Janus Kinase-1) and TYK2 (Tyrosine Kinase-2), which are[..]
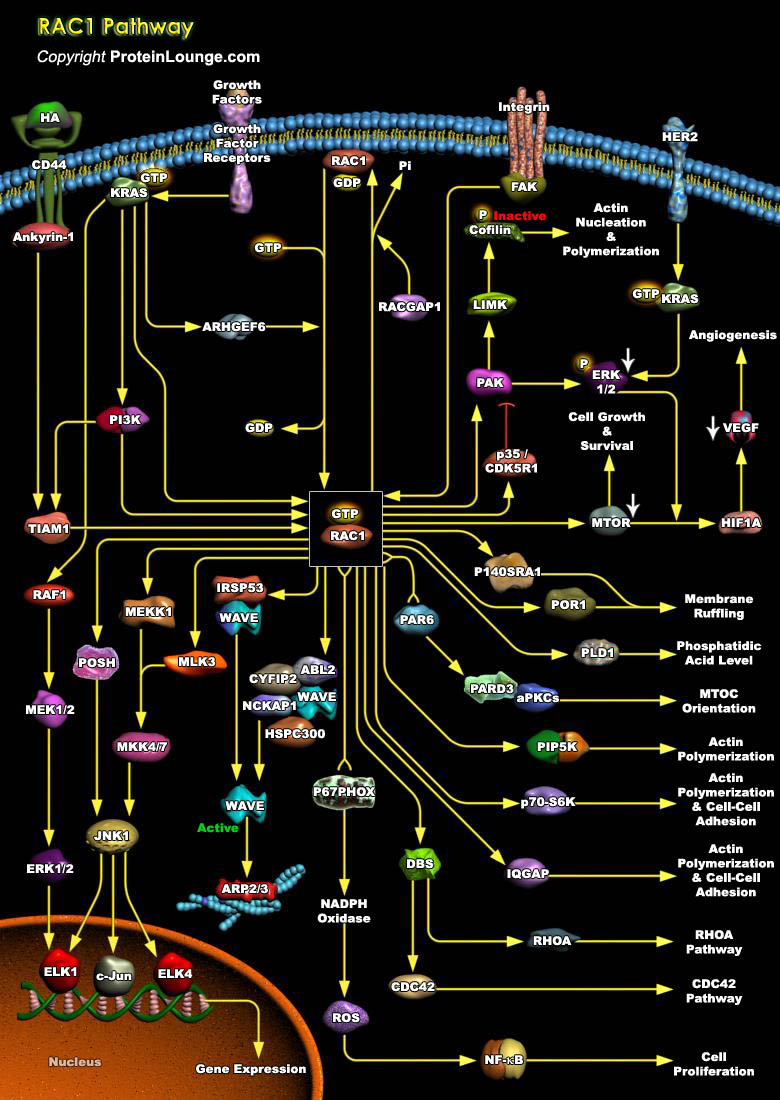
To achieve strong adhesion to their neighbors and sustain stress and tension, epithelial cells develop many different specialized adhesive structures. Breakdown of these structures occurs during tumor progression with the development of a fibroblastic morphology characteristic of metastatic cells. Adhesion receptors of the Cadherin family have been implicated in these cellular processes, which play an important role in the development and maintenance of the differentiated epithelial phenotype during organogenesis and adult life. Cadherin-mediated adhesion requires the activity of the cytosolic proteins of the Rho subfamily members, Rho, Rac and CDC42 (Cell Division Cycle-42). They belong to the Ras Superfamily of small GTPases, whose function is regulated[..]
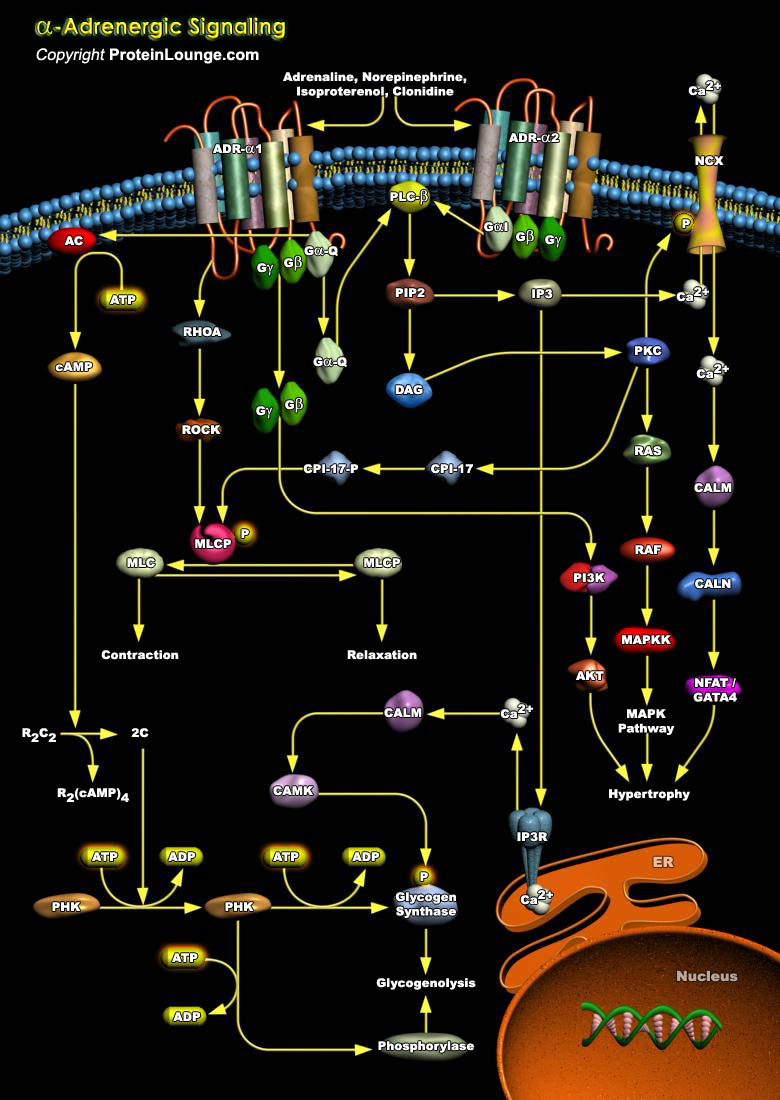
ADRs (Adrenergic Receptors) are expressed on virtually every cell type in the body and are the receptors for Adrenaline, Epinephrine and Norepinephrine within the Sympathetic Nervous System. They serve critical roles in maintaining homeostasis in normal physiologic settings as well as pathologic states. These receptors are also targets for therapeutically administered agonists and antagonists (Ref.1). ADRs are members of the super family of cell surface receptors that carry out signaling via GPCR (G-Protein Coupled Receptors) and are divided into nine distinct subtypes: ADR-Alpha1A, ADR-Alpha1B, ADR-Alpha1D, ADR-Alpha2A, ADR-Alpha2B, ADR-Alpha2C, ADR-Beta1, ADR-Beta2 and ADR-Beta3. ADR-Alpha2 is implicated in diverse physiological functions particularly of the[..]
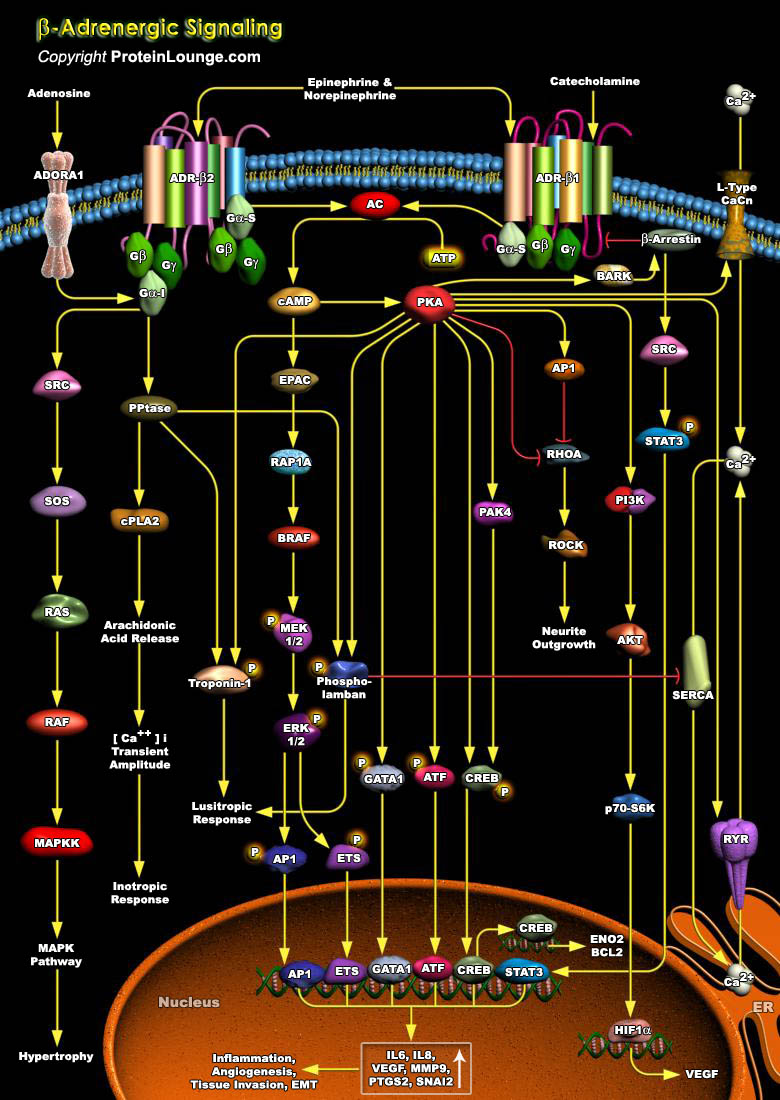
The rate and strength of beating of the heart is under the reciprocal control of the Adrenergic (sympathetic) and Cholinergic (parasympathetic) systems. Increased strength (inotropy) in cardiac beating in response to hormones like the blood-borne Epinephrine or to neurally delivered Norepinephrine is mediated by ADR-Beta (Beta-Adrenergic Receptors) , which are members of the superfamily of cell surface receptors that carry out signaling via coupling to G-proteins (Guanine nucleotide binding proteins)(Ref.1). ADR forms the interface between the endogenous Catecholamines, Adrenaline, Epinephrine and Norepinephrine and a wide array of target cells in the body to mediate the biological effects of the Sympathetic Nervous System. They serve critical roles in maintaining[..]
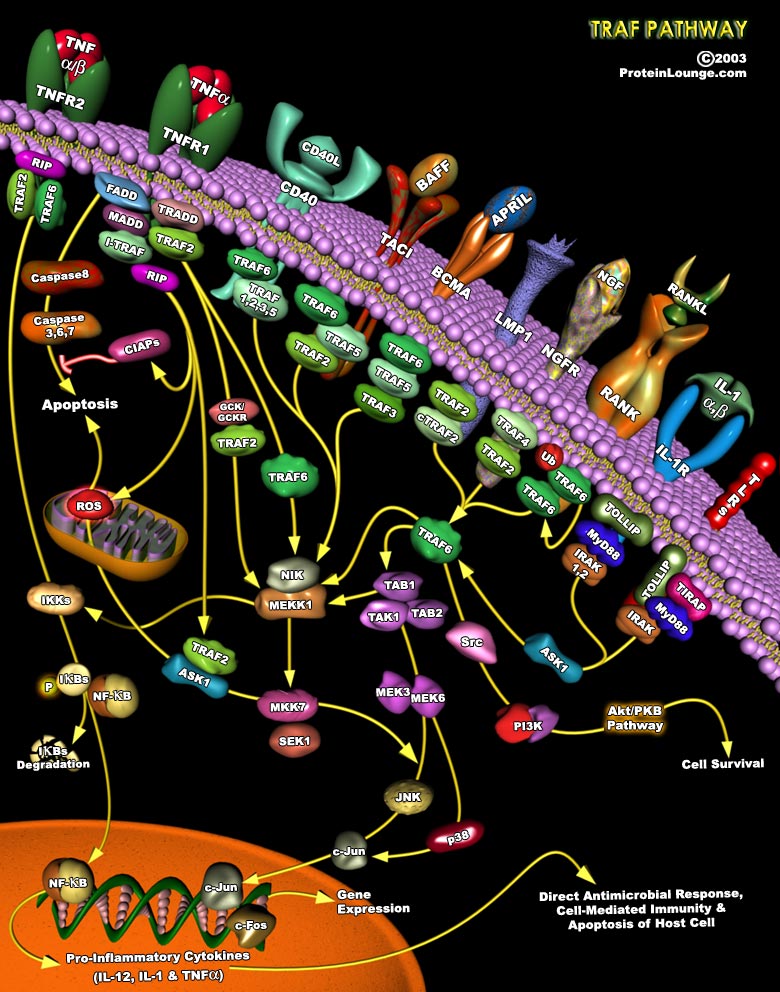
The structural and metabolic integrity of bone is maintained through the dynamic process of bone remodeling those results from the coordinate action of bone resorption and the formation of new bone by osteoblasts. Regulation of bone remodeling occurs through multiple mechanisms that ultimately converge on the interaction of osteoclasts or their precursors with osteoblasts and bone marrow stromal cells. Two key factors supplied by the stromal environment are CSF1 (Colony-Stimulating Factor-1) and the TNF family member, RANKL (Receptor Activator of Nuclear Factor-KappaB Ligand, also called TRANCE, ODF, OPGL). Signaling through RANK is essential for the differentiation and activation of osteoclasts, the cell principally responsible for bone resorption. RANK provokes[..]
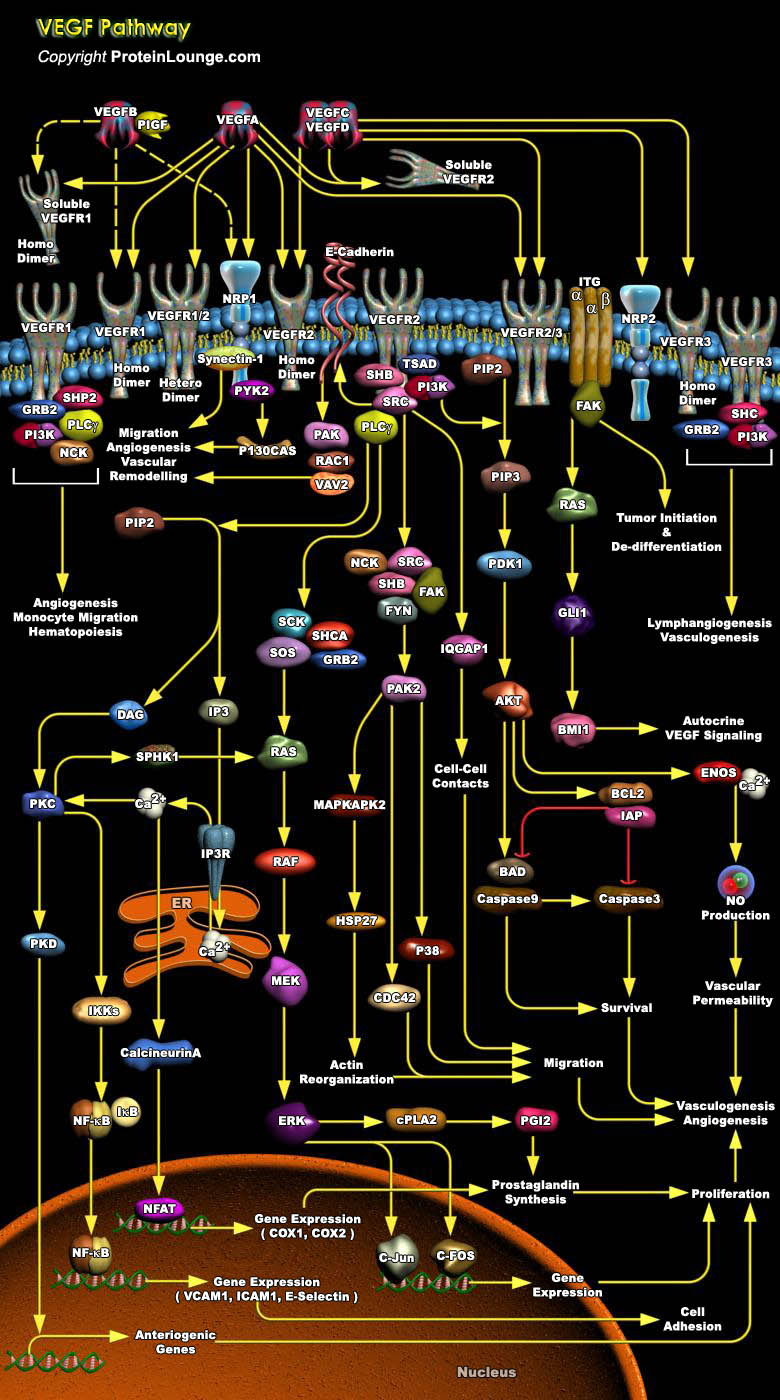
The vascular endothelial growth factor (VEGF) family of soluble protein growth factors is master regulators of in vasculogenesis and angiogenesis during blood vessel development. In mammals, the VEGF family consists of 5 members, VEGFA, B, C, and D and placenta growth factor (PIGF). VEGFs act through three structurally related VEGF receptor tyrosine kinases, denoted VEGFR1 (FLT1), VEGFR2 (FLK1), and VEGFR3 (FLT4). Binding of VEGF to its cognate VEGF receptor in cis or trans (e.g., by binding HS proteoglycans [HSPGs] on adjacent cells induces receptor homo- or heterodimerization (Ref.1 and 2). VEGFA binds VEGFR1 and VEGFR2. VEGFR2 is expressed mainly in endothelial cells, whereas VEGFR1 is expressed in endothelial cells as well as hematopoietic[..]
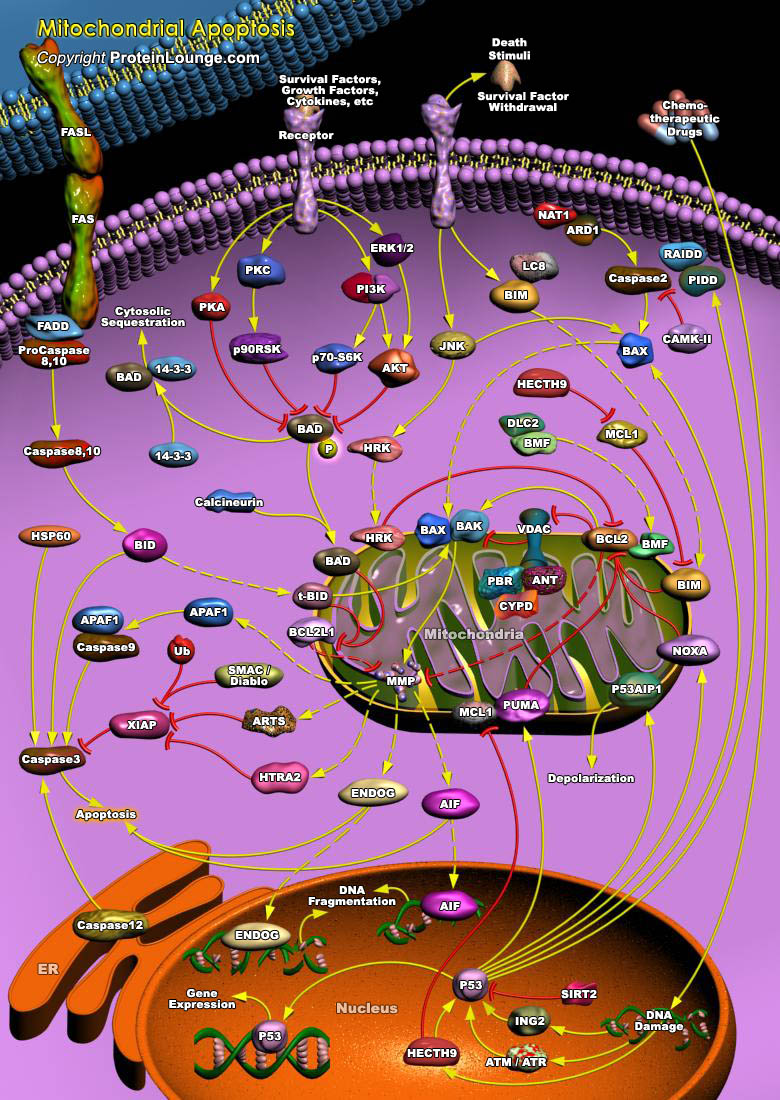
Apoptosis is a naturally occurring process by which a cell is directed to Programmed Cell Death. Apoptosis is based on a genetic program that is an indispensable part of the development and function of an organism. In this process, cells that are no longer needed or that will be detrimental to an organism or tissue are disposed of in a neat and orderly manner; this prevents the development of an inflammatory response, which is often associated with Necrotic cell death. There are at least two broad pathways that lead to Apoptosis, an "Extrinsic" and an "Intrinsic" Pathway. In both pathways, signaling results in the activation of a family of Cys (Cysteine) Proteases, named Caspases that act in a proteolytic cascade to dismantle and remove the dying cell. The extrinsic[..]

